Research | Open Access | Volume 9 (1): Article 36 | Published: 26 Feb 2026
Factors associated with tuberculosis in cattle in the urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Table 1: Knowledge of livestock farmers in urban and peri-urban areas about zoonoses and bovine tuberculosis in particular, Ouagadougou, Burkina Faso, 2021 | ||
|---|---|---|
| Questions | Frequency | Percent (%) |
| Possible transmission of pathogens from animals to humans | ||
| Yes | 17 | 70.8 |
| No | 7 | 29.2 |
| Brucellosis and tuberculosis as examples of zoonoses | ||
| Yes | 7 | 29.2 |
| No | 17 | 70.8 |
| Pathogen transmission from animals to humans | ||
| Contact with infected animals | 14 | 58.3 |
| No known mode | 10 | 41.7 |
| Exposure to zoonotic pathogens due to livestock activity | ||
| Yes | 20 | 83.3 |
| No | 4 | 16.7 |
| Methods for preventing the transmission of zoonotic pathogens from animals | ||
| Reduced contact with animals | 1 | 4.2 |
| Don’t know | 23 | 95.8 |
| Ever heard of bovine tuberculosis? | ||
| Yes | 23 | 95.8 |
| No | 1 | 4.2 |
| Ever heard of human tuberculosis? | ||
| Yes | 21 | 87.5 |
| No | 3 | 12.5 |
| Have you ever seen an animal with tuberculosis? | ||
| Yes | 0 | 0 |
| No | 24 | 100 |
| Do you know the symptoms of animal tuberculosis? | ||
| Don’t know | 24 | 100 |
| How is tuberculosis transmitted between cattle? | ||
| Contact between animals | 6 | 25.0 |
| Don’t know | 18 | 75.0 |
| How is bovine tuberculosis transmitted to humans? | ||
| In contact with infected animals | 3 | 12.5 |
| Consumption of infected animal products | 6 | 25.0 |
| Don’t know | 15 | 62.5 |
| Risk of exposure to bovine tuberculosis after consuming raw milk from infected animals | ||
| No | 7 | 29.2 |
| Yes | 17 | 70.8 |
| Risk of exposure to bovine tuberculosis after eating rare meat from infected animals | ||
| No | 16 | 66.7 |
| Yes | 8 | 33.3 |
Table 1: Knowledge of livestock farmers in urban and peri-urban areas about zoonoses and bovine tuberculosis in particular, Ouagadougou, Burkina Faso, 2021
| Table 2: Distribution of bovine tuberculosis cases by location on farms in the urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021 | ||||
|---|---|---|---|---|
| Communes | Total animals tested | Positive | Prevalence (%) | 95% CI (%) |
| Borough 2/Ouaga | 49 | 3 | 6.12 | [0–12.84] |
| Komsilga | 52 | 17 | 32.69 | [19.94–45.44] |
| Koubri | 52 | 5 | 9.61 | [1.60–17.63] |
| Saaba | 53 | 5 | 9.43 | [1.56–17.30] |
| Kompi-Ipala | 53 | 3 | 5.66 | [0–11.88] |
| Pabre | 56 | 3 | 5.35 | [0–11.25] |
| Tanghin-Dassouri | 56 | 7 | 12.5 | [3.84–21.16] |
| Total | 371 | 43 | 11.59 | [8.75–15.25] |
Table 2 : Distribution of bovine tuberculosis cases by location on farms in the urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021
| Table 3: Results of multivariate analysis of factors associated with bovine tuberculosis in herds in urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021 | ||
|---|---|---|
| Variables | ORa (95% CI) | P-value |
| Grazing (having at-risk grazing neighbors) | ||
| No (ref.) | 1 | |
| Yes | 1.70 [0.41–7.01] | 0.46 |
| Type of production system | ||
| No (ref.) | 1 | |
| Extensive | 2.78 [0.73–11.98] | 0.62 |
| Contact between cattle and other domestic animals | ||
| No (ref.) | 1 | |
| Yes | 3.04 [1.30–10.74] | 0.003 |
| Buying cattle from high-risk farms | ||
| No (ref.) | 1 | |
| Yes | 2.80 [1.04–10.60] | 0.04 |
| Sharing with animals from other farms | ||
| No (ref.) | 1 | |
| Yes | 2.06 [1.09–8.59] | 0.18 |
| Proximity to cattle | ||
| No (ref.) | 1 | |
| Yes | 2.81 [0.09–17.11] | 0.080 |
| Farm size | ||
| No (ref.) | 1 | |
| Yes | 1.66 [0.31–7.42] | 0.29 |
| Animal body condition | ||
| Good (ref.) | 1 | |
| Poor | 1.76 [0.29–12.03] | 0.98 |
| Education level | ||
| School children (ref.) | 1 | |
| Out of school | 2.50 [0.11–11.54] | 0.43 |
| Knowledge of zoonoses and bovine tuberculosis | ||
| Good (ref.) | 1 | |
| Poor | 1.63 [0.32–8.22] | 0.54 |
Table 3 : Results of multivariate analysis of factors associated with bovine tuberculosis in herds in urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021
Figures
Keywords
- Associated factors
- Prevalence
- Tuberculosis
- Burkina Faso
Djenebou Diakité1,2,&, Laibané Dieudonné Dahourou3, Djibril Barry2, Pauline Kiswendsida Yanogo2, Yoda Hermann2, Jéan Kaboré4, Brice Wilfrid Bicaba5, Nicolas Meda2 , Lamouni Habibata Zerbo6, Samba Thioub7, Lalidia Bruno Ouoba6, Sayouba Ouédraogo8, Désiré Nana9
1Ministry of Agriculture, Animal Resources and Fisheries, Directorate General of Veterinary Services, Ouagadougou, Burkina Faso, 2Burkina Field Epidemiology Laboratory Training Program/University Joseph Ki-Zerbo, Ouagadougou, Burkina Faso, 3Livestock Department, Institute of Environmental Sciences and Rural Development, University of Dedougou, Burkina Faso, 4Institute for Research in Health Sciences, National Center for Scientific and Technological Research, Ouagadougou, Burkina Faso, 5Ministry of Health, Management of the Health Emergencies Response Operations Center, Ouagadougou, Burkina Faso, 6Ministry of Agriculture, Animal and Fishery Resources, National Livestock Laboratory, Ouagadougou, Burkina Faso, 7Tambacounda Health District, Ministry of Health and Social Action, Tambacounda, Senegal, 8Management of the VET-Consulting Veterinary Clinic and Pharmacy Complex Rural town of Koubri, Burkina Faso, 9Center for International Cooperation in Agronomic Research for Development, University of Montpellier, France
&Corresponding author: Diakité Djenebou, Ministry of Agriculture, Animal Resources and Fisheries, Directorate General of Veterinary Services, Ouagadougou, Burkina Faso, Email: diakitdjnbou@yahoo.fr , ORCID: https://orcid.org/0009-0001-3165-175X
Received: 20 Jan 2025, Accepted: 24 Feb 2026, Published: 26 Feb 2026
Domain: One Health
Keywords: Associated factor, prevalence, tuberculosis, Burkina Faso
©Diakité Djenebou et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Diakité Djenebou et al., Factors associated with tuberculosis in cattle in the urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):36. https://doi.org/10.37432/jieph-d-25-00029
Abstract
Introduction: Bovine tuberculosis is endemic in Burkina Faso. It is a chronic zoonosis with a significant health and economic impact. Studies carried out on both sides of the country show the existence of bovine tuberculosis with a high prevalence ranging from 3.75 to 27.7% from 2004 to 2018. However, the factors associated with the circulation of the disease are poorly documented, while A better understanding of the risk factors for infection is necessary for its eradication. This study assessed the factors associated with tuberculosis on farms in the study area in 2021.
Methods: This was a questionnaire-based cross-sectional analytical study focusing mainly on environmental factors, livestock management and animal transmission. In total, 371 cattle were subjected to comparative intradermal tuberculin testing. Data were collected using Microsoft Excel® 2016 software, then imported into Epi info® 7.2.2 for analyses.
Results: The overall prevalence of bovine tuberculosis was 11.59% (95%CI: 8.75 -15.25). In multivariate analysis after adjustment, purchase of cattle from at-risk farms (aOR: 2.80, 95%CI: 1.04 -10.60, p = 0.04) and contact of cattle with other domestic animals (aOR=3.04, 95%CI: 1.30-10.74, p=0.003) remained significantly associated with developing bovine tuberculosis among the cattle.
Conclusion: Factors independently associated with bTB on farms in the study area are the purchase of cattle from at-risk farms and contact between domestic cattle. In view of these results, it is important to implement eradication measures, namely prevention, monitoring and control in our farms.
Introduction
Bovine tuberculosis is a chronic infectious disease caused by a bacterium called Mycobacterium bovis and, in some cases, Mycobacterium tuberculosis [1–3]. It is a zoonotic disease with a major impact on animal productivity, trade and commerce, and a threat to animal and public health [4,5]. This is an important zoonosis that has been notifiable to the World Organization for Animal Health (OIE) since 1964, in accordance with the OIE Terrestrial Animal Health Code [1]. All countries and territories are affected [1,6]. In some countries, infections caused by Mycobacterium bovis are estimated to account for up to 10% of tuberculosis cases in humans [1]. In 2016, according to WHO estimates, 147,000 new cases of zoonotic tuberculosis were reported in humans, and there were 12,500 human deaths due to the disease [7]. Bovine tuberculosis is endemic in Africa [3, 8, 9] in herds that supply meat and milk to the population.
In Burkina Faso, cattle farming produces over 30 million tonnes of beef and 264 million tonnes of milk per year [10]. Studies show the existence of bovine tuberculosis with a high prevalence ranging from 3.75 to 27.7% from 2004 to 2018[11–13] . In addition to being a health problem, bovine tuberculosis also has an economic impact, as it mainly leads to production losses on farms and seizures at slaughterhouses [1]. The cohabitation of humans and livestock in urban and peri-urban areas is not without risk to public health [8]. Although required by legislation, the application of the classic “test and slaughter” method of bovine tuberculosis control is not very effective. The prompt identification of the factors associated with the circulation of the bovin tuberculosis is required to guide effective implementation of the control measures and ensure eradication of the disease. Researchers have reported the link between the environment, farming methods, the animal and the specific characteristics of the pathogen [9,14]. We therefore investigated the factors associated with bovine tuberculosis in herds in the urban and peri-urban areas of Ouagadougou in Burkina Faso in 2021.
Methods
Study design and population
This was an analytical cross-sectional study conducted from November 01 to December 31, 2021. To be included, the farm owners must be available to respond to the survey and grant access to the farm for a clinical examination of the animals and the performance of the intradermal tuberculin test, as well as the proximity of the farm to the vaccination park, were required. Sick cattle and those located outside the study area were not included in the study.
Study area
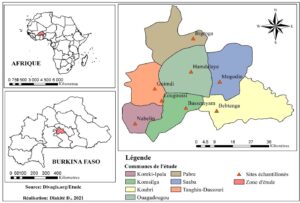
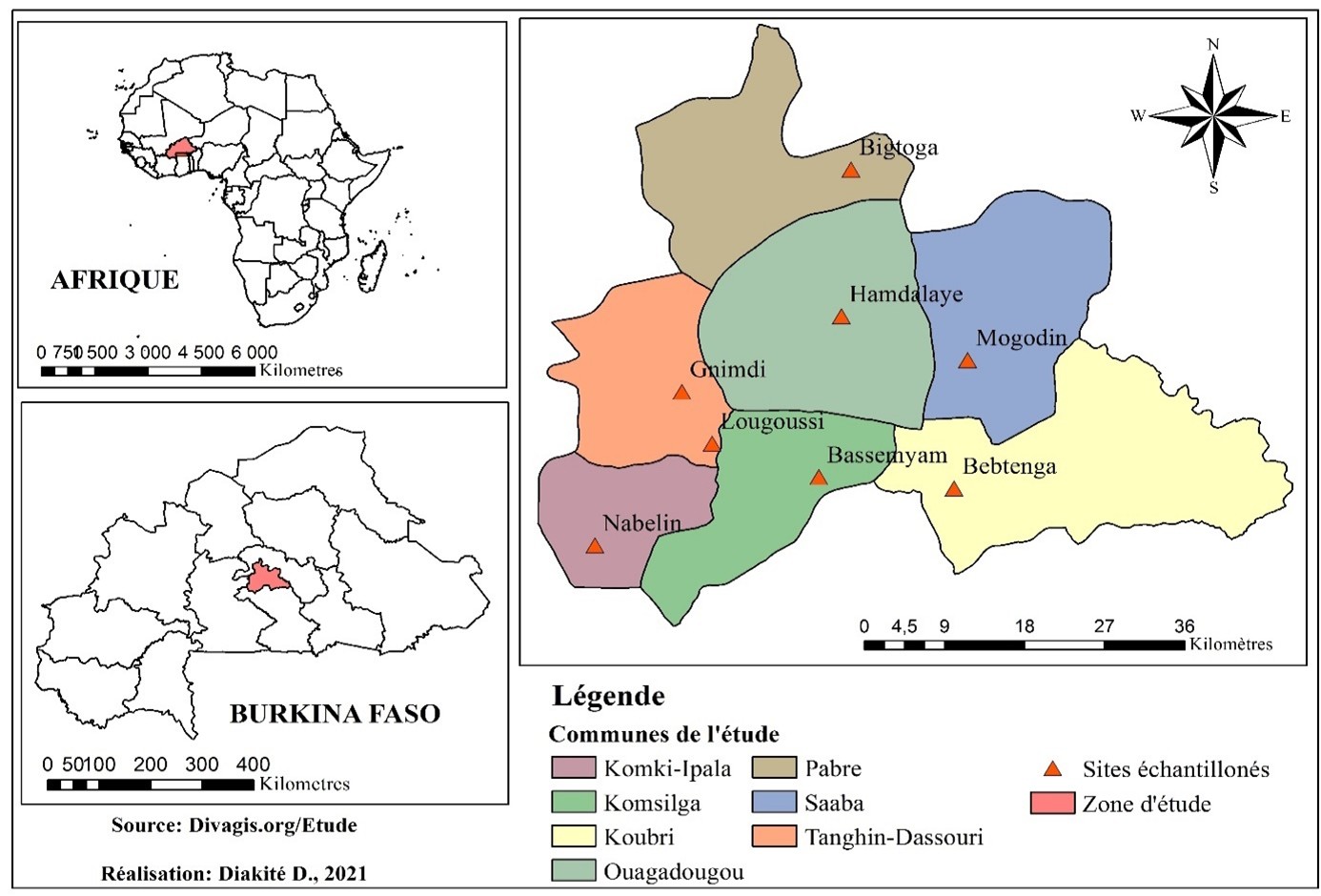
The study was conducted in Burkina Faso in West Africa. The country has a surface area of 270,200 km² and an estimated population of 20,487,979 in 2019[14]. Burkina Faso is divided into 13 administrative regions. The study was carried out in the Centre region, which comprises a single province subdivided into seven communes. This region is home to the capital Ouagadougou, with a population of 2,453,496 in 2019[14]. In 2020, the national cattle herd was estimated at around 10,300,000 head, including 177,000 head in the Centre region [15]. Current production systems are intensive, semi-intensive and extensive. Farmers practice fattening and dairy production on a family and commercial scale. This is an area of high milk production (Figure 1).
Sampling
Cattle breeders and cattle were sampled. Breeders were selected at random on the basis of the list of breeders drawn up by the heads of the livestock technical support zones (ZATE) and their proximity to a vaccination park. On each selected farm, simple random sampling was used to select the animals to be included in the study. On each farm, only healthy cattle over six months old were selected. The total herd size for the Central region of BF was 177,000 head of cattle in 2020.
We calculated our sample size using Open Epi® version 3 software. An expected prevalence of 6.05% [12], a precision of 5%, a calculation factor of 2 and a power of 95% were considered. The minimum sample size calculated was 87±9 (i.e. 10% of animals in which the result of the comparative intradermal tuberculin test could not be read) = 96 cattle. To improve the accuracy of the study, 371 cattle from 24 farms were registered for this study.
Data collection
A questionnaire was used to collect data on farmers’ knowledge. The questionnaire was administered in French or the local language during a face-to-face interview. To collect data on farm management and individual data on the animals to be analysed, a questionnaire was drawn up and administered to the farmers. To determine the tuberculosis status of the animals, we performed a comparative intradermal tuberculin test, based on the in vivo demonstration of a delayed hypersensitivity reaction following intradermal injection of avian and bovine tuberculins into the animal.
The comparative intradermal tuberculin test consists of injecting bovine (B) and avian (A) tuberculins into the dermis of the neck at two points 12-15 cm apart on day zero (D0), and assessing reactions at the injection points after 72 hours (D3) [16]. The results were interpreted according to the following rules for expressing individual results: for each animal that reacted, the thickening of the skin fold between the time intervals D0 and D3 was calculated. Let : ΔA =A3-A0 and ΔB =B3-B0, then look at ΔB :
- if ΔB is ≤ 2 mm, then the test is negative regardless of the difference (ΔB – ΔA).
- if ΔB is > 2 mm, then :
- if (ΔB – ΔA) >4 mm, the result is positive ;
- if (ΔB – ΔA) < 1 mm, the result is negative.
If 1 ≤ (ΔB – ΔA) ≤4 mm, the result is doubtful. [17]. That obtained for the thickening of the skin fold after injection of tuberculin B and that obtained by the difference between the thickenings of the folds after injection of tuberculins B and A [18].
Definition of key variables
We collected data on the following key variables, which were defined as follows:
Breeder: for the purposes of this work, we have indifferently referred to animal owners and keepers as breeders.
Pasture: land covered with grass and other low plants suitable for grazing animals, especially cattle or sheep [19].
Farm at risk: is a farm with an epidemiological link to an outbreak of bovine tuberculosis [20]. The status of the animals with regard to bovine tuberculosis is unknown.
Contact between domestic cattle: all interactions between cattle from one farm and those from other farms.
A high-risk tuberculosis pasture neighbour or high-risk tuberculosis farm: means a cattle farm (or other nearby livestock) with a high risk of transmission of bovine tuberculosis (Mycobacterium bovis) to another nearby farm, in particular due to: direct geographical proximity (adjoining pastures, pastures separated by a simple fence), frequent direct or indirect contact between animals (sharing water points, common crossings, insufficient barriers), an unfavourable health history, such as: recent or recurring cases of tuberculosis on the neighboring farm, suspicion or confirmation of tuberculosis without strict biosecurity measures in place.
High-risk tuberculosis grazing cattle or high-risk neighbour: cattle raised outdoors or on pasture in a geographical area where bovine tuberculosis is endemic or on the rise, and/or in direct or indirect contact with known reservoirs of the disease, such as infected wildlife (e.g. badgers, wild boar, deer), without sufficient biosecurity measures.
Bovine tuberculosis (caused by Mycobacterium bovis) can be transmitted in a variety of ways, and risk levels vary depending on environmental factors, husbandry practices, the species involved, and human interactions. The main risk levels for each transmission route are :
- Transmission between cattle (herd) — High risk
- Direct contact between animals, especially in enclosed or poorly ventilated spaces.
- Aerosols (respiratory droplets) are the primary route.
- Particularly high risk in the presence of an infected, shedding animal in a group.
- Interspecies transmission (wildlife/domestic animals) — Variable risk
- High risk if cohabiting with carrier species (badgers, deer, wild boar, etc.).
- Certain wild species can maintain a reservoir of the disease.
- Transmission through sharing water sources, food, or contaminated bedding.
- Transmission to humans (zoonosis) — Low to moderate risk
- Mainly through consumption of contaminated raw milk or unpasteurized dairy products.
- Less frequently, through inhalation of droplets or contact with open wounds.
- People at increased risk: farmers, veterinarians, slaughterers, and immunocompromised individuals.
- Environmental Transmission — Moderate Risk
- bovis can survive in the environment (soil, water, bedding) for several months depending on conditions.
- Moderate risk of indirect contamination, especially in poorly sanitised or highly populated farms.
- Transmission through human practices (equipment, transportation) — Low to Moderate Risk
- Risk of cross-contamination from undisinfected equipment, vehicles, or agents working on several farms.
- Diseases introduced through the purchase of untested animals or animals imported without quarantine.
Data management and analysis
The data collected was entered into Excel® 2016. They were then cleaned up, and any missing information was completed by contacting the owners again. Data from the Excel® 2016 spreadsheet were imported into Epi info® 7.2.2 for the various analyses. The analyses were carried out in two stages: descriptive and analytical. To assess farmers’ knowledge of zoonoses and bovine tuberculosis, scores ranging from zero to two were awarded, based on the completeness and accuracy of the farmers’ answers. A total of 14 questions were used for this exercise. Based on the accuracy and completeness of the answers given, knowledge scores were calculated. In total, when a breeder answered all the questions exactly, a maximum score of 20 was awarded. For a breeder to be considered knowledgeable about zoonoses, and bovine tuberculosis in particular, a minimum score of 12 out of 20 was required, which is equal to or greater than 60% according to the threshold of the Likert-type scale [21, 22].
In this respect, respondents were classified into two groups: satisfactory level of knowledge or unsatisfactory level of knowledge. The overall prevalence of bovine tuberculosis and the prevalence by commune were calculated as the ratio of the number of positive cases to the total number of animals tested by study area and by locality. Variations in prevalence by commune and the characteristics of intradermal tuberculin test-positive cattle were also presented.
The search for factors associated with bovine tuberculosis led to the performance of logistic regression (univariate and multivariate analysis) on complete data. For model fitting, independent variables associated with bovine TB infection with a p-value ≤ 0.20 were retained. Interactions between grazing (having at-risk grazing neighbours) and the other explanatory variables were tested in the final model. The grazing variable, being our main independent variable, regardless of its p-value of association with bovine tuberculosis, was included in all models. All other explanatory variables for bovine tuberculosis found in the literature were included in the model. The odds ratios and their confidence intervals, as well as the associated p-values, were estimated at the α <0.05 significance level.
Ethical considerations
The study was conducted in accordance with the principles of scientific research and animal welfare. At the same time, verbal authorisation to collect data in the field was also obtained from the provincial livestock directorate in the study area before the work was carried out. In order to obtain the consent of the study participants, the objectives of the work and the procedures were explained to them orally. This study also received the approval of the Joseph KI ZERBO University Animal Experimentation Ethics Committee (Reference Number CE-UJKZ/2020-03). The confidentiality of the data was ensured, as it was stored on a computer and password-protected. The identity of the breeders and any other participants in the study was kept anonymous.
Results
Farmers’ knowledge of zoonoses and bovine tuberculosis
In the study, 70.8%(17/24) of farmers were aware of the possibility of transmission of pathogens from animals to humans and vice versa. Also, 29.2% (7/24) identified bovine tuberculosis and brucellosis as examples of zoonoses. As for the mode of transmission of pathogens from animals to humans, 58.3% of farmers felt that contact with infected animals was a risk factor. Most farmers (83.3%) were aware that their activity exposed them to zoonotic pathogens. In addition, 95.8% (23/24) of farmers had heard of bovine tuberculosis. However, none of them had seen a bovine affected by bovine tuberculosis, and none knew the symptoms of tuberculosis in cattle (Table 1). Only 33.3% of the livestock farmers in the urban and peri-urban areas of Ouagadougou had satisfactory knowledge about zoonoses and bovine tuberculosis.
Prevalence of bovine tuberculosis
In this study, the overall prevalence of bovine tuberculosis was 11.6% (95% CI: 8.75 -15.25). Of the 24 farms, 17 had at least one positive animal, giving a herd prevalence of 70.8% (95% CI: 52.7%-89.0%). The distribution of prevalence by locality is shown in Table 2. The highest prevalence was recorded in the commune of Komsilga, at 32.7% (95% CI: 19.9-45.4). The 43 cattle that tested positive had the following characteristics: their age ranged from 5 to 10 years, 7 (16.3%) showed clinical signs such as cough, cold and emaciation, and 7 (30.2%) were of mixed breed.
Factors associated with bovine tuberculosis
In this study, having high-risk grazing neighbours was not significantly associated with bovine tuberculosis. In contrast, contact between domestic cattle (aOR = 3.04; 95% CI: 1.30 – 10.74) and the purchase of cattle from high-risk farms (aOR: 2.80; 95% CI: 1.04-10.60) were significantly associated with bovine TB infection (Table 3).
Discussion
The majority of farmers were aware of zoonoses and of bovine tuberculosis. Burkina Faso, like other countries, is facing an upward trend in zoonotic diseases. This level of information among farmers could probably be due to mass media campaigns on the risks of disease transmission from humans to animals, or vice versa. However, the level of knowledge specific to bovine tuberculosis was low. This gap creates a risk of exposure of farmers to animals carrying M. bovis. Our results are similar to those of Tschopp et al. 2015 in Ethiopia [23] et Sreeramareddy and al., 2013 in India [24] but contrary to Mohammed et al., 2021 in Malaysia [25]. For the prevention and control of bovine tuberculosis, it would be important to raise farmers’ awareness of bovine tuberculosis.
The purchase of cattle from at-risk farms is a factor associated with the transmission of bovine tuberculosis, since the introduction of animals increases the risk of bovine tuberculosis for a farm compared with a farm that does not purchase animals. This observation underlines the importance of herd contamination through the acquisition or keeping of animals. This result is similar to that found by Zehaira [29] in eastern Algeria in 2020 and Delafosse et. al, [14] in Chad in 2022. It would be prudent for farms to know the status of their herd in relation to bovine tuberculosis and to systematically screen every bovine animal they buy before introduction into their herd. Contact between domestic cattle is a factor associated with bovine tuberculosis due to the mode of transmission of bovine tuberculosis. The predominant mode of transmission of M. bovis is inhalation. Transmission is facilitated by prolonged contact between healthy and infected animals, so indirect transmission of M. bovis is strongly suspected between animals sharing the same territory and resources [20].
Many other domestic and wild animal species are susceptible to M. bovis and can act as reservoirs for the bacteria. This means that, in addition to transmission within a single farm, infection can spread to other farms, either through animal movements or by neighbouring an infected farm [20]. This result is similar to that found successively in 2014 and 2011 in Africa and France [6, 8, 30].
Limitations
In carrying out the study, we were faced with a number of limitations, such as insufficient financial resources, difficulties in acquiring tuberculin for the intradermal tuberculin test, and the reluctance of farmers to carry out the intradermal tuberculin test on their herds. These included the short time period devoted to this study, the fact that the study period coincided with the contagious bovine pleuropneumonia (CBPP) vaccination campaign in the field, and the acquisition of tuberculin for the CDI test.
Much of the information collected were self reported with the potential of social desirability bias. This primarily concerned the size of the herd and the origin of the animals, among other things. This situation would certainly have influenced the identification of factors associated with bTB in intra-urban and peri-urban herds of Ouagadougou in the BF in 2021. The questionnaires and data collection tools were administered in the local language help minimise this. Multivariate analysis was used to account for the confounding factors.
Conclusion
The factors independently associated with bovine tuberculosis on farms in the urban and peri-urban area of Ouagadougou, Burkina Faso, in 2021 were the purchase of cattle from high-risk farms and contact between domestic cattle. The prevalence of bovine tuberculosis in this area in 2021 was 11.59%. In view of our results, we recommend that full implementation of testing be carried out before the slaughtering of cattle for consumption. To improve the situation, we propose to carry out awareness-raising activities among farmers, so that they are better informed about bovine tuberculosis and more willing to participate in control effort.
What is already known about the topic
- Bovine tuberculosis is a zoonotic disease with a significant impact on animal productivity, animal trade and commerce, and a threat to animal and public health. So, in addition to being a health problem, the impact of bovine tuberculosis is also economic.
- In 2016, according to WHO estimates, 147,000 new cases of zoonotic tuberculosis were recorded in humans and there were 12,500 human deaths due to this disease. Bovine tuberculosis is endemic in Africa on farms which provide, among other things, meat and milk to populations.
- Studies carried out on both sides of Burkina Faso indicate the existence of bovine tuberculosis with a high prevalence varying between 3.75 to 27.7% from 2004 to 2018. But the factors linked to its spread are relatively few. documented.
What this study adds
- The results of this study provided recent data (2021) on the overall prevalence of bovine tuberculosis, 11.59% (95% CI: 8.75 -15.25) in urban and peri-urban areas of Ouagadougou in Burnina Faso.
- They also made it possible to know that the factors associated with the circulation of this disease which until then had been little documented are, among other things, contact between domestic cattle (ORa = 3.04; 95% CI: 1.30 – 10.74 ) and purchasing cattle from at-risk farms (aOR: 2.80; 95% CI: 1.04-10.60).
- In the study, 70.83% of breeders were aware of the possibility of transmission of pathogens from animals to humans and vice versa. Also, 29.16% identified bovine tuberculosis and brucellosis as examples of zoonosis.
Acknowledgements
The authors would like to express their deep appreciation and sincere thanks to the team of the provincial directorate of the Ministry of Agriculture, Animal and Fishery Resources of the Central region of Burkina Faso, to the training program in field epidemiology of Burkina Faso as well as all breeders.
Authors´ contributions
This study was carried out in an academic setting (obtaining a Master 2 diploma in epidemiology and field laboratory). D. Diakite, L. D. Dahouro and S. Thioub chose the theme of the study, validated the methodology, carried out field work, analyzed the data and wrote the manuscript. P.K. Yanogo, J. Kaboré, D. Barry and B. W. Bicaba contributed to the validation of the study protocol, supervised the various works and made a critical revision of the manuscript. L. H. Zerbo, L. B. Ouoba, S. Ouédraogo and D. Nana participated in data collection and critical revision of the manuscript.
| Table 1: Knowledge of livestock farmers in urban and peri-urban areas about zoonoses and bovine tuberculosis in particular, Ouagadougou, Burkina Faso, 2021 | ||
|---|---|---|
| Questions | Frequency | Percent (%) |
| Possible transmission of pathogens from animals to humans | ||
| Yes | 17 | 70.8 |
| No | 7 | 29.2 |
| Brucellosis and tuberculosis as examples of zoonoses | ||
| Yes | 7 | 29.2 |
| No | 17 | 70.8 |
| Pathogen transmission from animals to humans | ||
| Contact with infected animals | 14 | 58.3 |
| No known mode | 10 | 41.7 |
| Exposure to zoonotic pathogens due to livestock activity | ||
| Yes | 20 | 83.3 |
| No | 4 | 16.7 |
| Methods for preventing the transmission of zoonotic pathogens from animals | ||
| Reduced contact with animals | 1 | 4.2 |
| Don’t know | 23 | 95.8 |
| Ever heard of bovine tuberculosis? | ||
| Yes | 23 | 95.8 |
| No | 1 | 4.2 |
| Ever heard of human tuberculosis? | ||
| Yes | 21 | 87.5 |
| No | 3 | 12.5 |
| Have you ever seen an animal with tuberculosis? | ||
| Yes | 0 | 0 |
| No | 24 | 100 |
| Do you know the symptoms of animal tuberculosis? | ||
| Don’t know | 24 | 100 |
| How is tuberculosis transmitted between cattle? | ||
| Contact between animals | 6 | 25.0 |
| Don’t know | 18 | 75.0 |
| How is bovine tuberculosis transmitted to humans? | ||
| In contact with infected animals | 3 | 12.5 |
| Consumption of infected animal products | 6 | 25.0 |
| Don’t know | 15 | 62.5 |
| Risk of exposure to bovine tuberculosis after consuming raw milk from infected animals | ||
| No | 7 | 29.2 |
| Yes | 17 | 70.8 |
| Risk of exposure to bovine tuberculosis after eating rare meat from infected animals | ||
| No | 16 | 66.7 |
| Yes | 8 | 33.3 |
| Table 2: Distribution of bovine tuberculosis cases by location on farms in the urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021 | ||||
|---|---|---|---|---|
| Communes | Total animals tested | Positive | Prevalence (%) | 95% CI (%) |
| Borough 2/Ouaga | 49 | 3 | 6.12 | [0–12.84] |
| Komsilga | 52 | 17 | 32.69 | [19.94–45.44] |
| Koubri | 52 | 5 | 9.61 | [1.60–17.63] |
| Saaba | 53 | 5 | 9.43 | [1.56–17.30] |
| Kompi-Ipala | 53 | 3 | 5.66 | [0–11.88] |
| Pabre | 56 | 3 | 5.35 | [0–11.25] |
| Tanghin-Dassouri | 56 | 7 | 12.5 | [3.84–21.16] |
| Total | 371 | 43 | 11.59 | [8.75–15.25] |
| Table 3: Results of multivariate analysis of factors associated with bovine tuberculosis in herds in urban and peri-urban areas of Ouagadougou, Burkina Faso, 2021 | ||
|---|---|---|
| Variables | ORa (95% CI) | P-value |
| Grazing (having at-risk grazing neighbors) | ||
| No (ref.) | 1 | |
| Yes | 1.70 [0.41–7.01] | 0.46 |
| Type of production system | ||
| No (ref.) | 1 | |
| Extensive | 2.78 [0.73–11.98] | 0.62 |
| Contact between cattle and other domestic animals | ||
| No (ref.) | 1 | |
| Yes | 3.04 [1.30–10.74] | 0.003 |
| Buying cattle from high-risk farms | ||
| No (ref.) | 1 | |
| Yes | 2.80 [1.04–10.60] | 0.04 |
| Sharing with animals from other farms | ||
| No (ref.) | 1 | |
| Yes | 2.06 [1.09–8.59] | 0.18 |
| Proximity to cattle | ||
| No (ref.) | 1 | |
| Yes | 2.81 [0.09–17.11] | 0.080 |
| Farm size | ||
| No (ref.) | 1 | |
| Yes | 1.66 [0.31–7.42] | 0.29 |
| Animal body condition | ||
| Good (ref.) | 1 | |
| Poor | 1.76 [0.29–12.03] | 0.98 |
| Education level | ||
| School children (ref.) | 1 | |
| Out of school | 2.50 [0.11–11.54] | 0.43 |
| Knowledge of zoonoses and bovine tuberculosis | ||
| Good (ref.) | 1 | |
| Poor | 1.63 [0.32–8.22] | 0.54 |

References
- Eloit M. Panorama 2019-1: Eradication of bovine tuberculosis: a One Health issue [Internet]. Bull OIE. 2019 May 30 [cited 2026 Feb 26];2019(1):1-2. doi:10.20506/bull.2019.1.2909 Available from: https://bulletin.woah.org/?panorama=01-1-2019-1-eradication&lang=fr
- Euzéby JP. Dictionnaire de bactériologie vétérinaire [Dictionary of Veterinary Bacteriology] [Internet]. [cited 2026 Feb 26]. 8 p. Available from: https://docplayer.fr/42172333-J-p-euzeby-dictionnaire-de-bacteriologie-veterinaire.htm French
- Tschopp R. Tuberculose bovine à l’interface homme-bétail-faune sauvage en Afrique subsaharienne [Bovine tuberculosis at the human-livestock-wildlife interface in sub-Saharan Africa]. In: Schelling E, Tanner M, Waltner-Toews D, Whittaker MA, Zinsstag J, editors. One health, une seule santé: théorie et pratique des approches intégrées de la santé [Internet]. Versailles (France): Éditions Quæ; 2021 [cited 2026 Feb 26]. p. 229-244. Available from: http://books.openedition.org/quae/36150 French
- Firdessa R, Tschopp R, Wubete A, Sombo M, Hailu E, Erenso G, Kiros T, Yamuah L, Vordermeier M, Hewinson RG, Young D, Gordon SV, Sahile M, Aseffa A, Berg S. High Prevalence of Bovine Tuberculosis in Dairy Cattle in Central Ethiopia: Implications for the Dairy Industry and Public Health [Internet]. PLoS ONE. 2012 Dec 28 [cited 2026 Feb 26];7(12):e52851. doi:10.1371/journal.pone.0052851 Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0052851
- Caminiti A. Panorama 2019-1: Les coûts socio-économiques de la tuberculose bovine [Panorama 2019-1: The socio-economic costs of bovine tuberculosis] [Internet]. Bulletin de l’OIE. 2019 May 30 [cited 2026 Feb 26];2019(Panorama-1):1–2. doi:10.20506/bull.2019.1.2916 Available from: https://bulletin.woah.org/?lang=fr&panorama=3-05-costs-fr French
- Marsot M, Durand B, Scoizec A, Béral M, Mathevon Y, Courcoul A. Facteurs de risque de tuberculose bovine: une étude cas-témoins dans trois départements français [Risk factors for bovine tuberculosis: a case-control study in three French departments] (Ardennes, Côte d’Or et Dordogne) [Internet]. 2016 [cited 2026 Feb 26]. Available from: https://www.anses.fr/fr/system/files/SANT2012sa0103.pdf
- Organisation mondiale de la Santé, Organisation des Nations Unies pour l’alimentation et l’agriculture, Organisation mondiale de la santé animale. Feuille de route pour la tuberculose zoonotique [Roadmap for zoonotic tuberculosis] [Internet]. Genève (Suisse): Organisation mondiale de la Santé; 2017 [cited 2026 Feb 26]. 20 p. doi:10.13140/RG.2.2.36608.99840 Available from: https://apps.who.int/iris/handle/10665/259230
- Boukary AR. La tuberculose à Mycobacterium bovis en Afrique subsaharienne [Tuberculosis caused by Mycobacterium bovis in sub-Saharan Africa] [Internet]. Rev Elev Med Vet Pays Trop. 2011 [cited 2026 Feb 26];55(1):23-37. Available from: https://revues.cirad.fr/index.php/REMVT/article/view/9876 French
- Sidibé S, Dicko NA, Fané A, Doumbia RM, Sidibé CK, Kanté S, Mangané O, Konaté B, Koné AZ, Maïga MS, Fofana M. Tuberculose bovine au Mali : résultats d’une enquête épidémiologique dans les élevages laitiers de la zone périurbaine du district de Bamako [Bovine tuberculosis in Mali: results of an epidemiological survey in dairy farms in the peri-urban area of Bamako district] [Internet]. Rev Elev Med Vet Pays Trop. 2003 Mar 1 [cited 2026 Feb 26];56(3–4):115. doi:10.19182/remvt.9851 Available from: https://revues.cirad.fr/index.php/REMVT/article/view/9851
- Organisation des Nations Unies pour l’alimentation et l’agriculture. Aperçu sur les maladies zoonotiques et la RAM: l’argument en faveur d’un protocole d’élicitation d’experts au Burkina Faso [Overview of zoonotic diseases and AMR: the case for an expert elicitation protocol in Burkina Faso] [Internet]. Rome: FAO; 2017 [cited 2026 Feb 26]. Available from: https://www.fao.org/3/i8090fr/I8090FR.pdf French
- Traoré A, Tamboura HH, Bayala B, Rouamba DW, Yaméogo N, Sanou M. Prévalence globale des pathologies majeures liées à la production laitière bovine en système d’élevage intra-urbain à Hamdallaye (Ouagadougou) [Overall prevalence of major diseases related to bovine dairy production in intra-urban livestock farming systems in Hamdallaye (Ouagadougou)] [Internet]. Biotechnol Agron Soc Environ. 2004 [cited 2026 Feb 26];7. Available from: https://popups.uliege.be/1780-4507/index.php?id=1345
- Boussini H, Traore A, Tamboura HH, Bessin R, Boly H, Ouedraogo A. Prévalence de la tuberculose et de la brucellose dans les élevages bovins laitiers intra-urbains et périurbains de la ville d’Ouagadougou au Burkina Faso [Prevalence of tuberculosis and brucellosis in urban and peri-urban dairy cattle farms in Ouagadougou, Burkina Faso] [Internet]. Rev Sci Tech OIE. 2012 Dec 1 [cited 2026 Feb 26];31(3):943-51. doi:10.20506/rst.31.3.2168 Available from: https://www.oie.int/app/uploads/2021/03/rst-31-3-2168.pdf
- Ouoba LB. Prévalence et facteurs de risque de la brucellose et de la tuberculose bovine au Burkina Faso: étude rétrospective de 2013 à 2017 [Prevalence and risk factors of brucellosis and bovine tuberculosis in Burkina Faso: a retrospective study from 2013 to 2017] [master’s thesis on the Internet]. Dakar (Senegal): Cheikh Anta Diop University; 2018 [cited 2026 Feb 26]. Available from: https://bibnum.ucad.sn/handle/123456789/11234 French Subscription required to access full text
- Delafosse A, Goutard F, Thebaud E. Epidémiologie de la tuberculose et de la brucellose des bovins en zone périurbaine d’Abéché (Tchad) [Epidemiology of tuberculosis and brucellosis of cattle in the peri-urban area of Abéché (Chad)] [Internet]. Rev Elev Med Vet Pays Trop. 2002 Jan 1 [cited 2026 Feb 26];55(1):5. doi:10.19182/remvt.9846 Available from: https://revues.cirad.fr/index.php/REMVT/article/view/9846
- Institut National de la Statistique et de la Démographie (Burkina Faso). Cinquième Recensement Général de la Population et de l’Habitation du Burkina Faso [Fifth general census of population and housing of Burkina Faso] [Internet]. Burkina Faso (Ouagadougou); 2020 Sept [cited 2026 Feb 26]. 8 p. Available from: https://www.insd.bf/fr/file-download/download/public/2073
- Ministère des Ressources Animales et Halieutiques (Burkina Faso). Annuaire des statistiques de l’élevage 2018 [Yearbook of Livestock Statistics 2018] [Internet]. Ouagadougou (Burkina Faso): Ministère des Ressources Animales et Halieutiques; 2019 Oct [cited 2026 Feb 26]. 140 p. French
- Burkina Faso Ministry of Animal Resources. National Livestock Census 2020: Cattle Population Estimates [Internet]. Ouagadougou: Burkina Faso Ministry of Animal Resources; 2020 [cited 2026 Feb 26]. Available from: https://www.mrah.gov.bf/ (official ministry site; specific census document URL not publicly archived)
- Philizot S, Bénet JJ. Réalisation de l’intradermotuberculination comparative [Performance of comparative intradermal tuberculin testing] [Internet]. Bulletin-des-GTV. 2011 Sept [cited 2026 Feb 26];61:77-80. Available from: https://www.gdshautsdefrance.fr/wp-content/uploads/2019/03/Bulletin-des-GTV-Article-IDC.pdf French
- World Organisation for Animal Health. Bovine tuberculosis (infection with Mycobacterium bovis) [Internet]. In: Manual of diagnostic tests and vaccines for terrestrial animals. Paris (France): World Organisation for Animal Health; 2023 [cited 2026 Feb 26]. Available from: https://www.woah.org/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/
- Lingoland English-English Dictionary. Pasture [Internet] [cited 2026 Feb 26]. Available from: https://lingolandedu.com/en/english-english-dictionary/pasture
- Palisson A. Rôles des contacts entre bovins dans la circulation d’agents infectieux. Importance respective du commerce et du pâturage pour la tuberculose bovine [Cattle Trade, Contacts on Pastures and Infectious Disease Spread Between Cattle Herds. Application to Bovine Tuberculosis] [dissertation on the Internet]. Paris Saclay (France): Université Paris-Saclay; 2016 [cited 2026 Feb 26]. 259 p. Available from: https://theses.hal.science/tel-01470351v1/document French
- Sambo M, Lembo T, Cleaveland S, Ferguson HM, Sikana L, Simon C, Urassa H, Hampson K. Knowledge, Attitudes and Practices (KAP) about Rabies Prevention and Control: A Community Survey in Tanzania [Internet]. PLoS Negl Trop Dis. 2014 Dec 4 [cited 2026 Feb 26];8(12):e3310. doi:10.1371/journal.pntd.0003310 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0003310
- Xiang N, Shi Y, Wu J, Zhang S, Ye M, Peng Z, Zhou L, Zhou H, Liao Q, Huai Y, Li L, Yu Z, Cheng X, Su W, Wu X, Ma H, Lu J, McFarland J, Yu H. Knowledge, attitudes and practices (KAP) relating to avian influenza in urban and rural areas of China [Internet]. BMC Infect Dis. 2010 Dec [cited 2026 Feb 26];10(1):34. doi:10.1186/1471-2334-10-34 Available from: https://bmcinfectdis.biomedcentral.com/articles/10.1186/1471-2334-10-34
- Tschopp R, Bekele S, Moti T, Young D, Aseffa A. Brucellosis and bovine tuberculosis prevalence in livestock from pastoralist communities adjacent to Awash National Park, Ethiopia [Internet]. Preventive Veterinary Medicine. 2015 Jun 15 [cited 2026 Feb 26];120(2):187–94. doi:10.1016/j.prevetmed.2015.03.004 Available from: https://www.sciencedirect.com/science/article/pii/S016758771500077X
- Sreeramareddy CT, Harsha Kumar HN, Arokiasamy JT. Prevalence of self-reported tuberculosis, knowledge about tuberculosis transmission and its determinants among adults in India: results from a nation-wide cross-sectional household survey [Internet]. BMC Infect Dis. 2013 Dec [cited 2026 Feb 26];13(1):16. doi:10.1186/1471-2334-13-16 Available from: https://bmcinfectdis.biomedcentral.com/articles/10.1186/1471-2334-13-16
- Sadiq MB, Hamid NA, Yusri UK, Ramanoon SZ, Mansor R, Affandi SA, Watanabe M, Kamaludeen J, Syed-Hussain SS. Ruminant farmers’ knowledge, attitude and practices towards zoonotic diseases in Selangor, Malaysia [Internet]. Preventive Veterinary Medicine. 2021 Nov [cited 2026 Feb 26];196:105489. doi:10.1016/j.prevetmed.2021.105489 Available from: https://www.sciencedirect.com/science/article/pii/S016758772100225X
- Vikou R, Aplogan LG, Ahanhanzo C, Baba-Moussa L, Gbangboche AB. Prévalence de la brucellose et de la tuberculose chez les bovins au Bénin [Prevalence of brucellosis and tuberculosis in cattle in Benin] [Internet]. Int J Bio Chem Sci. 2018 Jun 1 [cited 2026 Feb 26];12(1):120. doi:10.4314/ijbcs.v12i1.9 Available from: https://www.ajol.info/index.php/ijbcs/article/view/169998
- Kanyala E, Shuaib YA, Schwarz NG, Andres S, Richter E, Sawadogo B, Sawadogo M, Germaine M, Lassina O, Poppert S, Frickmann H. Prevalence and Molecular Characterization of Mycobacterium bovis in Slaughtered Cattle Carcasses in Burkina Faso; West Africa [Internet]. Microorganisms. 2022 Jul 8 [cited 2026 Feb 26];10(7):1378. doi:10.3390/microorganisms10071378 Available from: https://www.mdpi.com/2076-2607/10/7/1378
- World Health Organization, Food and Agriculture Organization of the United Nations, World Organisation for Animal Health. Roadmap for zoonotic tuberculosis [Internet]. Geneva (Switzerland): World Health Organization; 2017 [cited 2026 Feb 26]. 20 p. Available from: https://www.who.int/publications/i/item/9789241513043
- Djafar ZR, Benazi N, Bounab S, Sayhi M, Diouani MF, Benia F. Distribution of seroprevalence and risk factors for bovine tuberculosis in east Algeria [Internet]. Preventive Veterinary Medicine. 2020 Oct 1 [cited 2026 Feb 26];183:105127. doi:10.1016/j.prevetmed.2020.105127 Available from: https://www.sciencedirect.com/science/article/pii/S016758772030312X
- Ayele WY, Neill SD, Zinsstag J, Weiss MG, Pavlik I. Bovine tuberculosis: an old disease but a new threat to Africa [Internet]. Int J Tuberc Lung Dis. 2004 Aug [cited 2026 Feb 26];8(8):924-37. Available from: https://pubmed.ncbi.nlm.nih.gov/15305435/
