Research | Open Access | Volume 9 (2): Article 53 | Published: 01 Apr 2026
Global trends in bioterrorism and biosecurity research publications (1997–2025): A Scopus bibliometric analysis
Menu, Tables and Figures
On Pubmed
On Google Scholar
Navigate this article
Tables
| Table 1: Top five leading affiliations of authors and fields of study in bioterrorism and biosecurity research (1997–2025) | |
|---|---|
| Affiliation of authors | Publication (n) |
| Centers for Disease Control and Prevention | 30 |
| U.S. Army Medical Research Institute of Infectious Diseases | 29 |
| University of Pittsburgh Medical Center | 26 |
| Johns Hopkins University | 24 |
| National Institutes of Health (NIH) | 21 |
| Field of study (Discipline) | |
| Medicine | 577 |
| Social Sciences | 265 |
| Biochemistry, Genetics, and Molecular Biology | 258 |
| Immunology and Microbiology | 258 |
| Environmental Science | 188 |
Table 1: Top five leading affiliations of authors and fields of study in bioterrorism and biosecurity research (1997–2025)
| Table 2: Top five countries with the most citations of biosafety and biosecurity research | ||
|---|---|---|
| Country | Publication (n) | Number of citations |
| United States | 570 | 19643 |
| United Kingdom | 66 | 1934 |
| Germany | 37 | 1531 |
| France | 37 | 1018 |
| Australia | 35 | 697 |
Table 2: Top five countries with the most citations of biosafety and biosecurity research
Figures
Keywords
- Bioterrorism
- Biosecurity
- Preparedness
- Public health
- Biological agents
Festus Mulakoli1,&, Samwel Gesaka2, John Mbataru2
1School of Nursing and Midwifery, Aga Khan University, Nairobi, Kenya, 2Department of Biomedical Sciences, Aga Khan University, Nairobi, Kenya
&Corresponding author: Festus Mulakoli, School of Nursing and Midwifery, Aga Khan University, Nairobi, Kenya, Email: mulakolifesto@gmail.com, ORCID: https://orcid.org/0000-0002-1540-5938
Received: 20 Sep 2025, Accepted: 25 Mar 2026, Published: 01 Apr 2026
Domain: Global Health Security
Keywords: Bioterrorism, biosecurity, preparedness, public health, biological agents
©Festus Mulakoli et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Festus Mulakoliu et al., Global trends in bioterrorism and biosecurity research publications (1997–2025): A Scopus bibliometric analysis. Journal of Interventional Epidemiology and Public Health. 2026; 9(2):53. https://doi.org/10.37432/jieph-d-25-00200
Abstract
Introduction: Bioterrorism and biosecurity preparedness are critical pillars of global public health protection and resilience of health systems worldwide. Although this field has grown over the past few decades, there remains limited clarity on how research efforts have evolved and where critical gaps persist, particularly regarding global collaboration and emerging biological threats. To address this, we conducted a bibliometric analysis to map publication trends, influential contributors, and collaboration networks in bioterrorism and biosecurity from 1997 to 2025. This study analyzed global research output, collaboration, and thematic evolution in bioterrorism and biosecurity and identified gaps relevant to preparedness and policy.
Methods: We searched the Scopus database for peer-reviewed articles, reviews, and conference papers in English using the terms “bioterrorism” OR “biological terrorism” OR “biological warfare” AND “biosecurity” OR “biosafety” OR “biodefense. Extracted data were analyzed for publication trends, that is, annual, country, author, and institutional outputs. Citation analysis, keyword co‑occurrence, international co‑authorship, and collaboration were analyzed using VOSviewer (v1.6.20).
Results: A total of 1,222 records were included. The United States led the output (n=570), followed by the United Kingdom (n=66). The highest number of annual publications occurred in 2004 (n=112). The leading institutions included the Centers for Disease Control and Prevention (n=30) and the U.S. Army Medical Research Institute of Infectious Diseases (n=29). Medicine, social sciences, and biochemistry/genetics/molecular biology were the dominant disciplines. Keyword networks clustered around “biological warfare,” “humans,” and “bioterrorism.” Arnon’s 2001 paper received the most citations (n=1,485).
Conclusions: Publication trends in biosafety and biosecurity are highly event-driven, as shown in this analysis, and are concentrated in high-income countries. Fewer publications are produced in low- and middle-income countries, such as in Africa. These findings underscore the need for sustained collaboration, equitable partnerships, and structured support for low- and middle-income countries.
Introduction
Bioterrorism and biosecurity refer to the deliberate release of biological agents such as viruses, bacteria, toxins, or other harmful pathogens that cause illness, death, or panic among humans, animals, and plants [1]. It is distinct from other forms of terrorism because of its silent nature, low cost, ease of replication, and ability to transcend geographical boundaries [2]. Illustrative incidents include the 2001 anthrax attacks in the United States, which resulted in five fatalities and numerous infections, and the 1984 Salmonella contamination orchestrated by the Rajneeshee cult in Oregon, which led to more than 750 documented cases of foodborne illness [3, 4]. Other examples include Aum Shinrikyo’s experiments with biological agents and the Soviet Union’s bioweapons program, including an anthrax release in Sverdlovsk [5, 6]. In response, biosecurity measures have been strengthened globally with initiatives such as the U.S. BioWatch Program and international regulations aimed at improving surveillance and laboratory biosafety [7, 8].
The Centers for Disease Control and Prevention (CDC) and other organizations have classified bioterrorism and biosecurity agents into three categories (A, B, and C) based on their severity and potential public health impact [9-11]. Category A agents are characterized by the highest severity, are highly contagious, have high mortality rates, and have the potential to cause pandemics. Thus, specific public health measures are required to mitigate their impact on global health. Biological and public safety (BPS) agents in this category include anthrax, botulism, plague, smallpox, tularemia, and viral hemorrhagic fever. Category B agents are the second-highest priority, being moderately easy to spread, causing moderate illness with low mortality, and requiring enhanced surveillance. Examples include brucellosis, food and water safety threats, Q fever, and viral encephalitis. Category C agents, including the Nipah virus, hantaviruses, yellow fever, and pandemic influenza strains, are third-highest priority emerging pathogens that can be engineered for mass use [12].
Despite the potential of these disease-causing agents, which could lead to catastrophic consequences, large-scale biological weapons attacks have been relatively unsuccessful in the past [13]. However, small-scale bioterrorism and biosecurity-related activities continue to threaten global safety in some regions with political instability. Recent technological advances and easy access to biological agents present major challenges for biosafety and biosecurity globally [14, 15]. For instance, recent biosafety threats involve dual-use technologies (artificial intelligence, synthetic biology, and CRISPR), which enable the creation of dangerous pathogens, increase bioterrorism, and laboratory accident risks, as highlighted by the impact of COVID-19 and compounded by cyber threats to biological data, thus requiring stronger governance and security [16]. This is similar to bioterrorism and biosecurity related incidents, such as the 47 anthrax hoaxes and threats reported in the United States between 1992 and mid-February 1999 [17].
Preparedness to deal with bioterrorism and biosecurity threats is essential for public health systems and healthcare providers worldwide. Conducting studies that address biosafety and biosecurity concerns is crucial for regularly reassessing these threats. Such studies provide valuable information for the development of effective biosafety and biosecurity-related policies and protocols to manage such events should they occur in any country. Through such investigations, emerging biological risks that may arise from natural pathogens, laboratory accidents, or intentional misuse can be identified. Studies have assessed containment measures, protective equipment, and decontamination procedures to enhance biosafety [18]. In biosecurity, research contributes to securing facilities, pathogen-tracking systems, and “dual-use research of concern” (DURC) oversight frameworks to prevent hazardous material theft [19].
In many vulnerable countries, local healthcare providers serve as frontline responders to bioterrorism events and broader biosecurity threats. However, historically, these frontline systems have been chronically underfunded and insufficiently integrated into national and global preparedness planning, limiting their capacity to detect, contain, and respond effectively [20]. Studies have identified factors that influence preparedness, including perceived coping efficacy and frontline preparedness [21]. Effective preparedness involves developing detailed response plans, assessing strengths and weaknesses, and collaborating with international agencies [22]. While most healthcare professionals express a willingness to assist during bioterrorism and biosecurity incidents, many report feeling underprepared [23-25]. A study of pediatricians in Michigan found that 80% of the respondents believed that terrorism was a threat, but only 12% felt adequately prepared for biological attacks [25]. Similarly, a national survey revealed that only 43% of emergency physicians and 21% of primary care physicians felt well prepared to respond to bioterrorism and biosecurity [23].
Bioterrorism and biosecurity preparedness are critical components of both national and global health systems. This requires a multifaceted approach involving scientific studies, educational programs, training capacity, security planning, and coordination across various sectors and levels of the global government [26-28]. Although perceptions of bioterrorism and biosecurity attacks have increased, many countries lack the capacity to manage these threats. Some of the key highlights identified by local health departments and healthcare providers in various global regions include a lack of adequate knowledge of biosafety/biosecurity and resources to respond effectively to such events [24].
Studies have shown that healthcare professionals are willing to assist in bioterrorism and biosecurity response efforts despite their limited training and preparedness [24, 29-32]. However, discrepancies exist between the perceived risks of bioterrorism and biosecurity across global regions. The United States views bioterrorism primarily through the lens of advanced biotechnology misuse and synthetic biology threats, as emphasized in biosecurity analyses [16, 33].
Europe, the Middle East, and the Gulf perceive risks through agroterrorism and food system vulnerabilities, reflecting regional food security and geopolitical concerns [34, 35]. Biosafety and biosecurity frameworks across Europe, the Middle East, and the Gulf exhibit significant variations shaped by differing levels of regulatory maturity, geopolitical pressure, and scientific capacity. In Europe, institutionalized biosafety governance is supported by long‑standing professional bodies such as the European Biosafety Association (EBSA), which promotes standardized competency development, BSL‑3/BSL‑4 facility compliance, and the adoption of international standards, including ISO 35001 and CWA 16335, reflecting the region’s emphasis on harmonized, skills‑based biosafety systems [36]. European states, such as Germany, the United Kingdom, and other scientific powerhouses, have also integrated biosecurity considerations into broader “science diplomacy” strategies, which increasingly shape global biosecurity governance and promote multilateral cooperation on emerging biotechnologies [37]. In contrast, countries in the Middle East face more acute challenges due to political instability, limited regulatory oversight, and expanding biotechnology sectors that increase exposure to dual‑use research risks. Studies have shown that biosafety and biosecurity systems in the region remain uneven, with gaps in laboratory governance, inconsistent implementation of international biosafety protocols, and low levels of DURC oversight, issues highlighted in regional analyses of biotechnological expansion and biological weapon-related vulnerabilities [38, 39]. Across the Gulf and broader Eastern Mediterranean Region, WHO assessments reveal fragmented national laboratory systems and persistent deficiencies in biosafety and biosecurity governance, particularly in the areas of regulatory coherence, high-risk pathogen management, and workforce capacity, despite accelerated investment following the COVID‑19 pandemic [40]. Complementing these findings, the UNICRI’s regional evaluation illustrates that Gulf states continue to prioritize strengthening their biosafety and biosecurity capacities in response to transboundary biological threats. However, they remain hindered by uneven implementation of Joint External Evaluation (JEE) recommendations, gaps in real-time surveillance, and limited emergency response integration [41]. Collectively, these studies underscore the need for enhanced cross-regional cooperation, harmonized regulatory standards, and integrated biosafety and biosecurity governance mechanisms to ensure resilient preparedness across Europe, the Middle East, and the Gulf.
In Asia, particularly in countries with advancing biotechnology sectors, risk perceptions focus on the dual-use potential of emerging technologies influenced by historical events, such as Japan’s Unit 731 [42]. These examples show that global regions perceive bioterrorism differently, based on distinct contextual realities, leading to varied threat assessments and preparedness priorities [43]. Biosafety and biosecurity efforts across Asian countries, particularly in Southeast and East Asia, have expanded significantly in response to escalating biological threats; however, substantial disparities remain in national preparedness and regulatory capacity. In Southeast Asia, countries such as Singapore, Malaysia, Indonesia, Thailand, Vietnam, and the Philippines continue to strengthen their laboratory biosafety systems and biosecurity governance driven by recurring outbreaks of emerging infectious diseases and the increasing availability of advanced biotechnologies [44]. Regional assessments highlight that while several nations have improved laboratory standards and national pathogen‑control lists, inconsistent implementation of frameworks for dual‑use research of concern (DURC) poses persistent risks, compounded by uneven institutional awareness and limited regulatory expertise [45]. ASEAN leaders have also acknowledged that differences in biosafety capacities and legal instruments across member states can lead to vulnerabilities in pathogen handling, high‑containment laboratory safety, and the prevention of accidental or deliberate release of dangerous agents [46]. These concerns have motivated calls for stronger harmonization of biosafety protocols, expanded cross-border laboratory networking, and joint training programs to elevate minimum safety standards [47]. Simultaneously, Asia’s rapid growth in biotechnology, ranging from genetic engineering laboratories in Singapore and Malaysia to the expanding virology and synthetic biology sectors in China, Japan, and South Korea, has intensified the need for robust biosecurity oversight to prevent misuse or unregulated experimentation [44]. Strategic dialogues hosted in Singapore underscore the importance of regional cooperation, emphasizing medical countermeasures, laboratory security, and emerging technological risks, such as cyber‑biosecurity and AI‑enabled biological design [48]. Collectively, these studies demonstrate that although Asia has made notable progress in advancing biosafety and biosecurity systems, sustained regional frameworks, harmonized governance, and strengthened DURC oversight remain essential for mitigating biological risks across regions characterized by high population density, rapid technological expansion, and diverse national capacities.
African states frame bioterrorism risks around weak biosafety infrastructure, with gaps in surveillance, laboratory capacity, and response systems that increase their vulnerability to biological threats. Biosafety and biosecurity systems across Africa face substantial challenges due to structural weaknesses in laboratory capacity, regulatory oversight, and sustainable workforce development. Many countries, including Nigeria, Kenya, Uganda, Ghana, Tanzania, Ethiopia, South Africa, Senegal, Rwanda, and the Democratic Republic of Congo, continue to experience shortages in high‑containment laboratories, inadequate biosafety equipment, and inconsistent implementation of national biorisk management frameworks [49, 50]. These vulnerabilities were highlighted during major outbreaks, such as Ebola in Sierra Leone, Liberia, and Guinea, where gaps in biosafety practices, inconsistent use of personal protective equipment, and limited laboratory networks accelerated transmission and hindered early detection [51]. Countries such as Uganda, the DRC, and South Sudan regularly encounter viral hemorrhagic fever outbreaks; they remain dependent on external partners for advanced genomic sequencing and pathogen characterization. Meanwhile, nations such as Kenya, South Africa, and Senegal, home to institutions like KEMRI, NICD, and Institut Pasteur Dakar, demonstrate comparatively stronger capacities, but they still face cross‑border risks due to uneven regional preparedness. Governance gaps also persist across the continent, with many states lacking comprehensive national policies for dual‑use research oversight, biosafety certification systems, and secure pathogen transport protocols. Although initiatives led by the Africa CDC, the African Union, and regional laboratory networks have advanced capacity building and harmonization of biosafety norms, sustained political commitment and long‑term investment remain essential to strengthen the continent’s resilience against both intentional biological threats and accidental laboratory incidents.
Biosafety and biosecurity capacity across Africa remains highly variable and constrained by significant structural gaps in laboratory infrastructure, regulatory oversight, and sustainable workforce development. Countries such as Nigeria, Kenya, Uganda, Ghana, Ethiopia, South Africa, Senegal, Tanzania, Rwanda, the Democratic Republic of Congo, and Sierra Leone continue to face shortages in high-containment laboratories, inconsistent application of biorisk management standards, and limited national mechanisms for governing dual-use research and pathogen security [52, 53]. These gaps have been repeatedly highlighted during major public health emergencies; for example, the Ebola outbreaks in Guinea, Liberia, and Sierra Leone demonstrated how inadequate biosafety procedures and weak surveillance systems can exacerbate transmission dynamics and delay effective response efforts [51, 54]. While some countries, such as Kenya, South Africa, and Ethiopia, have developed comparatively stronger regulatory and institutional architectures through national biosafety laws and specialized public health laboratories, wide disparities persist across the continent [53]. Recent evaluations show that many states still lack comprehensive legal frameworks, trained biosafety officers, secure pathogen inventories, and standardized national certification pathways for biosafety professionals, leaving them vulnerable to accidental release or deliberate misuse of biological materials [52, 55]. Nonetheless, progress is accelerating: the Africa CDC’s continental Biosafety and Biosecurity Initiative, the establishment of Regional Technical Working Groups across Central, Eastern, Northern, Southern, and West Africa, and the operationalization of regional Centers of Excellence in South Africa, Tanzania, and Senegal have strengthened governance coordination, emergency readiness, and training systems [52, 55]. Sustained investment, domestic policy reform, and expanded regional collaboration remain essential for building a resilient continental architecture capable of managing both intentional biological threats and unintentional laboratory-associated risks.
To address these challenges, a comprehensive bioterrorism and biosecurity preparedness strategy should focus on enhancing knowledge through continuous education and training, developing detailed response plans, improving detection technologies, and fostering collaboration among local, national, and international stakeholders [56, 57]. Additionally, incorporating complexity theory into preparedness efforts may better equip healthcare organizations to manage the unpredictable nature of bioterrorism [58].
This bibliometric analysis of bioterrorism and biosecurity is valuable for several reasons. Bibliometric analysis can provide a comprehensive overview of the research landscape in bioterrorism and biosecurity preparedness, helping to identify key trends, influential authors, and seminal works in the field [59]. This approach will allow researchers and policymakers to understand the evolution of bioterrorism and biosecurity preparedness research over time, highlighting emerging areas of focus and potential knowledge gaps in the literature. Such an analysis could reveal interdisciplinary connections and collaborations, as bioterrorism and biosecurity preparedness involve various sectors, including public health, medicine, emergency response, and national security [60]. By mapping these connections, researchers can identify opportunities for further interdisciplinary work and knowledge sharing. A bibliometric study would help quantify the impact of different research areas on bioterrorism and biosecurity preparedness, such as detection technologies, response strategies, and healthcare system preparedness [61]. This information can guide future research priorities and funding allocations. Although there is a significant body of literature on bioterrorism and biosecurity preparedness, many studies have indicated a lack of practical preparedness among healthcare professionals and local health departments [24, 62]. Bibliometric analysis can help bridge this gap by identifying the most influential and practical research areas in the field of implementation science.
Global research on bioterrorism and biosecurity has grown in recent years, driven by significant biological events and technological advancements. However, a notable gap persists between academic understanding and practical preparedness, particularly in low- and middle-income countries (LMICs). This study intends to inform biosecurity and bioterrorism preparedness efforts in Africa by identifying global trends that highlight gaps in research capacity, surveillance infrastructure, and scientific collaboration. Because African nations face disproportionate vulnerability to biological threats owing to limited laboratory capacity, dependence on external genomic sequencing, and fragmented preparedness systems, mapping global research dynamics can help contextualize where Africa remains under-resourced and where strategic investment is most needed. To address this issue, this study undertook a bibliometric analysis of global research trends to examine trends in bioterrorism and biosecurity from 1997 to 2025.
Methods
This study employed bibliometric analysis to examine global research trends in bioterrorism and biosecurity from 1997 to 2025. Data were extracted from the Scopus database using keywords such as (“bioterrorism” OR “biological terrorism” OR “biological warfare”) AND (“biosecurity” OR “biosafety” OR “biodefense”). Publications were analyzed for annual output, leading countries/institutions, and authorship patterns using the Scopus database. Co-authorship networks and keyword co-occurrences were analyzed using VOSviewer software. This study also assessed thematic evolution and collaborative networks to identify shifts in research focus over time. Only peer-reviewed articles, reviews, and conference papers published in English were included. These findings provide insights into the progression of bioterrorism and biosecurity research, highlighting key contributors and emerging trends.
Search strategy
A comprehensive literature search was conducted using the Scopus database to identify relevant publications on bioterrorism and biosecurity preparedness between 1997 and 2025. The search strategy included keywords such as (“bioterrorism” OR “biological terrorism” OR “biological warfare”) AND (“biosecurity” OR “biosafety” OR “biodefense”). Boolean operators (AND) and (OR) were used to refine the search results.
Inclusion and exclusion criteria
We included peer-reviewed articles, review papers, and conference proceedings in English that focused on bioterrorism, biosecurity policies, threat preparedness, and response strategies. Non-English publications, editorials, letters, news articles, and studies unrelated to bioterrorism or biosecurity were excluded.
Data extraction and analysis
The extracted metadata included the title, authors, affiliations, publication year, citations, keywords, and abstracts. Bibliometric analysis was conducted using VOSviewer (version 1.6.20) and Scopus to examine annual publication trends, leading countries, institutions, authors, collaboration patterns, and keyword co-occurrence.
Study quality and risk of bias assessment
Given the bibliometric nature of this study, a traditional risk of bias assessment (e.g., PRISMA) was not applicable. However, source reliability was ensured by selecting only peer-reviewed publications from indexed journals. Duplicate records were removed, and data consistency was verified manually. This methodology provides a systematic approach to mapping global research trends in bioterrorism and biosecurity preparedness and identifying key contributions and knowledge gaps.
Ethical considerations
This study, as a bibliometric analysis of published literature, did not require formal ethical approval because it exclusively utilized publicly available scientific data from the Scopus database without involving human or animal subjects, confidential records, or sensitive personal information. All data were extracted from peer-reviewed journal articles and conference proceedings in compliance with standard academic research ethics, ensuring proper attribution through citations while maintaining transparency in the methodology and analytical processes. The research was conducted with academic integrity, free from conflicts of interest, and designed solely to contribute to the scholarly understanding of global trends in bioterrorism and biosecurity preparedness, without misrepresenting the findings or using restricted materials.
Results
Authors with the highest number of publications on biosafety and biosecurity
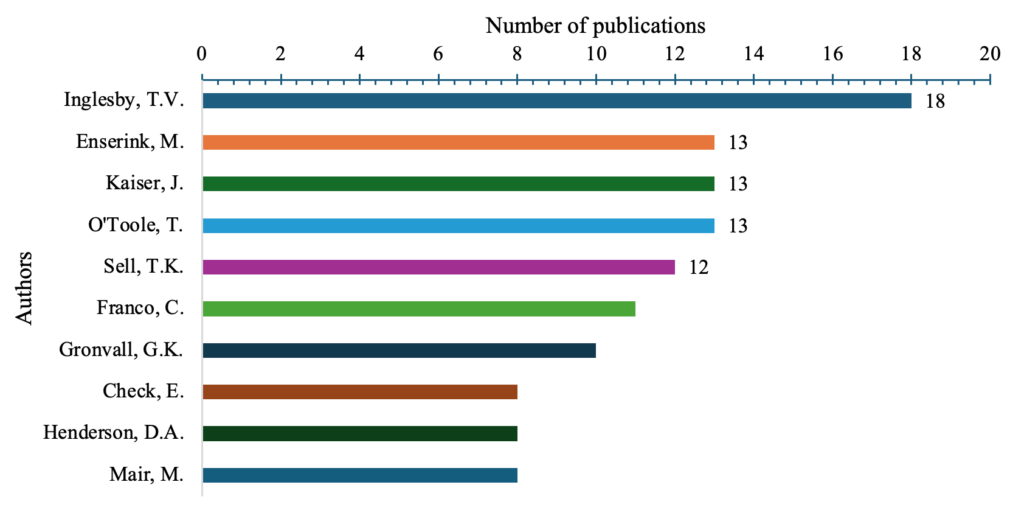
Among the listed authors, Inglesby, T. V. (n=18), had the highest number of publications (Figure 1), followed by Enserink, M. (n=13). Other notable contributors include Kaiser, J., O’Toole, T., Sell, T.K., Franco, C., Gronvall, G.K., Check, E., Henderson, D.A., and Mair, M., with publication counts ranging from (n=8) to (n=13).
The top five countries with the highest publications on biosafety and biosecurity
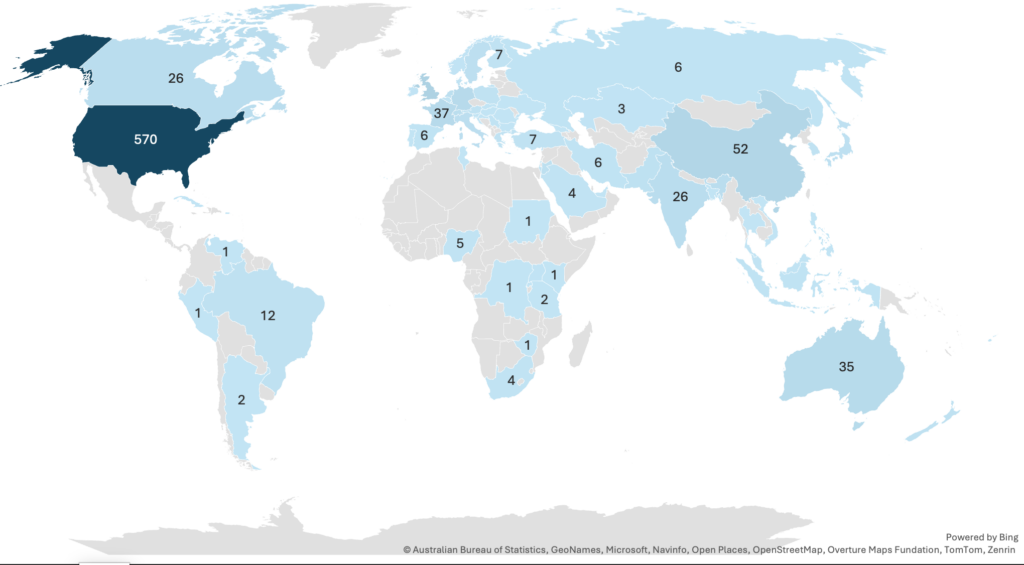
The world map (Figure 2) visualizes the distribution of data points across various countries using blue shading to indicate frequency, with darker shades representing higher numbers. The United States leads significantly, with 570 occurrences, far surpassing all other nations. The United Kingdom ranked second (n=66), and China ranked third (n=52). This distribution highlights the strong concentration of data from North America and Oceania, with notable contributions from Europe, South America, and Asia.
Global trends in publications
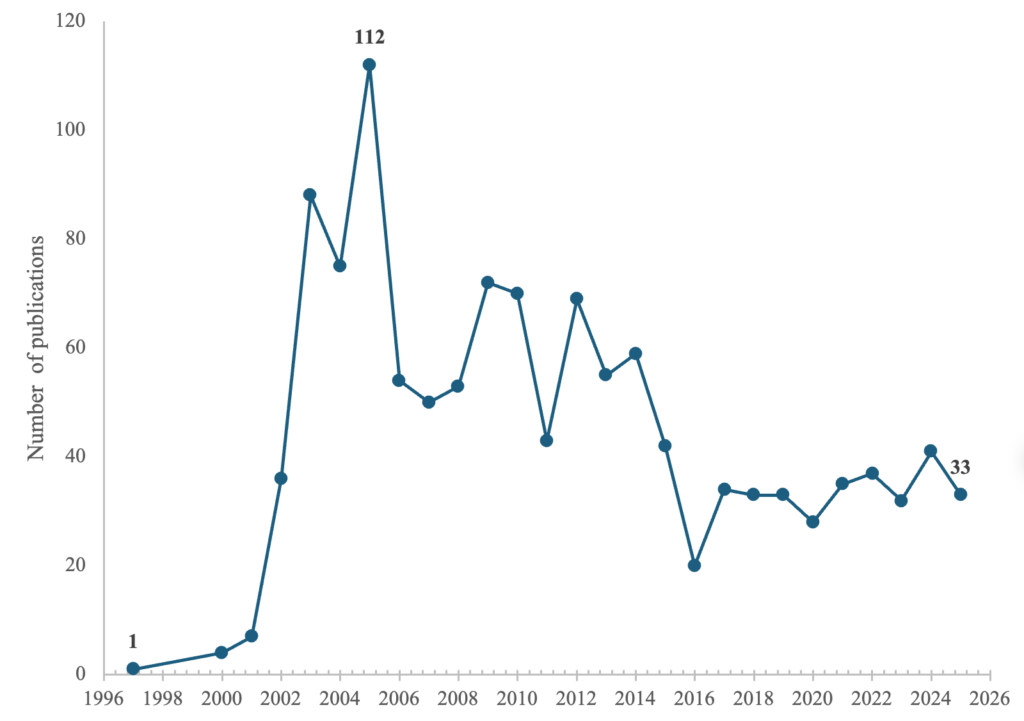
Figure 3 illustrates the annual publication output in the field from 1997 to 2025. A sharp increase in scholarly interest began around 2000, culminating in a peak of 112 publications in 2004. A notable decrease was observed between 2016. From 2017 onwards, publication activities remained steady, with at least 28 to 41 publications per annum.
Authors’ affiliations and disciplinary focus of studies on biosafety and biosecurity
Table 1 presents data on the number of publications related to bioterrorism and biosecurity, categorized by affiliation and discipline. The analysis of author affiliations and research domains revealed significant institutional and disciplinary contributions to the bioterrorism and biosecurity literature. The Centers for Disease Control and Prevention (CDC) led with (n=30) publications, closely followed by the U.S. Army Medical Research Institute of Infectious Diseases (n=29), University of Pittsburgh Medical Center (n=26), Johns Hopkins University (n=24), and National Institutes of Health (n=21). In terms of disciplinary focus, medicine dominated the field with 577 publications, followed by social sciences (n=265), biochemistry, genetics, and molecular biology (n=258), immunology, microbiology (n=258), and environmental science (n=188).
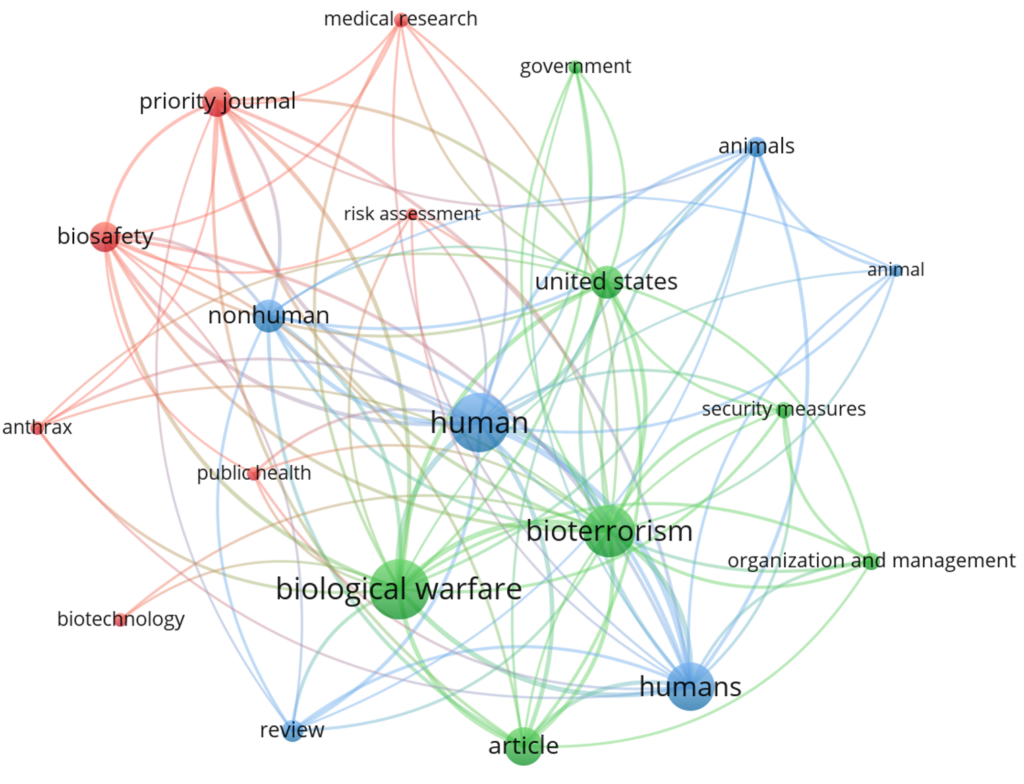
Co-occurrence of index keywords used in publications on biosafety and biosecurity
Network visualization reveals the structural relationships between key topics in biosecurity and bioterrorism. Figure 4 shows multiple nodes, each representing a distinct topic, and edges denoting conceptual or thematic connections between them. The nodes are color-coded into three primary clusters (red, blue, and green), suggesting thematic groupings and communities within the dataset. Prominent nodes, such as “biological warfare,” “human,” and “information,” occupy central positions, indicating a high degree of connectivity and relevance to multiple subtopics.
Scientific journal with the highest number of publications on biosafety and biosecurity
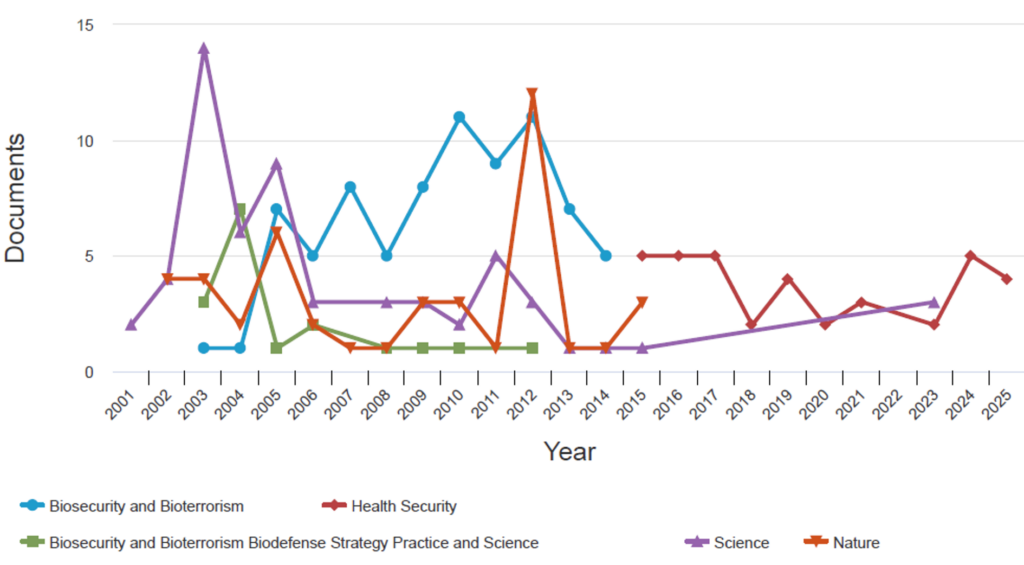
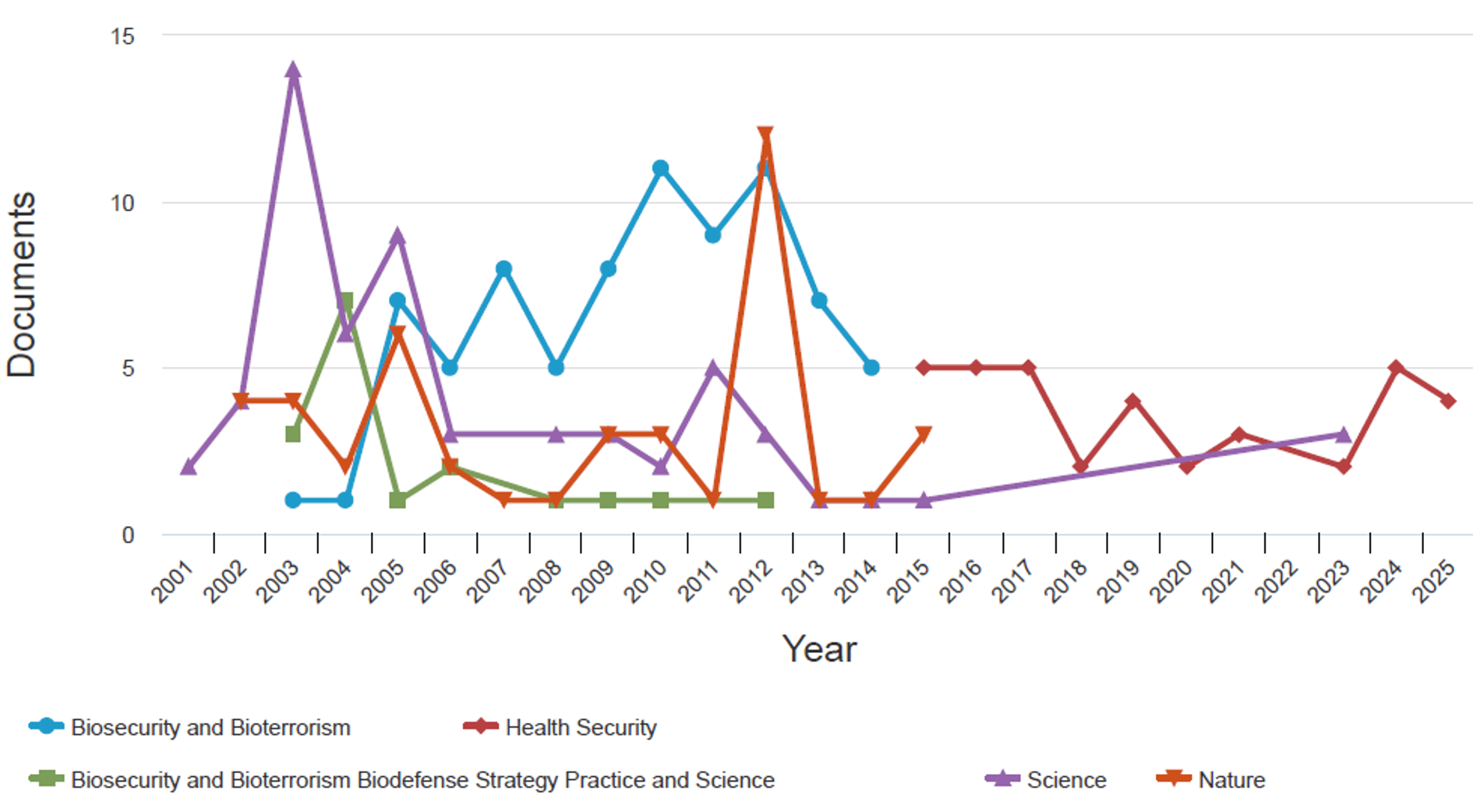
Between 2001 and 2025, publication trends across five key journals and categories revealed distinct patterns in bioterrorism and biosecurity research. The journals Biosecurity and Bioterrorism led to 78 publications, followed by Science (n=60), Nature (n=4), Health Security (n=33), and Biosecurity and Bioterrorism: Biodefense Strategy, Practice, and Science (n=17). Health security demonstrated a steady upward trajectory in response to global health emergencies, peaking between 2018 and 2020. Biosecurity and bioterrorism exhibited notable spikes in 2011–2012, reflecting increased scholarly attention during that period. In contrast, Biosecurity and Bioterrorism: Biodefense Strategy, Practice, and Science maintained a lower overall publication volume, with a modest increase between 2010 and 2012 (Figure 5).
Collaboration and Co-authorship between countries with an interest in biosafety and biosecurity
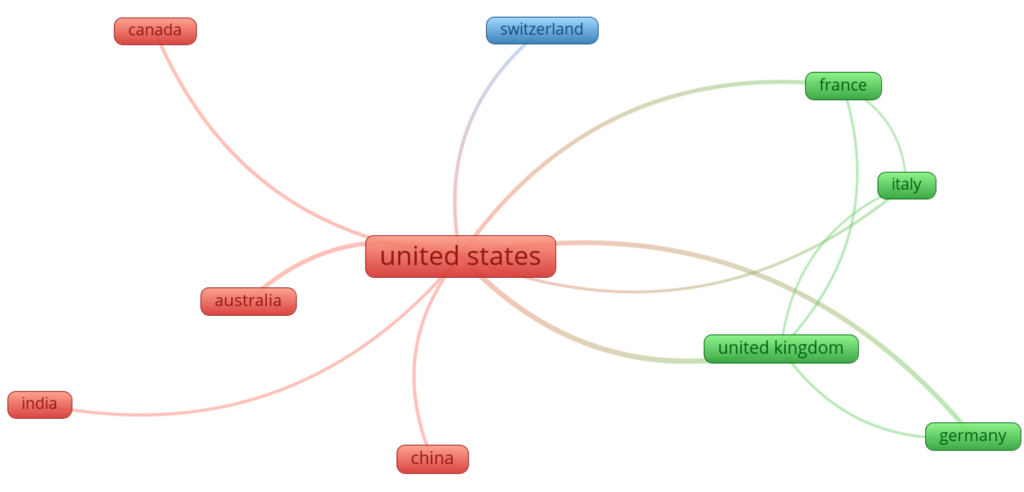
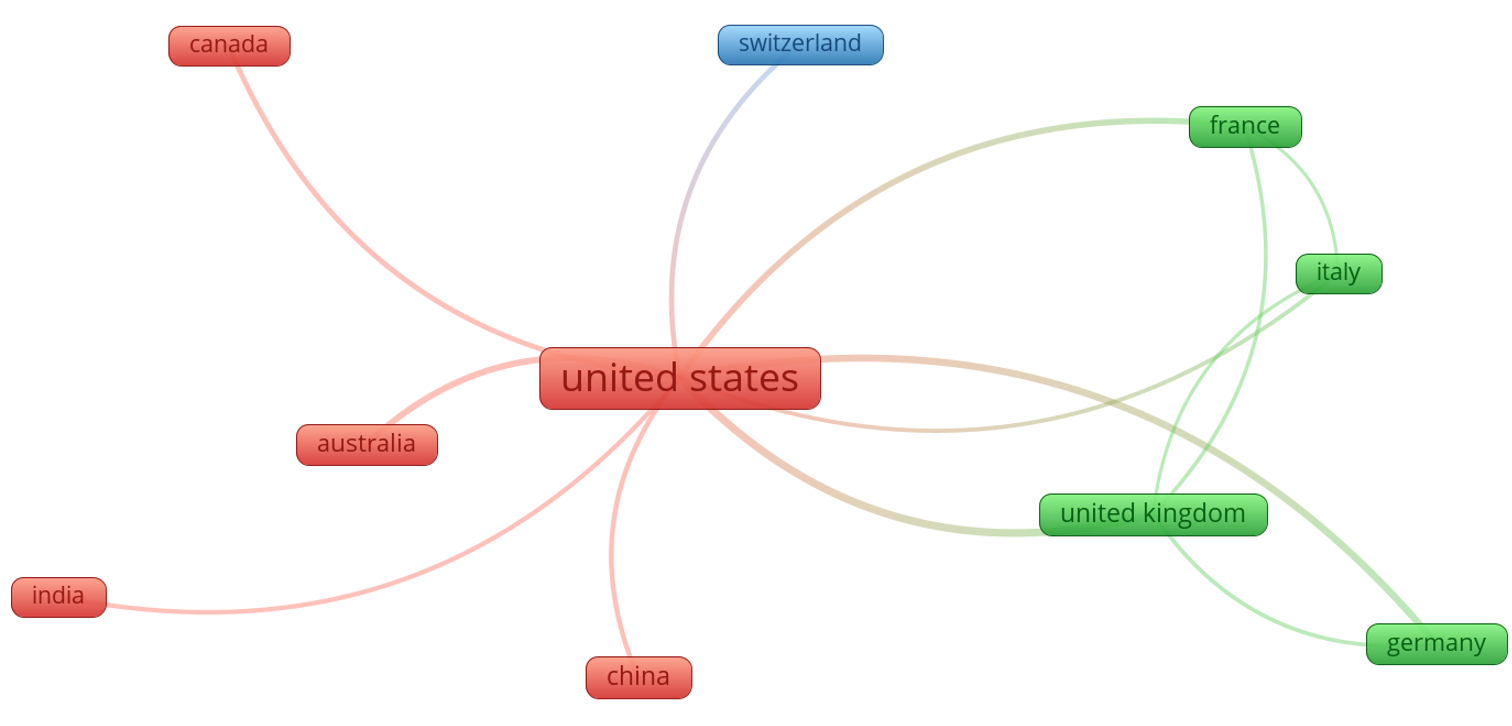
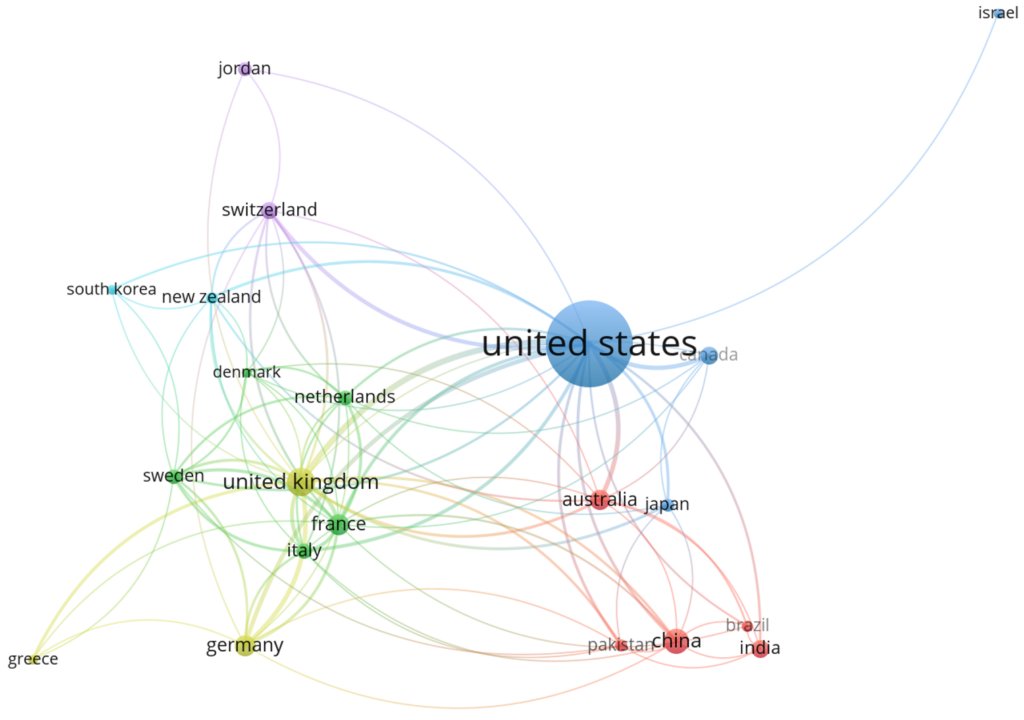
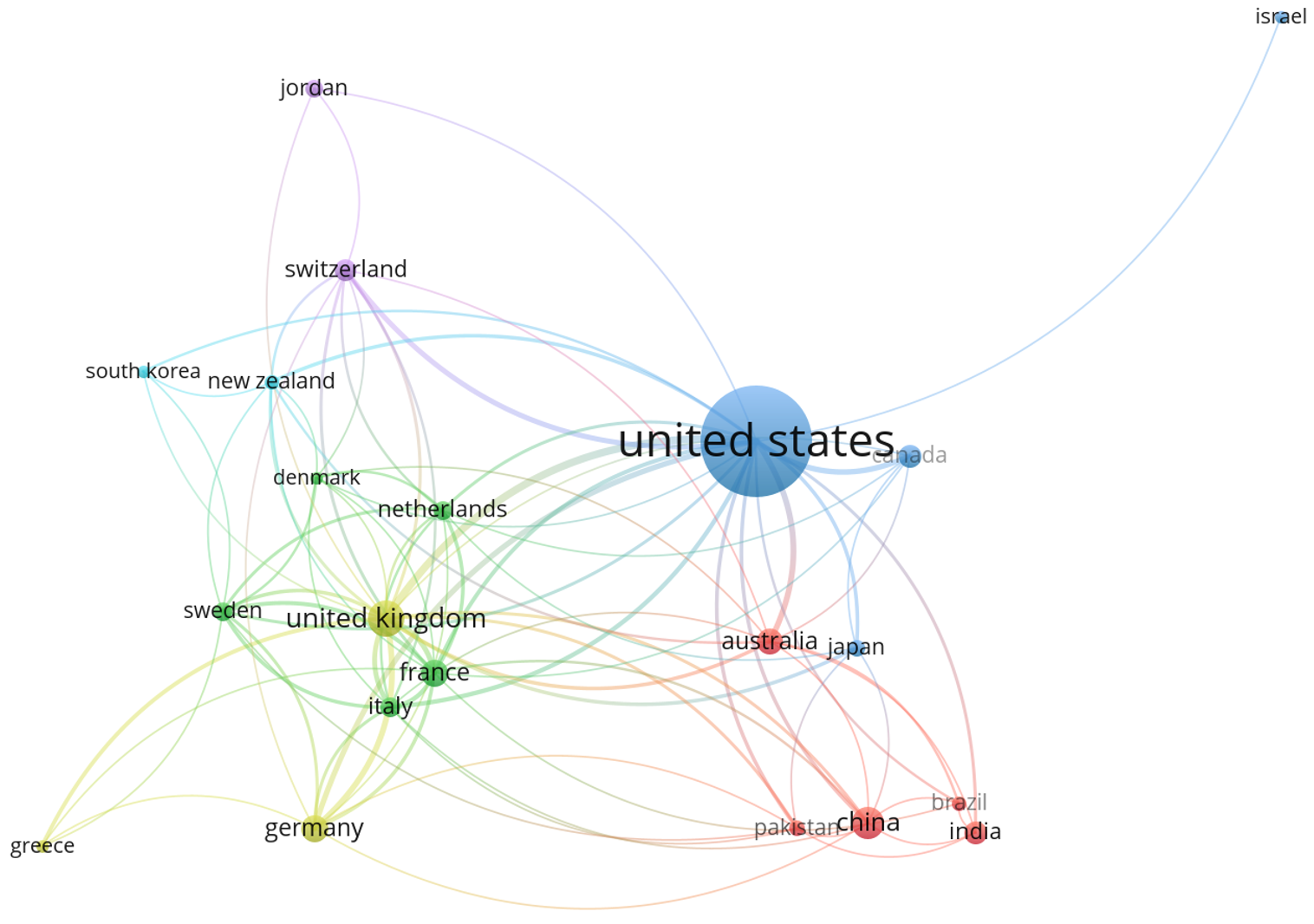
Figure 6 shows the top ten countries that collaborated on biosafety and biosecurity studies during the review period. Three clusters (green, red, and blue) were identified, in which the USA was the main collaborator in the red cluster, the United Kingdom was the main collaborator in the green cluster, and Switzerland was the main collaborator in the blue cluster. In general, the USA collaborates with China, India, Canada, and Australia. It has collaborations with the United Kingdom, Italy, Germany, and France, all from the green cluster.
Figure 7 illustrates co-authorship among various countries, where each node represents a country, and each edge indicates a bilateral or multilateral relationship. After the analysis, five clusters were identified (purple, red, blue, orange, and green). Node size reflects the relative influence or centrality of each country in the network. The United States (purple cluster) emerged as the most prominent node, suggesting a high degree of connectivity and influence in global affairs. Other significant nodes include the United Kingdom, Germany, and China, each of which demonstrates substantial linkages with other nations.
Authors and countries with the highest number of citations
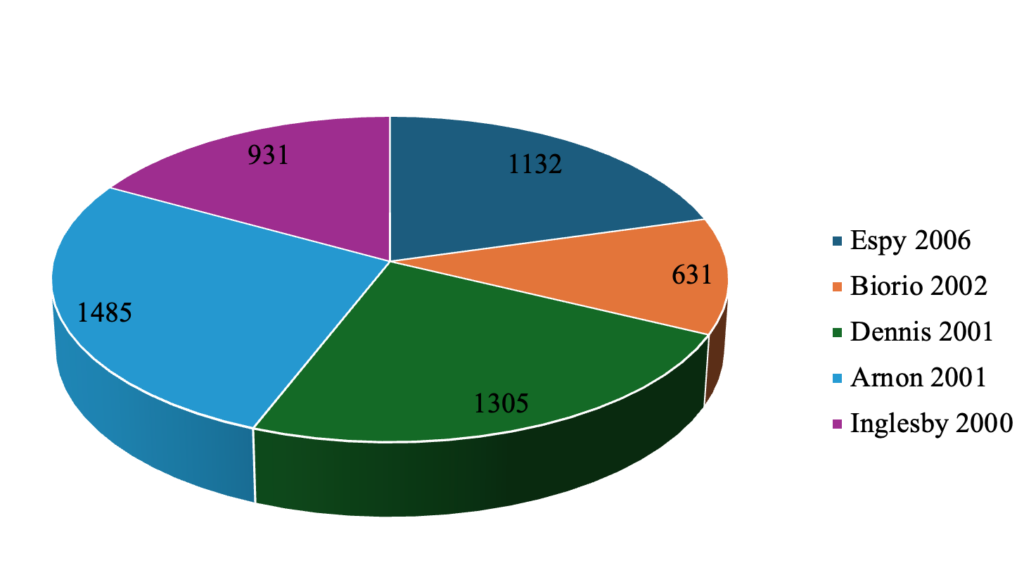
Figure 8 summarizes the authors with the highest number of publications during the review period. A paper published by Armon in 2001 had the highest number of citations (1485), followed by Dennis (2001) with 1305 citations and Espy (2006) with 1132 citations. Table 2 summarizes the countries with the highest number of citations. The United States had the highest number of citations (19643) as of December 2025.
Discussion
This bibliometric analysis maps the evolution of global bioterrorism and biosecurity research from 1997 to 2025 and reveals a field strongly influenced by recurring and emerging biological threats. These findings demonstrate substantial geographic disparities in research productivity, with high-income countries contributing the majority of publications, whereas low- and middle-income countries, despite being disproportionately vulnerable to biosafety and biosecurity breaches, remain significantly underrepresented. By systematically quantifying these patterns, the analysis underscores persistent global inequities in scientific output and highlights critical research gaps that continue to separate well-resourced regions from those at higher risk.
The most significant finding of this study is that the research output on biosafety and biosecurity is highly event-driven. This is evident from the significant increase in the number of publications after 2000, which peaked in 2004, directly corresponding to the global response to the 2001 anthrax attacks. This attack served as a catalyst that shaped the trajectory of modern biodefense policies in most countries [63]. Conversely, this pattern confirms that real-world security threats function as primary catalysts for scholarly output and funding opportunities. This phenomenon is well documented in biosafety and biosecurity research publications in major scholarly databases [64]. The subsequent fluctuations and notable drop between 2018 and 2019 are a good illustration of persistent failure to maintain sustained research output, with attention spiking episodically rather than being embedded in a long-term biosafety and biosecurity mitigation strategy [65]. Beyond the surge in anthrax‑related research output in 2001, the slight increase in publications from 2020 onward observed in this analysis is closely associated with the COVID‑19 pandemic, which exposed significant systemic vulnerabilities and renewed global debate on the increasingly blurred boundary between naturally occurring outbreaks and deliberate biological threats [66]. This reactive pattern aligns with assessments from the Global Health Security Index, which emphasizes that despite increased awareness, the world remains dangerously underprepared for future biological catastrophes [67].
A clear concentration of research output is evident in this bibliometric analysis, with the United States contributing the largest share of publications. This reflects the country’s leading role in biodefense policy and funding, supported by efforts such as the National Biodefense Strategy and major investments made after anthrax attacks [68-70]. The strong performance of other high-income countries, including the United Kingdom and Australia, reflects their sustained commitment to building robust national biosafety and biosecurity frameworks. These nations have prioritized strengthening laboratory safety standards, enhancing surveillance systems, and developing coordinated biodefense strategies, which collectively contribute to their substantial research output in this field [71]. However, these geographic imbalances in biosafety and biosecurity research highlight a persistent and concerning gap in global research capacity. While high-income countries dominate scientific output due to stronger funding, infrastructure, and established research networks, many low- and middle-income countries (LMICs) continue to face fundamental challenges, including limited laboratory capacity, inadequate surveillance systems, and insufficient investment in biosecurity initiatives that hinder their ability to contribute to and benefit from advances in the field [72]. This imbalance is reflected in the region’s notably low publication output. For example, within Africa, only a few countries, such as South Africa, Ethiopia, and Kenya, have made measurable contributions to biosafety and biosecurity research, underscoring the broader capacity gaps across the continent.
Consequently, this imbalance in research capacity feeds into a broader skew in global research focus. Consequently, the research agenda is disproportionately shaped by the priorities of the Global North, potentially overlooking context-specific threats in regions that are often on the frontlines of emerging infectious diseases [73]. These disparities are especially evident when examining specific areas of biosafety and biosecurity practices. For example, in high-income countries (HICs) such as the United States and member states of the European Union, biosafety and biosecurity systems are supported by long-standing regulatory frameworks, substantial financial investment, and well-developed institutional capacity. These mature and legally enforceable structures provide robust oversight, standardized laboratory protocols, and strong accountability mechanisms that collectively enable sustained research productivity and innovation [74, 75]. In contrast, many low- and middle-income countries (LMICs) operate with limited laboratory infrastructure, fragmented regulatory oversight, and constrained financial resources, resulting in biosafety and biosecurity systems that are far less developed. These constraints hinder effective risk management, reduce research output, and limit the ability of LMICs to detect, prevent, and respond to biological threats with the same level of rigor as in high-income settings [76]. The 2014–2016 Ebola outbreak in West Africa exemplified this, in which critical gaps in basic biosafety protocols contributed to its initial spread among local communities, resulting in the death of thousands of people [77].
A further regional illustration of these disparities can be seen in parts of the Middle East and South Asia. Middle Eastern countries, such as Jordan, Iran, and Israel, along with neighboring nations like India, have played a significant role in advancing biosafety and biosecurity capacities in the broader Asian region. However, in recent years, this region has frequently become an epicenter of biosafety and biosecurity-related challenges due to persistent geopolitical instability. Such instability has strained national health security systems and, in some cases, weakened biological containment frameworks, thereby complicating efforts to prevent, detect, and respond effectively to biological threats [78]. Amidst these developments, countries such as Jordan have significantly contributed through state-led initiatives to fortify the Biological Weapons Convention (BWC) in their statutes. By hosting the internationally supported Jordan Biosafety and Biosecurity Center, Jordan enhances regional resilience through a ‘train-the-trainer’ model, effectively addressing the dual-use dilemma inherent in the region’s expanding biotechnology sectors [46]. This approach positions the Middle East as an active contributor in shaping norms and mechanisms for mitigating biological threats. Jordan is a significant contributor to regional biosecurity and genomics research, establishing itself as a hub for scientific capacity in the Levant. The Jordanian National Biosecurity System, developed in collaboration with research institutions, is central to this effort. Other studies by the Jordan University of Science and Technology (JUST) have characterized the genetic diversity of pathogens and provided data for national risk assessment. [79]. Furthermore, research from King Abdullah University Hospital has utilized next-generation sequencing to track hospital-acquired infections, directly informing the national biosafety protocols and antimicrobial stewardship programs [24, 80, 81]. These institution-led studies have not only strengthened Jordan’s domestic biosecurity framework but also provided a model for pathogen surveillance and biorisk management that is shared with neighboring countries.
The infrastructure divide was equally profound across all regions. High-income countries invest billions in high-containment laboratories and advanced surveillance systems, whereas LMICs often struggle with unreliable electricity, insufficient safety equipment, and broken autoclaves [49]. The COVID-19 vaccine disparity, in which HICs secured vast stockpiles, while LMICs faced critical shortages, demonstrated how resource inequality directly translated into security vulnerability [82]. The human capital gap further exacerbates these challenges because HICs support the professional ecosystems of trained experts, whereas many LMICs rely on ad hoc project-dependent training, leading to high turnover and inconsistent oversight [50, 83].
A critical thematic finding is the field’s maturation from a narrow focus on “bioterrorism” towards a broader, more interdisciplinary concept of “biosecurity” and “health security.” While the disciplinary focus remains on medicine and life sciences, strong evidence from the social sciences indicates the essential integration of policy, ethics, law, and risk communication [84]. This interdisciplinarity is visually confirmed by keyword co-occurrence analysis, in which nodes such as “biological warfare” (security), “human” (public health), and “information” (communication) are centrally connected, echoing the argument for an “all-of-society” approach that integrates diverse domains of knowledge [73, 85]. The evolution of key journals reflects this trend, with the steady rise of Health Security demonstrating a field moving beyond intentional terrorism to encompass pandemics, laboratory safety, and strengthening health systems.
Despite ongoing research efforts, a significant disconnect persists between the theories produced in academic circles and the real-world skills required. This analysis reinforces what many have observed: healthcare professionals, especially those on the frontlines, often find themselves ill-prepared owing to insufficient formal training in bioterrorism preparedness. This critical issue requires the attention of all stakeholders [24]. Local health departments are often chronically underfunded and undertrained, indicating that translating academic research into tangible readiness remains a paramount and unmet challenge. This implementation gap suggests that future research should focus not only on generating new knowledge but also on effective knowledge translation and capacity building in LMICs through sustainable international partnerships [50, 86].
Citation analysis demonstrates that influence within bioterrorism and biosecurity research is anchored by a few seminal works, with Arnon and Schechter [87] emerging as the most frequently cited study because it provided one of the earliest and most authoritative consensus-based assessments of botulinum toxin as a biological weapon. In their landmark paper, the authors characterized botulinum toxin as “the most poisonous substance known” and emphasized its extreme potency, relative ease of production, and potential for aerosol or foodborne dissemination, factors that elevate it to one of the highest-risk biological agents owing to its lethality and the prolonged respiratory support required in severe cases [13, 87]. Their work also offered detailed clinical guidance on the early recognition of botulism, urgent antitoxin administration, and coordinated public health actions, thereby forming a foundational operational framework for U.S. biodefense planning during a period when global attention was intensified by the 2001 anthrax attacks [69, 70]. This partly explains why Arnon’s publication accumulated 1,485 citations, surpassing Dennis [88], who garnered 1,305 citations through their contributions to biodefense system strengthening, and Espand, Uhl [89] with 1,132 citations for advancing molecular diagnostic approaches crucial to biothreat detection. Moreover, the overwhelming citation dominance of the United States, with 19,643 total citations, reflects its extensive biodefense research infrastructure and leadership through institutions such as the Centers for Disease Control and Prevention (CDC) and the U.S. Army Medical Research Institute of Infectious Diseases, which have historically shaped global biosecurity discourse [90].
The findings of this bibliometric analysis paint a picture of a research field catalyzed by crises, dominated by a few nations, and evolving towards a more holistic and interdisciplinary understanding of biological threats. The consistent growth of integrated approaches reflects the recognition that technical solutions alone are insufficient without governance frameworks to support them. The need for international collaboration through frameworks such as the Biological Weapons Convention (BWC) and the Global Health Security Agenda (GHSA) is more critical than ever [91]. However, as emerging biotechnologies, such as CRISPR and synthetic biology, accelerate, existing governance structures become strained [92, 93]. Managing dual-use research of concern (DURC) in a fraught geopolitical landscape remains a pressing and unresolved challenge [92]. Therefore, future efforts must prioritize closing the implementation gap, fostering equitable global partnerships, and developing interdisciplinary governance models that can address biological risks.
This study offers a thorough overview; however, it is constrained by its dependence on the Scopus database, which might overlook non-English publications and gray literature (such as government reports and policy white papers), which are particularly relevant to this field. Future bibliometric studies should include these sources to provide a more comprehensive understanding of biosecurity.
Conclusion
The findings from this bibliometric analysis showed that publication trends in bioterrorism and biosecurity research during the review period were event-driven, with notable increases after major incidents. Research output is dominated by high-income countries, particularly the United States. Significant gaps remain in low-resource countries despite increasing global collaboration. Strengthening global biosafety and biosecurity requires sustained collaboration and equitable partnerships with African countries.
What is already known about the topic
- Bioterrorism and biosecurity pose significant threats to public health and require coordinated preparedness and response strategies to mitigate their impact.
- Healthcare providers often report feeling underprepared to manage bioterrorism-related incidents, despite recognizing their importance.
- Developed countries have established robust biosafety and biosecurity frameworks, whereas many low- and middle-income countries face infrastructure and resource challenges in this regard.
- Research output in bioterrorism and biosecurity has increased significantly, especially after the major global health crises.
What this study adds
- This study provides the first comprehensive bibliometric analysis of bioterrorism and biosecurity research over two decades.
- This identifies key global trends, influential authors, and leading institutions that shape the field.
- This analysis highlights the disconnection between academic research and practical preparedness, particularly in low-resource settings such as India.
- This underscores the need for interdisciplinary collaboration and capacity building to improve biosecurity readiness.
Acknowledgements
The authors acknowledge the support of the Aga Khan University Library for providing access to research resources and institutional guidance during the study. We also extend our appreciation to our colleagues at the School of Nursing and Midwifery and the Department of Biomedical Sciences for their valuable insights during the conceptualization of this work.
Authors´ contributions
Festus Mulakoli conceptualized the study, led the bibliometric analysis, and drafted the manuscript. Samwel Gesaka and John Mbataru provided critical revisions, contributed to the discussion and conclusion sections, and ensured methodological rigor. All authors have reviewed and approved the final manuscript.
| Table 1: Top five leading affiliations of authors and fields of study in bioterrorism and biosecurity research (1997–2025) | |
|---|---|
| Affiliation of authors | Publication (n) |
| Centers for Disease Control and Prevention | 30 |
| U.S. Army Medical Research Institute of Infectious Diseases | 29 |
| University of Pittsburgh Medical Center | 26 |
| Johns Hopkins University | 24 |
| National Institutes of Health (NIH) | 21 |
| Field of study (Discipline) | |
| Medicine | 577 |
| Social Sciences | 265 |
| Biochemistry, Genetics, and Molecular Biology | 258 |
| Immunology and Microbiology | 258 |
| Environmental Science | 188 |
| Table 2: Top five countries with the most citations of biosafety and biosecurity research | ||
|---|---|---|
| Country | Publication (n) | Number of citations |
| United States | 570 | 19643 |
| United Kingdom | 66 | 1934 |
| Germany | 37 | 1531 |
| France | 37 | 1018 |
| Australia | 35 | 697 |








References
- Jansen HJ, Breeveld FJ, Stijnis C, Grobusch MP. Biological warfare, bioterrorism, and biocrime. Clin Microbiol Infect [Internet]. 2014 Jun 16 [cited 2026 Apr 01];20(6):488-96. doi: 10.1111/1469-0691.12699
- Ostfield ML. Strengthening Biodefense Internationally: Illusion and Reality. Biosecur Bioterror [Internet]. 2008 Sep [cited 2026 Apr 01];6(3):261–8. doi: 10.1089/bsp.2008.0033
- Dhawan B, Desikan-Trivedi P, Chaudhry R, Narang P. Bioterrorism: a threat for which we are ill prepared. Natl Med J India [Internet]. 2001 Jul-Aug [cited 2026 Apr 01];14(4):225-30. Available from: https://www.ncbi.nlm.nih.gov/pubmed/1154753
- Long CM, Marzi A. Biodefence research two decades on: worth the investment? Lancet Infect Dis [Internet]. 2021 Aug [cited 2026 Apr 01];21(8):e222-e233. doi: 10.1016/S1473-3099(21)00382-0
- D’Amelio E, Gentile B, Lista F, D’Amelio R. Historical evolution of human anthrax from occupational disease to potentially global threat as bioweapon. Environ Int [Internet]. 2015 Dec [cited 2026 Apr 01];85:133-46. doi: 10.1016/j.envint.2015.09.009
- Rosenau W. Aum Shinrikyo’s Biological Weapons Program: Why Did it Fail? Studies in Conflict & Terrorism [Internet]. 2001 Jul [cited 2026 Apr 01];24(4):289–301. doi: 10.1080/10576100120887
- Grundmann O. The current state of bioterrorist attack surveillance and preparedness in the US. Risk Manag Healthc Policy [Internet]. 2014 Oct 9 [cited 2026 Apr 01];7:177-87. doi: 10.2147/rmhp.s56047
- Linger SP, Brown MJ, Daniel WB, Ivey AD, Street GE, McPherson TN, et al. Sensing biological threats. GeoSpatial Solutions [Internet]. 2005 [cited 2026 Apr 01];15(4):58. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-17444421870&partnerID=40&md5=b01f02fee6efd38f6eccd0b167f80b8e
- Verma AK, MS, N, RT, KD, SVS. Glanders-A Re-emerging Zoonotic Disease: A Review. J of Biological Sciences [Internet]. 2013 Dec 15 [cited 2026 Apr 01];14(1):38–51. doi: 10.3923/jbs.2014.38.51
- Revathy AR, Menezes G. Overview of bioterrorism and its agents. International Journal of Pharma and Bio Sciences [Internet]. 2013 [cited 2026 Apr 01];4(3):B647-B53. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-84881276170&partnerID=40&md5=b205c690d369ac7d72ad0ff4f3694229
- Janoutová J, Filipčíková R, Bílek K, Janout V. Biological agents of bioterrorism – preparedness is vital. Epidemiol Mikrobiol Imunol [Internet]. 2020 Winter [cited 2026 Apr 01];69(1):42-47. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85083899202&partnerID=40&md5=5b6d73e8f43fc88064f389033364e5d3
- Centers for Disease Control and Prevention (US), Department of Health and Human Services (US). Possession, Use, and Transfer of Select Agents and Toxins; Biennial Review of the List of Select Agents and Toxins and Enhanced Biosafety Requirements. Final rule. Fed Regist [Internet]. 2017 Jan 19 [cited 2026 Apr 01];82(12):6278-94. Available from: https://www.ncbi.nlm.nih.gov/pubmed/28106357
- Karwa M, Currie B, Kvetan V. Bioterrorism: Preparing for the impossible or the improbable. Crit Care Med [Internet]. 2005 Jan [cited 2026 Apr 01];33(Supplement):S75–95. doi: 10.1097/01.CCM.0000151070.56915.22
- Green MS, LeDuc J, Cohen D, Franz DR. Confronting the threat of bioterrorism: realities, challenges, and defensive strategies. Lancet Infect Dis [Internet]. 2019 Jan [cited 2026 Apr 01];19(1):e2–13. doi: 10.1016/S1473-3099(18)30298-6
- Sharma D, Mishra A, Newaskar V, Khasgiwala A. Bioterrorism: Law Enforcement, Public Health & Role of Oral and Maxillofacial Surgeon in Emergency Preparedness. J Maxillofac Oral Surg [Internet]. 2016 [cited 2026 Apr 01];15(2):137-43. doi: 10.1007/s12663-015-0834-x
- De Lima RC, Quaresma JAS. Emerging technologies transforming the future of global biosecurity. Front Digit Health [Internet]. 2025 Jun [cited 2026 Apr 01];7:1622123. doi: 10.3389/fdgth.2025.1622123
- Cole LA. Risks of publicity about bioterrorism: anthrax hoaxes and hype. Am J Infect Control [Internet]. 1999 [cited 2026 Apr 01];27(6):470-3. doi: 10.1016/s0196-6553(99)70023-4
- Salerno RM, Hickok LT. Strengthening Bioterrorism Prevention: Global Biological Materials Management. Biosecurity and Bioterrorism: Biodefense Strategy, Practice, and Science [Internet]. 2007 Jun [cited 2026 Apr 01];5(2):107–16. doi: 10.1089/bsp.2006.0027
- Hurst DJ, Bobier CA. Dual-Use Research and Publication Policies: A Comparison of Journals in Life Sciences and Artificial Intelligence. Applied Biosafety [Internet]. 2025 Jun 1 [cited 2026 Apr 01];30(2):97–106. doi: 10.1089/apb.2024.0034
- Morse A. Bioterrorism Preparedness for Local Health Departments. Journal of Community Health Nursing [Internet]. 2002 Dec [cited 2026 Apr 01];19(4):203–11. doi: 10.1207/S15327655JCHN1904_01
- Kim MJ, Hall CM, Bonn M. Does International Travel Frequency Affect COVID-19 Biosecurity Behavior in the United States? IJERPH [Internet]. 2021 Apr 13 [cited 2026 Apr 01];18(8):4111. doi: 10.3390/ijerph18084111
- Terriff CM, Tee AM. Citywide pharmaceutical preparation for bioterrorism. American journal of health-system pharmacy: AJHP: official journal of the American Society of Health-System Pharmacists [Internet]. 2001 [cited 2026 Apr 01];58(3):233-7. doi: 10.1093/ajhp/58.3.233
- Alexander GC, Larkin GL, Wynia MK. Physicians’ preparedness for bioterrorism and other public health priorities. Acad Emerg Med [Internet]. 2006 Nov [cited 2026 Apr 01];13(11):1238-41. doi: 10.1197/j.aem.2005.12.022
- Nofal A, AlFayyad I, AlJerian N, Alowais J, AlMarshady M, Khan A, Heena H, AlSarheed AS, Abu-Shaheen A. Knowledge and preparedness of healthcare providers towards bioterrorism. BMC Health Serv Res [Internet]. 2021 Dec [cited 2026 Apr 01];21(1):426. doi: 10.1186/s12913-021-06442-z
- Stankovic C, Mahajan P, Ye H, Dunne RB, Knazik SR. Bioterrorism: Evaluating the preparedness of pediatricians in Michigan. Pediatr Emerg Care [Internet]. 2009 Feb [cited 2026 Apr 01];25(2):88-92. doi: 10.1097/PEC.0b013e318196ea81
- Appelt S, Rohleder AM, Invernizzi C, Mikulak R, Brinkmann A, Nitsche A, et al. Strengthening the United Nations Secretary-General’s Mechanism to an alleged use of bioweapons through a quality-assured laboratory response. Nat Commun [Internet]. 2021 May 25 [cited 2026 Apr 01];12(1):3078. doi: 10.1038/s41467-021-23296-5
- Enserink M, Lawler A. Research Chiefs Hunt for Details in Proposal for New Department. Science [Internet]. 2002 Jun 14 [cited 2026 Apr 01];296(5575):1944–5. doi: 10.1126/science.296.5575.1944
- Kamoie B. The National Response Plan: a new framework for homeland security, public health, and bioterrorism response. Journal of health law [Internet]. 2005 [cited 2026 Apr 01];38(2):287-318. Available from: https://www.ncbi.nlm.nih.gov/pubmed/16270671
- Eze U, Okafor N, Ozota G, Nworie K, Asogwa C, Richard I, et al. Assessment of the knowledge of healthcare workers on monkeypox in Nigeria. GMS Hyg Infect Control [Internet]. 2024 Aug 20 [cited 2026 Apr 01];19:Doc38. doi: 10.3205/dgkh000493
- Cubelo F. Policy reform on the qualification pathway of internationally educated nurses in Greenland and its relationship with the Danish system: a qualitative discourse analysis. Policy Polit Nurs Pract [Internet]. 2024 [cited 2026 Apr 01];25(3):172-81. doi: 10.1177/15271544241245975
- Rathish B, Pillay R, Wilson A, Pillay VV. Comprehensive review of bioterrorism. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Mar 27 [updated 2026 Jan] [cited 2026 Apr 01]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK570614/
- Tadesse MT, Ayalke LL, Tedla BF, Weldemeskel FA, Yirdaw AS, Abebe TA. Biosafety Management Practices Among Clinical Laboratory Professionals in Debre Berhan Town Governmental Hospitals, Ethiopia: A Cross-Sectional Study. Health Sci Rep [Internet]. 2025 Jan 27 [cited 2026 Apr 01];8(1):e70361. doi: 10.1002/hsr2.70361
- Trump BD, Florin MV, Perkins E, Linkov I. Biosecurity for synthetic biology and emerging biotechnologies: critical challenges for governance. In: Trump BD, Florin MV, Perkins E, Linkov I. Emerging threats of synthetic biology and biotechnology: addressing security and resilience issues. Dordrecht (NL): Springer [Internet]; 2021 [cited 2026 Apr 01]. p. 1-12. doi: 10.1007/978-94-024-2086-9_1
- Pourian HR, Khoobdel M, Alizadeh M. Stored-grains pests and their control with emphasis on military food warehouses in Iran: a review. J Mil Med [Internet]. 2019 [cited 2026 Apr 01];21(4):313-324. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85072517179&partnerID=40&md5=bd86b0c15e2a549fd52012cf36c56e67
- Majid MA. Meeting the biosecurity concerns of genetically modified organisms (GMOs) through Malaysia’s Strategic Trade Act 2010 and Biosafety Act 2007. In: New Forest, UK [Internet]; 2012 [cited 2026 Apr 01]. p. 325–35. Available from: http://library.witpress.com/viewpaper.asp?pcode=EID12-029-1 doi: 10.2495/EID120291
- European Biosafety Association (EBSA). Welcome to EBSA – European Biosafety Association [Internet]. 2026 [cited 2026 Apr 01]. Available from: https://www.ebsaweb.eu/
- Namdeo SK, Zhang JY. Rethinking science diplomacy and global biosecurity: challenges, emerging practices and the way forward. International Affairs [Internet]. 2024 Nov 4 [cited 2026 Apr 01];100(6):2623–35. doi: 10.1093/ia/iiae187
- AL-Eitan L, Alnemri M. Biosafety and biosecurity in the era of biotechnology: The Middle East region. Journal of Biosafety and Biosecurity [Internet]. 2022 Nov 21 [Version of Record 2022 Dec 21; cited 2026 Apr 01];4(2):130–45. doi: 10.1016/j.jobb.2022.11.002
- Al Eitan L, Ali M. Biosafety system evaluations across Middle Eastern biotechnology sectors. Middle East Biosecur Rev [Internet]. 2025 [cited 2026 Apr 01].
- World Health Organization (Eastern Mediterranean Office). Accelerating action on health emergency preparedness and response [Internet]. Cairo (Egypt): WHO Regional Office for the Eastern Mediterranean; 2025 [cited 2026 Apr 01]; [about 3 screens]. Available from: https://www.emro.who.int/media/news/accelerating-action-on-health-emergency-preparedness-and-response-2.html
- United Nations Interregional Crime and Justice Research Institute (UNICRI). Threat response and risk mitigation: security governance [Internet]. Turin (Italy): UNICRI; 2025 [cited 2026 Apr 01]; [about 4 screens]. Available from: https://unicri.org/threat-response-and-risk-mitigation-security-governance
- Lederman Z, Lai Ivan LH, Liu CN, Hames A, Menikoff J. Unit 731 and history-informed professional identity formation. The Lancet [Internet]. 2025 Feb [cited 2026 Apr 01];405(10477):465. doi: 10.1016/S0140-6736(24)02682-5
- Shadel BN, Chen JJ, Newkirk RW, Lawrence SJ, Clements B, Evans RG. Bioterrorism risk perceptions and educational needs of public health professionals before and after September 11, 2001: a national needs assessment survey. JPHMP [Internet]. 2004 [cited 2026 Apr 01];10(4):282-9. doi: 10.1097/00124784-200407000-00004
- Asia Centre for Health Security. Regional biosafety and laboratory preparedness in Southeast Asia. Singapore (SG): Asia Centre for Health Security; 2025 [cited 2026 Apr 01]; [about 2 screens]. Available from: https://asia-chs.org/event/laboratory-biosafety-and-biosecurity-workshop-2024/
- Caballero-Anthony M, Montesclaros JML, Jeselyn, Trajano JC. Dual-Use Research of Concern Landscape in Southeast Asia: Prioritization, Gaps, and Challenges. Appl Biosaf [Internet]. 2025 Jun 1 [cited 2026 Apr 01];30(2):178–88. doi: 10.1089/apb.2024.0055
- Medina PB, Armon S, Bin Abdul Aziz MF, Cheong IH, De Leon MP, Drobysz S, Fikry Bin Haji Abdul Momin MH, Garcia DL, Iskandriati D, Kozlakidis Z, Cui L, Mao S, Miranda ME, Mya KM, Nallenthiran L, Obusan MC, Phimmakong K, Sabai P, Saejung C, Sathasivam HP, Jafar FLB, Vitor RJS, Yabes AM, Calaor AB, Vijayan V, Lin RTP. A Review of Regulatory Frameworks for Biobanking in Southeast Asia. Biopreservation and Biobanking [Internet]. 2025 Jun 1 [cited 2026 Apr 01];23(3):165–76. doi: 10.1089/bio.2024.0044
- Association of Southeast Asian Nations. ASEAN Leaders’ Declaration on Strengthening Biosafety and Biosecurity [Internet]. Jakarta (Indonesia): Association of Southeast Asian Nations; 2024 Oct 9 [cited 2026 Apr 01]. 2 p. Available from: https://asean.org/wp-content/uploads/2024/10/6-ASEAN-Leaders_-Declaration-on-Strengthening-Regional-Biosafety-and-Biosecurity.pdf
- Johns Hopkins Center for Health Security. Southeast Asia Strategic Dialogue on Biosecurity [Internet]. Baltimore (MD): Hopkins Center for Health Security; 2023 Apr 26-28 [cited 2026 Apr 01]. 21 p. Available from: https://centerforhealthsecurity.org/southeast-asia-strategic-dialogue-on-biosecurity-bios
- Ondoa P, Datema T, Keita-Sow MS, Ndihokubwayo JB, Isadore J, Oskam L, et al. A new matrix for scoring the functionality of national laboratory networks in Africa: introducing the LABNET scorecard. Afr J Lab Med [Internet]. 2016 Oct 31 [cited 2026 Apr 01];5(3):498. doi: 10.4102/ajlm.v5i3.498
- Shrivastava R, Poxon R, Rottinghaus E, Essop L, Sanon V, Chipeta Z, et al. Leveraging gains from African Center for Integrated Laboratory Training to combat HIV epidemic in sub-Saharan Africa. BMC Health Serv Res [Internet]. 2021 Dec [cited 2026 Apr 01];21(1):22. doi: 10.1186/s12913-020-06005-8
- Rushton S, Kamradt-Scott A. The revised International Health Regulations and outbreak response. In: The politics of surveillance and response to disease outbreaks: the new frontier for states and non-state actors. 1st ed [Internet]. London (UK): Routledge; 2015 [cited 2026 Apr 01]. p. 114–132. Purchase or subscription required to access full text.
- Africa CDC. Africa CDC Annual Progress Report 2019 [Internet]. Addis Ababa (Ethiopia): Africa CDC; 2021 Aug 11 [cited 2026 Apr 01]. 21 p. Available from: https://africacdc.org/download/africa-cdc-annual-progress-report-2019/
- Dhruvin PaZ, Y. Biorisk Governance in Africa: A Landscape Analysis of Policies, Challenges, and Emerging Trends [Internet]. Annapolis (Maryland): Tech Governance Project; 2024 Oct [cited 2026 Apr 01]. 14 p. Available from: https://www.tgov.org/post/biorisk-governance-in-africa-landscape-analysis
- World Health Organization. National stakeholder workshop for piloting the WHO global guidance framework for the responsible use of the life science in Uganda [Internet]. Geneva (Switzerland): World Health Organization; 2023 Nov 8 [cited 2026 Apr 01]; [about 2 screens]. Available from: https://www.who.int/news/item/08-11-2023-national-stakeholder-workshop-for-piloting-the-who-global-guidance-framework-for-the-responsible-use-of-the-life-science-in-uganda
- Africa Society of Laboratory Medicine. Continental biosafety and biosecurity strengthening updates [Internet]. Nairobi (Kenya): 2025 Jun 13 [cited 2026 Apr 01]; [about 2 screens]. Available from: https://aslm.org/east-africa-unites-in-nairobi-to-shape-2025-2030-biosafety-and-biosecurity-strategy/
- Lee S, Kim Y. Predictors of bioterrorism preparedness among clinical nurses: A cross-sectional study. Nurse Education Today [Internet]. 2023 Mar [cited 2026 Apr 01];122:105727. doi: 10.1016/j.nedt.2023.105727
- Rebmann T. Early warnings: health care preparedness. Occup Health Saf [Internet]. 2005 Nov [cited 2026 Apr 01];74(11):53-6, 97. Available from: https://www.ncbi.nlm.nih.gov/pubmed/16355587
- Bebber R, Liberman A. Bioterrorism preparedness through the lens of complexity theory. IJPP [Internet]. 2008 [cited 2026 Apr 01];3(5/6):328. doi: 10.1504/IJPP.2008.02098
- Donthu N, Kumar S, Mukherjee D, Pandey N, Lim WM. How to conduct a bibliometric analysis: An overview and guidelines. Journal of Business Research [Internet]. 2021 Sep [cited 2026 Apr 01];133:285–96. doi: 10.1016/j.jbusres.2021.04.070
- Department of biotechnology, RV college of engineering., Vatsa KD. Bioterrorism Preparedness and Response Strategies: Safeguarding Against Biohazards. IJSREM [Internet]. 2023 Jul 27 [cited 2026 Apr 01];07(07). doi: 10.55041/IJSREM24859
- Kim Y, Lee H. Bioterrorism-Related Training Programs for Healthcare Workers: A Systematic Review. Sage Open [Internet]. 2023 Oct [cited 2026 Apr 01];13(4):21582440231211373. doi: 10.1177/21582440231211373
- Kat AR, Nekorchuk DM, Holck PS, Hendrickson LA, Imrie AA, Effler PV. Hawaii Physician and Nurse Bioterrorism Preparedness Survey. Prehosp Disaster Med [Internet]. 2006 Dec [cited 2026 Apr 01];21(6):404–13. doi: 10.1017/S1049023X00004118
- Koblentz GD. From biodefence to biosecurity: the Obama administration’s strategy for countering biological threats. Int Aff [Internet]. 2012 Jan [cited 2026 Apr 01];88(1):131-48. doi: 10.1111/j.1468-2346.2012.01061.x
- Gronvall GK, Wang L, McGrath PF, Cicero AJ, Yuan Y, Parker MI, Zhang W, Sun Y, Xue Y, Zhang J, Zhang X, Yu L, Song J, Trotochaud M. The Biological Weapons Convention should endorse the Tianjin Biosecurity Guidelines for Codes of Conduct. Trends Microbiol [Internet]. 2022 Dec [cited 2026 Apr 01];30(12):1119-20. doi: 10.1016/j.tim.2022.09.014
- Singh V, Chouhan APS. Microbiology as Forensic Tool in investigation of Bioterrorism. Indian J Forensic Med Pathol [Internet]. 2021 [cited 2026 Apr 01];14(3 Special Issue):651-7. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85134729304&partnerID=40&md5=5be37f193129336aae64c7d898d83051
- Schmidt M, López VT, Tobías M, Grinberg E, Merlinsky G. Social and environmental conflicts caused by agrochemical use in Salta, Santiago del Estero and Santa Fe, Argentina. Cien Saude Colet [Internet]. 2022 Mar [cited 2026 Apr 01];27(3):1061–72. doi: 10.1590/1413-81232022273.04852021
- Wilson N, Boyd M, Baker M, Nelson C. The 2021 Global Health Security (GHS) Index: Aotearoa New Zealand’s improving capacity to manage biological threats must now be consolidated. NZMJ [Internet]. 2022 Aug 19 [cited 2026 Apr 01];135(1560):89–98. doi: 10.26635/6965.5661
- Aquino LL, Wu JJ. Cutaneous manifestations of category A bioweapons. J Am Acad Dermatol [Internet]. 2011 Dec [cited 2026 Apr 01];65(6):1213.e1-1213.e15. doi: 10.1016/j.jaad.2010.08.040
- Bush LM, Perez MT. The Anthrax Attacks 10 Years Later. Ann Intern Med [Internet]. 2012 Jan 3 [cited 2026 Apr 01];156(1_Part_1):41–4. doi: 10.7326/0003-4819-155-12-201112200-00373
- Allen KC. Applications. In: Disaster Epidemiology [Internet]. Elsevier; 2018 [cited 2026 Apr 01]. p. 143–51. doi: 10.1016/B978-0-12-809318-4.00019-8
- Broom J, Broom A, Kirby E. Context-sensitive antibiotic optimization: a qualitative interviews study of a remote Australian hospital setting. J Hosp Infect [Internet]. 2018 Nov [cited 2026 Apr 01];100(3):265–9. doi: 10.1016/j.jhin.2018.06.002
- Walsh PF. Health Security Intelligence [Internet]. London (UK): Routledge; 2024 [cited 2026 Apr 01]. 13 p.
- Novossiolova T, Whitby S, Dando M, Shang L. Strengthening biological security after COVID-19: Using cartoons for engaging life science stakeholders with the Biological and Toxin Weapons Convention (BTWC). J Biosaf Biosecur [Internet]. 2022 Jun [cited 2026 Apr 01];4(1):68–74. doi: 10.1016/j.jobb.2022.03.001
- Gronvall G, Boddie C, Knutsson R, Colby M. One Health Security: An Important Component of the Global Health Security Agenda. Biosecurity and Bioterrorism: Biodefense Strategy, Practice, and Science [Internet]. 2014 Sep [cited 2026 Apr 01];12(5):221–4. doi: 10.1089/bsp.2014.0044
- Millett K. Financial Woes Spell Trouble for the Biological Weapons Convention. Health Security [Internet]. 2017 Jun 1 [cited 2026 Apr 01];15(3):320–2. doi: 10.1089/hs.2017.0030
- Bennett B, Cohen IG, Davies SE, Gostin LO, Hill PS, Mankad A, Phelan AL. Future-proofing global health: Governance of priorities. Global Public Health [Internet]. 2018 May 4 [cited 2026 Apr 01];13(5):519–27. doi: 10.1080/17441692.2017.1296172
- Rushton S, Kamradt-Scott A. The Politics of Surveillance and Response to Disease Outbreaks: The New Frontier for States and Non-State Actors [Internet]. London (UK): Routledge; 2016 [cited 2026 Apr 01]. Available from: https://www.routledge.com/The-Politics-of-Surveillance-and-Response-to-Disease-Outbreaks-The-New-Frontier-for-States-and-Non-state-Actors/Davies-Youde/p/book/9780815377771
- Truppa C, Yaacoub S, Valente M, Celentano G, Ragazzoni L, Saulnier D. Health systems resilience in fragile and conflict-affected settings: a systematic scoping review. Confl Health [Internet]. 2024 Jan 3 [cited 2026 Apr 01];18(1):2. doi: 10.1186/s13031-023-00560-7
- Haqaish WSA, Obeidat H, Patel P, Walker S. The Jordan Food and Drug Administration: Comparison of its Registration Process with Australia, Canada, Saudi Arabia and Singapore. Pharm Med [Internet]. 2017 Feb [cited 2026 Apr 01];31(1):21–30. doi: 10.1007/s40290-016-0172-4
- Almukhlifi Y, Crowfoot G, Wilson A, Hutton A. Disaster Knowledge, Skills, and Preparedness among Emergency Medical Services in Saudi Arabia. Disaster Med Public Health Prep [Internet]. 2024 Oct [cited 2026 Apr 01];18:e233. doi: 10.1017/dmp.2024.276
- Alkaabi N, Aldubayee M, Masuadi E, Alwan I, Babiker A. Opportunities, barriers, and expectations towards international voluntary medical missions among health trainees in Saudi Arabia. Sudan J Paed [Internet]. 2023 [cited 2026 Apr 01];187–98. doi: 10.24911/SJP.106-1696628750
- Berkley S. Even after COVID, the world’s vaccine strategy is failing. Nature [Internet]. 2022 Dec 15 [cited 2026 Apr 01];612(7940):377–377. doi: 10.1038/d41586-022-04423-8
- Schmidt M. A metric space for semantic containment: Towards the implementation of genetic firewalls. Biosystems [Internet]. 2019 Nov [cited 2026 Apr 01];185:104015. doi: 10.1016/j.biosystems.2019.104015
- Khan AS, Amara PS, Morse SA. Forensic public health. In: Microbial Forensics [Internet]. Elsevier; 2020 [cited 2026 Apr 01]. p. 105–22. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780128153796000088 doi: 10.1016/B978-0-12-815379-6.00008-8
- Trump BD, Galaitsi S, Appleton E, Bleijs DA, Florin M, Gollihar JD, Hamilton RA, Kuiken T, Lentzos F, Mampuys R, Merad M, Novossiolova T, Oye K, Perkins E, Garcia‐Reyero N, Rhodes C, Linkov I. Building biosecurity for synthetic biology. Molecular Systems Biology [Internet]. 2020 Jul [cited 2026 Apr 01];16(7):e9723. doi: 10.15252/msb.20209723
- Fischer I, Avrashi S, Oz T, Fadul R, Gutman K, Rubenstein D, Kroliczak G, Goerg S, Glöckner A. The behavioural challenge of the COVID-19 pandemic: indirect measurements and personalized attitude changing treatments (IMPACT). R Soc Open Sci [Internet]. 2020 Aug [cited 2026 Apr 01];7(8):201131. doi: 10.1098/rsos.201131
- Arnon SS, Schechter R, Inglesby TV, Henderson DA, Bartlett JG, Ascher MS, Eitzen E, Fine AD, Hauer J, Layton M, Lillibridge S, Osterholm MT, O’Toole T, Parker G, Perl TM, Russell PK, Swerdlow DL, Tonat K, For The Working Group On Civilian Biodefense. Botulinum Toxin as a Biological Weapon: Medical and Public Health Management. JAMA [Internet]. 2001 Feb 28 [cited 2026 Apr 01];285(8):1059. doi: 10.1001/jama.285.8.1059
- Dennis C. The bugs of war. Nature [Internet]. 2001 May 17 [cited 2026 Apr 01];411(6835):232–5. doi: 10.1038/35077161
- Espy MJ, Uhl JR, Sloan LM, Rosenblatt JE, Cockerill FR, Smith TF. Detection of Vaccinia Virus, Herpes Simplex Virus, Varicella-Zoster Virus, and Bacillus anthracis DNA by LightCycler Polymerase Chain Reaction After Autoclaving: Implications for Biosafety of Bioterrorism Agents. Mayo Clinic Proceedings [Internet]. 2002 Jul [cited 2026 Apr 01];77(7):624–8. doi: 10.4065/77.7.624
- Use of Anthrax Vaccine in Response to Terrorism: Supplemental Recommendations of the Advisory Committee on Immunization Practices. JAMA [Internet]. 2002 Dec 4 [cited 2026 Apr 01];288(21):2681. doi: 10.1001/jama.288.21.2681-JWR1204-3-1
- Imperiale MJ, Howard D, Casadevall A. The Silver Lining in Gain-of-Function Experiments with Pathogens of Pandemic Potential. In: Yamauchi Y, editor. Influenza Virus [Internet]. New York, NY: Springer New York; 2018 [cited 2026 Apr 01]. p. 575–87. Available from: http://link.springer.com/10.1007/978-1-4939-8678-1_28 doi: 10.1007/978-1-4939-8678-1_28
- Lea-Smith DJ, Hassard F, Coulon F, Partridge N, Horsfall L, Parker KDJ, Smith RDJ, McCarthy RR, McKew B, Gutierrez T, Kumar V, Dotro G, Yang Z, EBIC partners, Curtis TP, Golyshin P, Heaven S, Jefferson B, Jeffrey P, Jones DL, Le Corre Pidou K, Liu Y, Lyu T, Smith C, Yakunin A, Zhang Y, Krasnogor N. Engineering biology applications for environmental solutions: potential and challenges. Nat Commun [Internet]. 2025 Apr 14 [cited 2026 Apr 01];16(1):3538. doi: 10.1038/s41467-025-58492-0
- Zhang Y, Li Z, Milon Essola J, Ge K, Dai X, He H, Xiao H, Weng Y, Huang Y. Biosafety materials: Ushering in a new era of infectious disease diagnosis and treatment with the CRISPR/Cas system. Biosafety and Health [Internet]. 2022 Apr [cited 2026 Apr 01];4(2):70–8. doi: 10.1016/j.bsheal.2022.03.010
