Research | Open Access | Volume 9 (2): Article 64 | Published: 17 Apr 2026
Caregivers’ awareness, attitudes, uptake, and willingness toward the RTS,S/AS01 malaria vaccine, Oti Region, Ghana 2023
Menu, Tables and Figures
On Pubmed
On Google Scholar
Navigate this article
Tables
| Variable | n | % (95% CI) |
|---|---|---|
| Caregiver characteristics | ||
| Age (years) | ||
| 15–17 | 6 | 1.6 (0.6–3.5) |
| 17–28 | 189 | 51.5 (46.3–56.7) |
| 28–45 | 169 | 46.0 (40.9–51.2) |
| 45–65 | 3 | 0.8 (0.2–2.4) |
| Sex | ||
| Female | 342 | 93.2 (90.0–95.5) |
| Male | 25 | 6.8 (4.5–10.0) |
| Marital status | ||
| Married | 302 | 82.3 (78.0–85.9) |
| Not married* | 65 | 17.7 (14.1–22.0) |
| Education level | ||
| No formal education | 47 | 12.8 (9.7–16.6) |
| Primary | 82 | 22.3 (18.3–26.9) |
| JHS/Middle | 151 | 41.1 (36.1–46.3) |
| SHS/Vocational | 40 | 10.9 (8.0–14.6) |
| Tertiary | 47 | 12.8 (9.7–16.6) |
| Employment status | ||
| Employed | 268 | 73.0 (68.2–77.4) |
| Not employed | 99 | 27.0 (22.6–31.8) |
| Religion | ||
| Christianity | 205 | 55.9 (50.7–61.0) |
| Islam | 162 | 44.1 (39.0–49.3) |
| Ethnicity | ||
| Ewe | 128 | 34.9 (30.2–39.9) |
| Kotokoli | 121 | 33.0 (28.4–37.9) |
| Akan/Twi | 33 | 9.0 (6.4–12.4) |
| Basare | 26 | 7.1 (4.9–10.1) |
| Hausa | 13 | 3.5 (2.1–6.0) |
| Other | 46 | 12.5 (9.5–16.3) |
| Child characteristics | ||
| Sex of child | ||
| Male | 202 | 55.0 (49.8–60.1) |
| Female | 165 | 45.0 (39.9–50.2) |
| Age group (months) | ||
| 5–11 | 78 | 21.3 (17.4–25.9) |
| 12–23 | 112 | 30.5 (26.0–35.4) |
| 24–59 | 177 | 48.2 (43.1–53.4) |
| Awareness of malaria vaccine | ||
| Ever heard of a malaria vaccine (Yes) | 323 | 87.5 (83.7–90.5) |
| Heard of the RTS, S vaccine (Yes) | 221 | 59.9 (54.8–64.8) |
| Sources of information† | ||
| Healthcare providers | 321 | 87.0 (83.2–90.0) |
| Friends/family | 15 | 4.1 (2.5–6.7) |
| Radio/Television | 4 | 1.1 (0.4–2.8) |
| Social media | 4 | 1.1 (0.4–2.8) |
| Other sources | 16 | 4.3 (2.7–6.9) |
| Notes: • Values are presented as frequency (percentage with 95% confidence interval) • Not married includes single, divorced/separated, and widowed • †Multiple responses allowed; percentages may not sum to 100% | ||
Table 1: Socio-demographic Characteristics and Awareness of Malaria Vaccine among Caregivers (n = 367)
| Characteristics | Awareness | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Good (n = 264) | Poor (n = 103) | ||||
| Age group (years) | |||||
| 15–17 | 3 (50.0) | 3 (50.0) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 128 (67.7) | 61 (32.3) | 189 (100) | 2.10 (0.41–10.8), 0.372 | 1.96 (0.36–10.6), 0.438 |
| 28–45 | 131 (77.5) | 38 (22.5) | 169 (100) | 3.45 (0.67–17.7), 0.139 | 3.12 (0.58–16.8), 0.186 |
| 45–65 | 2 (66.7) | 1 (33.3) | 3 (100) | 2.00 (0.12–33.6), 0.620 | 1.88 (0.10–34.1), 0.650 |
| Sex | |||||
| Female | 248 (72.5) | 94 (27.5) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 16 (64.0) | 9 (36.0) | 25 (100) | 0.67 (0.29–1.57), 0.355 | 0.71 (0.29–1.75), 0.458 |
| Marital status | |||||
| Not married | 41 (63.1) | 24 (36.9) | 65 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Married | 223 (73.8) | 79 (26.2) | 302 (100) | 1.66 (0.95–2.89), 0.074 | 1.58 (0.87–2.85), 0.129 |
| Religion | |||||
| Christianity | 155 (75.6) | 50 (24.4) | 205 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Islam | 109 (67.3) | 53 (32.7) | 162 (100) | 0.66 (0.43–1.02), 0.061 | 0.71 (0.45–1.12), 0.142 |
| Ethnicity | |||||
| Akan/Twi | 20 (60.6) | 13 (39.4) | 33 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Kotokoli | 82 (67.8) | 39 (32.2) | 121 (100) | 1.37 (0.60–3.15), 0.454 | 1.31 (0.55–3.12), 0.545 |
| Ewe | 98 (76.6) | 30 (23.4) | 128 (100) | 2.13 (0.93–4.86), 0.072 | 1.98 (0.84–4.69), 0.118 |
| Hausa | 7 (53.8) | 6 (46.2) | 13 (100) | 0.76 (0.21–2.73), 0.679 | 0.81 (0.22–3.02), 0.754 |
| Basare | 17 (65.4) | 9 (34.6) | 26 (100) | 1.23 (0.42–3.60), 0.706 | 1.19 (0.39–3.62), 0.758 |
| Others | 40 (87.0) | 6 (13.0) | 46 (100) | 4.33 (1.39–13.5), 0.012 | 3.91 (1.18–12.9), 0.026 |
| Education level | |||||
| No education | 24 (51.1) | 23 (48.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 67 (81.7) | 15 (18.3) | 82 (100) | 4.28 (1.94–9.44), <0.001 | 3.96 (1.74–9.01), 0.001 |
| JHS/Middle | 111 (73.5) | 40 (26.5) | 151 (100) | 2.66 (1.36–5.19), 0.004 | 2.41 (1.18–4.92), 0.016 |
| SHS/Vocational | 30 (75.0) | 10 (25.0) | 40 (100) | 2.88 (1.16–7.17), 0.023 | 2.62 (1.01–6.80), 0.047 |
| Tertiary | 32 (68.1) | 15 (31.9) | 47 (100) | 2.04 (0.94–4.45), 0.071 | 1.88 (0.83–4.24), 0.130 |
| Employment status | |||||
| Not employed | 63 (63.6) | 36 (36.4) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 201 (75.0) | 67 (25.0) | 268 (100) | 1.71 (1.04–2.82), 0.034 | 1.62 (0.94–2.79), 0.081 |
| Sex of child | |||||
| Female | 113 (68.5) | 52 (31.5) | 165 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 151 (74.8) | 51 (25.2) | 202 (100) | 1.36 (0.88–2.10), 0.170 | 1.29 (0.81–2.05), 0.287 |
| Age of child (months) | |||||
| 5–11 | 49 (62.8) | 29 (37.2) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 | 78 (69.6) | 34 (30.4) | 112 (100) | 1.36 (0.73–2.54), 0.334 | 1.29 (0.67–2.48), 0.448 |
| 24–60 | 137 (77.4) | 40 (22.6) | 177 (100) | 2.02 (1.11–3.67), 0.022 | 1.86 (0.98–3.51), 0.056 |
Table 2: Factors Associated with Awareness of Malaria Vaccine among Caregivers (n = 367)
| Characteristics | Attitude | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Good (n = 309) | Poor (n = 58) | ||||
| Awareness | |||||
| Poor | 63 (61.2) | 40 (38.8) | 103 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Good | 246 (93.2) | 18 (6.8) | 264 (100) | 8.73 (4.66–16.4), <0.001 | 3.98 (2.05–7.71), <0.001 |
| Age group (years) | |||||
| 15–17 | 4 (66.7) | 2 (33.3) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 150 (79.4) | 39 (20.6) | 189 (100) | 1.92 (0.35–10.6), 0.453 | 1.78 (0.31–10.1), 0.514 |
| 28–45 | 152 (89.9) | 17 (10.1) | 169 (100) | 4.47 (0.79–25.4), 0.090 | 3.91 (0.66–23.1), 0.137 |
| 45–65 | 3 (100) | 0 (0.0) | 3 (100) | — | — |
| Sex | |||||
| Female | 289 (84.5) | 53 (15.5) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 20 (80.0) | 5 (20.0) | 25 (100) | 0.73 (0.26–2.06), 0.553 | 0.78 (0.26–2.33), 0.655 |
| Marital status | |||||
| Not married | 49 (75.4) | 16 (24.6) | 65 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Married | 260 (86.1) | 42 (13.9) | 302 (100) | 2.02 (1.05–3.89), 0.036 | 1.83 (0.91–3.67), 0.089 |
| Religion | |||||
| Christianity | 178 (86.8) | 27 (13.2) | 205 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Islam | 131 (80.9) | 31 (19.1) | 162 (100) | 0.64 (0.37–1.12), 0.119 | 0.71 (0.39–1.29), 0.262 |
| Ethnicity | |||||
| Akan/Twi | 25 (75.8) | 8 (24.2) | 33 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Kotokoli | 101 (83.5) | 20 (16.5) | 121 (100) | 1.62 (0.64–4.09), 0.307 | 1.49 (0.57–3.89), 0.418 |
| Ewe | 113 (88.3) | 15 (11.7) | 128 (100) | 2.41 (0.93–6.23), 0.069 | 2.12 (0.79–5.67), 0.135 |
| Hausa | 9 (69.2) | 4 (30.8) | 13 (100) | 0.72 (0.17–3.04), 0.655 | 0.77 (0.17–3.49), 0.735 |
| Basare | 21 (80.8) | 5 (19.2) | 26 (100) | 1.34 (0.37–4.83), 0.656 | 1.29 (0.35–4.75), 0.700 |
| Others | 40 (87.0) | 6 (13.0) | 46 (100) | 2.13 (0.63–7.17), 0.221 | 1.98 (0.57–6.86), 0.283 |
| Education level | |||||
| No education | 30 (63.8) | 17 (36.2) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 67 (81.7) | 15 (18.3) | 82 (100) | 2.54 (1.14–5.64), 0.022 | 2.31 (0.99–5.41), 0.053 |
| JHS/Middle | 134 (88.7) | 17 (11.3) | 151 (100) | 4.46 (2.09–9.52), <0.001 | 3.91 (1.75–8.72), 0.001 |
| SHS/Vocational | 34 (85.0) | 6 (15.0) | 40 (100) | 3.21 (1.11–9.26), 0.031 | 2.84 (0.94–8.61), 0.064 |
| Tertiary | 44 (93.6) | 3 (6.4) | 47 (100) | 8.31 (2.25–30.7), 0.002 | 6.98 (1.79–27.2), 0.005 |
| Employment status | |||||
| Not employed | 78 (78.8) | 21 (21.2) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 231 (86.2) | 37 (13.8) | 268 (100) | 1.68 (0.94–3.01), 0.079 | 1.52 (0.82–2.82), 0.184 |
| Sex of child | |||||
| Female | 135 (81.8) | 30 (18.2) | 165 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 174 (86.1) | 28 (13.9) | 202 (100) | 1.38 (0.79–2.40), 0.254 | 1.29 (0.72–2.31), 0.392 |
| Age of child (months) | |||||
| 5–11 | 60 (76.9) | 18 (23.1) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 | 90 (80.4) | 22 (19.6) | 112 (100) | 1.23 (0.63–2.40), 0.546 | 1.16 (0.58–2.32), 0.672 |
| 24–60 | 159 (89.8) | 18 (10.2) | 177 (100) | 2.65 (1.36–5.15), 0.004 | 2.31 (1.14–4.68), 0.020 |
Table 3: Factors Associated with Attitude towards Malaria Vaccine among Caregivers (n = 367)
| Characteristics | Willingness | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Willing (n = 344) | Not willing (n = 23) | ||||
| Awareness | |||||
| Poor | 88 (85.4) | 15 (14.6) | 103 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Good | 256 (97.0) | 8 (3.0) | 264 (100) | 5.45 (2.19–13.6), <0.001 | 3.21 (1.18–8.72), 0.023 |
| Attitude | |||||
| Negative | 47 (81.0) | 11 (19.0) | 58 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Positive | 297 (96.1) | 12 (3.9) | 309 (100) | 5.80 (2.55–13.2), <0.001 | 4.12 (1.71–9.92), 0.002 |
| Age group (years) | |||||
| 15–17 | 5 (83.3) | 1 (16.7) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 177 (93.7) | 12 (6.3) | 189 (100) | 2.95 (0.32–27.3), 0.342 | 2.61 (0.27–25.2), 0.403 |
| 28–45 | 160 (94.7) | 9 (5.3) | 169 (100) | 3.56 (0.38–33.4), 0.263 | 3.01 (0.31–29.5), 0.340 |
| 45–65 | 2 (66.7) | 1 (33.3) | 3 (100) | 0.40 (0.02–7.02), 0.530 | 0.36 (0.02–6.54), 0.493 |
| Sex | |||||
| Female | 321 (93.9) | 21 (6.1) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 23 (92.0) | 2 (8.0) | 25 (100) | 0.75 (0.16–3.48), 0.713 | 0.81 (0.17–3.89), 0.793 |
| Education level | |||||
| No education | 40 (85.1) | 7 (14.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 77 (93.9) | 5 (6.1) | 82 (100) | 2.69 (0.84–8.64), 0.095 | 2.11 (0.61–7.25), 0.238 |
| JHS/Middle | 144 (95.4) | 7 (4.6) | 151 (100) | 3.60 (1.18–10.9), 0.024 | 2.84 (0.87–9.23), 0.083 |
| SHS/Vocational | 38 (95.0) | 2 (5.0) | 40 (100) | 3.33 (0.68–16.3), 0.137 | 2.91 (0.55–15.4), 0.207 |
| Tertiary | 45 (95.7) | 2 (4.3) | 47 (100) | 3.94 (0.81–19.2), 0.090 | 3.12 (0.60–16.3), 0.175 |
| Employment status | |||||
| Not employed | 89 (89.9) | 10 (10.1) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 255 (95.1) | 13 (4.9) | 268 (100) | 2.21 (0.95–5.15), 0.065 | 1.87 (0.76–4.59), 0.173 |
| AEFI experienced | |||||
| No | 197 (95.6) | 9 (4.4) | 206 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Yes | 147 (91.3) | 14 (8.7) | 161 (100) | 0.48 (0.20–1.12), 0.090 | 0.39 (0.16–0.94), 0.036 |
Table 4: Factors Associated with Willingness to Accept Malaria Vaccine among Caregivers (n = 367)
| Characteristics | Vaccination Status | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Fully vaccinated n (%) | Not fully vaccinated n (%) | ||||
| Age of child (months) | |||||
| 5–11 (Dose 1) | 71 (91.0) | 7 (9.0) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 (Doses 1–3) | 78 (69.6) | 34 (30.4) | 112 (100) | 0.23 (0.10–0.52), <0.001 | 0.26 (0.11–0.60), 0.002 |
| 24–59 (Doses 1–4) | 89 (50.3) | 88 (49.7) | 177 (100) | 0.11 (0.05–0.24), <0.001 | 0.14 (0.06–0.31), <0.001 |
| Caregiver education | |||||
| No education | 24 (51.1) | 23 (48.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 56 (68.3) | 26 (31.7) | 82 (100) | 2.05 (1.01–4.17), 0.046 | 1.89 (0.90–3.96), 0.093 |
| JHS/Middle | 109 (72.2) | 42 (27.8) | 151 (100) | 2.47 (1.28–4.78), 0.007 | 2.21 (1.11–4.39), 0.024 |
| SHS/Vocational | 28 (70.0) | 12 (30.0) | 40 (100) | 2.26 (0.95–5.35), 0.066 | 1.98 (0.80–4.93), 0.141 |
| Tertiary | 21 (44.7) | 26 (55.3) | 47 (100) | 0.76 (0.33–1.76), 0.524 | 0.81 (0.34–1.96), 0.642 |
| Caregiver attitude | |||||
| Negative | 24 (41.4) | 34 (58.6) | 58 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Positive | 214 (69.3) | 95 (30.7) | 309 (100) | 3.17 (1.78–5.66), <0.001 | 2.74 (1.47–5.12), 0.001 |
| AEFI experienced | |||||
| No | 156 (75.7) | 50 (24.3) | 206 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Yes | 82 (50.9) | 79 (49.1) | 161 (100) | 0.34 (0.22–0.54), <0.001 | 0.41 (0.25–0.67), <0.001 |
| Employment status | |||||
| Not employed | 58 (58.6) | 41 (41.4) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 180 (67.2) | 88 (32.8) | 268 (100) | 1.44 (0.89–2.33), 0.136 | 1.29 (0.77–2.18), 0.337 |
| Overall | 238 (64.9) | 129 (35.1) | 367 (100) | — | — |
Table 5: Age-Appropriate Full Malaria Vaccination Status and Associated Factors among Children Aged 6–59 Months (n = 367)
| AEFI Characteristic | Frequency (n) | Percentage (%) |
|---|---|---|
| Experienced any AEFI | 161 | 43.9 |
| No AEFI reported | 206 | 56.1 |
| Type of AEFI reported | ||
| Fever | 147 | 91.3 |
| Swelling at the injection site | 9 | 5.6 |
| Vomiting | 3 | 1.9 |
| Others | 2 | 1.2 |
| AEFI severity | ||
| Non-serious | 145 | 90.1 |
| Serious | 16 | 9.9 |
| AEFI outcome | ||
| Recovered | 161 | 100 |
| Did AEFI affect the acceptance of other vaccines? | ||
| No | 125 | 77.6 |
| Yes | 36 | 22.4 |
Table 6: Adverse Events Following Immunization (AEFI) after RTS, S Malaria Vaccination (n = 367)
Figures


Keywords
- Caregivers
- Malaria vaccine
- RTS, S/AS01
- Oti Region
- Ghana
John Sonnyinado Duako Baffoe1,2,3,4,&, Joseph Abdul-Latif4,5, Samuel Agyiri4,6, Vida Dabaloro4,7, Elijah Wamale Nlamba4,8, Gregory Amenuvegbe4, Joy Sefakor Mawusi4, Eric Takyi1, Rita Agyekumwah Asante2, George Akowuah2, Wisdom Kwame Takramah4, Joyce Berkumwin Der4, Donne Kofi Ameme2, Austin Gideon Adobasom-Anane9, George Asumah-Karikari Adu10
1District Health Directorate, Kadjebi, Oti Region, Ghana, 2Ghana Field Epidemiology and Laboratory Training Programme, School of Public Health, University of Ghana, 3Reaching The Unreached International (NGO), Ghana, 4Fred N. Binka School of Public Health, Hohoe, Ghana, 5District Health Directorate, Kassena Nankana West, Upper East Region, Ghana, 6Municipal Health Directorate, Birim Central, Eastern Region, Ghana, 7District Health Directorate, Techiman North, Bono East Region, Ghana, 8Municipal Health Directorate, Kassena Nankana, Upper East Region, Ghana, 9Kumasi Centre for Collaborative Research in Tropical Medicine, Kumasi, Ghana, 10National Malaria Elimination Programme, Accra, Ghana
&Corresponding author: John Sonnyinado Duako Baffoe, 1District Health Directorate, Kadjebi, Oti Region, Ghana, Email: baffoejohn22@gmail.com ORCID: https://orcid.org/0000-0002-4021-4172
Received: 27 Sep 2025, Accepted: 12 Apr 2026, Published: 17 Apr 2026
Domain: Infectious Disease Epidemiology
Keywords: Caregivers, malaria vaccine, RTS, S/AS01, Oti Region, Ghana
©John Sonnyinado Duako Baffoe et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: John Sonnyinado Duako Baffoe et al. Caregivers’ awareness, attitudes, uptake, and willingness toward the RTS,S/AS01 malaria vaccine, Oti Region, Ghana 2023. Journal of Interventional Epidemiology and Public Health. 2026; 9(2):64. https://doi.org/10.37432/jieph-d-25-00209
Abstract
Introduction: Malaria remains a major public health challenge, particularly among children under five years of age. The World Health Organization’s approval of the RTS, S/AS01 malaria vaccine for pilot implementation in Ghana, Kenya, and Malawi on May 1, 2019, marked a significant milestone in malaria prevention. This study assessed caregivers’ awareness, attitudes, vaccine uptake, adverse events following immunization (AEFI), and willingness to accept the RTS, S vaccine in the Oti Region of Ghana.
Methods: A community-based cross-sectional study was conducted in 2023 across five sub-districts. Using a multistage sampling technique, 39 communities and 367 caregivers of children aged 6–59 months were selected. Data were collected using a semi-structured questionnaire administered via Google Forms (Google LLC, Mountain View, CA, USA), and RTS, S vaccination status was verified using Child Welfare Clinic cards. Descriptive statistics and multivariable logistic regression analyses were performed, with adjusted odds ratios (AORs) and 95% confidence intervals (CIs) reported.
Results: Of the 367 caregivers, 342 (93.2%) were female, with a median age of 25 years (IQR: 9). Awareness of malaria vaccination was high (323/367; 87.5%), and 309/367 (84.2%; 95% CI: 80.2–87.6) demonstrated a positive attitude toward the vaccine. RTS, S uptake declined progressively from 96.5% for dose 1 to 50.4% for dose 4. Age-appropriate full vaccination was achieved by 238/367 (64.9%) children. Positive caregiver attitude (AOR = 2.74; 95% CI: 1.47–5.12) and higher educational attainment were independently associated with full vaccination, whereas older child age and prior AEFI (aOR=0.41 95%CI:0.25–0.67) were associated with lower completion. Overall, 161/367 (43.9%) caregivers reported AEFI, the majority of which were non-serious.
Conclusion: Although awareness, positive attitudes, and willingness to accept the RTS, S vaccine were high, completion of later doses remained suboptimal. Strengthening caregiver engagement, addressing concerns related to AEFI, reinforcing Evidence for Action (E4A) strategies, and enhancing follow-up systems are essential to improve full vaccination coverage and maximise the public health impact of the malaria vaccine.
Introduction
Malaria remains one of the most devastating public health challenges globally, with Plasmodium falciparum (Pf) as the primary cause of morbidity and mortality in sub-Saharan Africa [1,2]. From centuries of human struggle and numerous control interventions, malaria continues to claim over 400,000 lives annually, most of them children under five [3–5]. In response to this persistent threat, the RTS, S/AS01 malaria vaccine, commercially known as Mosquirix™ was developed through a decades-long collaboration between GlaxoSmithKline (GSK), PATH, and the Walter Reed Army Institute of Research, with funding support from the Bill & Melinda Gates Foundation [6,7].
The vaccine combines components of the Plamodium falciparum circumsporozoite protein and hepatitis B surface antigen, forming a fusion protein that targets the parasite’s early stages [8]. Following promising results from early trials, the WHO recommended pilot implementation of RTS, S in Ghana, Kenya, and Malawi in 2019. The pilot demonstrated a meaningful reduction in severe malaria cases and hospitalizations among vaccinated children [2]. Age-appropriate full malaria vaccination was defined according to the child’s age at assessment and eligibility for RTS, S doses, as verified from Child Welfare Clinic cards: children aged 5–11 months were considered fully vaccinated if they had received RTS, S dose 1; those aged 12–23 months if they had received RTS, S doses 1–3; and those aged 24–59 months if they had received all four RTS, S doses, while children who had not received all age-eligible doses were classified as not fully vaccinated [9,10]. These results affirmed RTS, S as a viable tool in the fight against malaria, especially in high-transmission areas. However, despite its promise, vaccine uptake has varied across regions.
In Ghana, challenges such as cold chain logistics, financial constraints, and caregiver hesitancy have hindered optimal coverage. In the Kadjebi District of the Oti Region, one of the pilot districts, coverage rates for RTS, S doses have consistently fallen short of the national 90% target. For instance, in 2021, RTS, S1 coverage reached 79.4%, while RTS, S4 lagged at 57% from the Kadjebi District Health Directorate 2021 Annual Performance Review. We assessed caregivers’ knowledge, attitudes, uptake, adverse effects following immunization (AEFI), and willingness to accept the malaria vaccine in Kadjebi District. Understanding these factors is crucial to improving vaccine implementation and achieving Sustainable Development Goal 3.3: to end the malaria epidemic by 2030 [11]. By exploring community-level perceptions and experiences, this research seeks to provide actionable insights for policymakers and health stakeholders. The findings will help strengthen malaria vaccine strategies and ensure that children in endemic regions receive the protection they need.
Methods
Study design
This study employed a community-based quantitative cross-sectional design to assess caregivers’ awareness, attitudes, willingness to accept malaria vaccination, vaccine uptake, and adverse events following immunization (AEFI) in the Kadjebi District [12,13].
Study site description
The study was conducted in the Kadjebi District, located in the middle sector of the Oti Region of Ghana. The district was carved out of the Jasikan-Buem Local Council and established as an Assembly by Legislative Instrument (L.I.) 1465 in 1989. Kadjebi is one of nine Municipal and District Assemblies in the Oti Region, encompassing a total land area of approximately 690 square kilometres [14]. It is bordered to the north by Nkwanta South District, to the south by Jasikan Municipal, to the southwest by Biakoye District, and to the northwest by Krachi East Municipal, with a long eastern border adjoining the Republic of Togo (Figure 1).
For primary health care planning and implementation, the district is divided into five sub-districts: Kadjebi-Asato, Dodo, Dodi, Ahamansu, and Pampawie (Kadjebi District Assembly, 2010). The district comprises 22 health facilities, including one hospital managed by the Christian Health Association of Ghana (CHAG), five health centres, and 36 Community-based Health Planning and Services (CHPS) zones, with 14 CHPS compounds. According to the 2021 Ghana Population and Housing Census, the projected population of the district for 2023 is 77,098, of which 51.2% (39,474) are males. Children under five constitute approximately 20% (15,425), and children aged under one year 4% (3,083).
Malaria remains highly endemic in the district, exacerbated by high rainfall levels and suboptimal usage of long-lasting insecticide-treated nets (LLINs). The region’s climatic and geographical features contribute to persistent malaria transmission, emphasizing the importance of targeted malaria interventions, including vaccination. Ghana’s national malaria vaccine protocol follows the introduction of the RTS, S/AS01 (Mosquirix) vaccine into the routine Expanded Programme on Immunization (EPI) for children living in moderate-to-high malaria transmission settings. The vaccine is administered in a four-dose schedule at 6, 7, 9, and 18 months of age, in accordance with recommendations from the World Health Organization (WHO. Ghana began piloting the vaccine in 2019 under the Malaria Vaccine Implementation Programme. It expanded nationwide in 2024 to all eligible districts, including those in the Oti Region, with support from UNICEF Ghana, Gavi, and other partners [15]. While early uptake of the first three doses has generally been high due to integration with routine immunization services, administrative data from the RTS, S pilot implementation in Ghana indicate that coverage for the first three doses exceeded 85–90%, whereas completion of the fourth dose declined to approximately 50–60%. This drop in coverage has been attributed to caregiver knowledge gaps, access challenges, and missed opportunities within health facilities.[16]. In the Kadjebi, where malaria transmission remains high, anecdotal programmatic reports suggest similar patterns, with relatively good initial acceptance but declining follow-up for later doses, underscoring the need for strengthened community engagement, defaulter tracing, and health education to maximize the vaccine’s protective impact against severe malaria [17,18].
Study population, inclusion, and exclusion criteria
The study population comprised caregivers residing in the Kadjebi District who had at least one child aged 6–59 months. This age group was selected because it corresponds to the target population for the malaria vaccination schedule, thereby enabling an appropriate assessment of caregivers’ knowledge, attitudes, uptake, AEFI, and willingness to receive the malaria vaccine. Caregivers were excluded if their eligible child was seriously ill at the time of data collection or if the child was unavailable for assessment or caregiver declined consent during the study period.
Sample size and sampling technique
To determine the minimum sample size for the study, the Cochran formula 1972 was used as illustrated below.
\[
n = \frac{Z^2 P (1 – P)}{d^2} \times \left(1 + p(m – 1)\right)
\]
Where n = desired sample size, Z = statistic corresponding to the level of confidence (1.96), P = expected coverage 75% RTS, S3 coverage (was a proxy for EPI), based on the MVIP Quarterly Data Bulletin in Ghana 2021, and d = desired level of precision (0.05 margin of error). A 10% adjustment for the non-response rate, p = Intra-cluster correlation (0.005), m = number of individuals in each cluster (9,252), and Design effect (Df) (47), when computed into the formula, yields 367 respondents.
In determining the proportionate sample size for each sub-district (strata), the formula A= y/z×n was used where “A” is the sample size for each stratum (sub-district), ‘y’ the sub-district yearly target population for children under one years in 2023, and ‘z’ the Kadjebi District 2023 target population (3,083) and “n” the total sample size for the study (367) [19,20]. The sample comprised 53 children from Ahamansu (population: 449), 105 from Dodi (population: 884), 73 from Dodo (population: 612), 102 from Kadjebi-Asato (population: 853), and 34 from Pampawie (population: 285) Sub-districts.
A stratified multistage cluster sampling technique was employed to obtain a representative sample of caregiver–child pairs in Kadjebi District. First, the district, which is administratively divided into five sub-districts, was stratified to ensure geographic representativeness. Accordingly, the calculated sample size was proportionally allocated to each sub-district based on the population of children aged 6–59 months, such that sub-districts with larger eligible populations contributed a greater number of respondents. Importantly, the sub-districts themselves were not sampled but served only as strata for sample distribution.
Subsequently, the primary sampling frame consisting of 174 communities was considered. Based on the allocated sample size within each stratum, communities were selected proportionately from each sub-district. In total, thirty-nine communities were selected using simple random sampling generated by Microsoft Excel 365, thereby ensuring that each community had an equal probability of selection within its respective stratum.
Following community selection, households within each selected community were identified with the assistance of community health volunteers and local guides. The number of households interviewed in each community was determined by dividing the allocated sub-district sample size by the number of selected communities in that sub-district. Thereafter, a random starting household was identified at the centre of the community, after which systematic household sampling was applied by selecting households at a fixed interval until the required number of eligible households was obtained. Only households with at least one child aged 6–59 months were considered eligible for participation.
Where more than one eligible child was present in a household, a simple random selection using concealed ballot allocation was conducted to select one child–caregiver pair, thereby ensuring equal probability of inclusion and preventing intra-household clustering. The caregiver of the selected child was then interviewed through face-to-face administration of a structured questionnaire. Thus, the sampling unit was the community (cluster), while the ultimate study unit was the child aged 6–59 months represented by the caregiver respondent.
Regarding data verification, vaccination status was primarily confirmed using Child Welfare Clinic (CWC) cards. However, where cards were unavailable, caregivers were asked to retrieve them on the same day, if feasible. If this was not possible, vaccination history was obtained through caregiver recall and cross-checked with health facility immunization registers, where available. Nevertheless, for variables requiring objective verification, including RTS, S malaria vaccine dose receipt and vaccination dates, only information confirmed by CWC cards or facility records was included in the final analysis. Consequently, records lacking verification were coded as missing and excluded from analyses requiring documented vaccination evidence using listwise deletion [20,21].
Outcome and independent variables
The outcome factors that can be influenced by the independent variables were evaluated, including attitude level, uptake, Adverse Event Following Immunization (AEFI), and whether caregivers were willing to receive the malaria vaccines. Due to their ability to be affected by and controlled by other variables of measurement, these variables were chosen as dependents. Because the measure is unaffected, the independent factors were the respondents’ age, sex, occupation, marital status, education level, religion, ethnicity, and age of child [22,23].
Data collection and tool measurement
Data were collected from caregivers of children aged 6–59 months residing in the Kadjebi District using a semi-structured questionnaire administered electronically via Google Forms (Google LLC, Mountain View, CA, USA), a web-based software platform. In addition to caregiver interviews, Child Welfare Clinic (CWC) cards were reviewed to verify malaria vaccine uptake, thereby minimizing recall bias objectively.
The questionnaire was designed and structured strictly in accordance with the study objectives and the manuscript scope, which focused on caregivers’ knowledge, attitudes, uptake, adverse events following immunization (AEFI), and willingness to accept the malaria vaccine. The data collection instrument comprised six sections, each aligned with a specific outcome of interest:
Section I: Socio-demographic characteristics
This section captured background variables that can influence malaria vaccine-related outcomes, including caregiver age, sex, educational level, marital status, religion, employment status, ethnicity, child’s sex, and child’s age (in months).
Section II: Caregivers’ awareness of the malaria vaccine
Awareness was assessed by whether the caregivers had heard of the malaria vaccine and specifically the RTS, S vaccine, and the sources of information.
Section III: Caregivers’ attitudes toward the malaria vaccine
Attitude was assessed as caregivers’ beliefs and evaluative judgments regarding the malaria vaccine. Items examined perceptions of vaccine effectiveness in preventing severe and life-threatening malaria, beliefs about duration of protection, and social perceptions related to vaccination.
Section IV: Malaria vaccine uptake
Vaccine uptake was measured using four items corresponding to receipt of RTS, S doses 1, 2, 3, and 4. Uptake information was verified through review of CWC cards to ensure accuracy.
Section V: Adverse Events Following Immunization (AEFI)
This section assessed caregiver-reported AEFI following malaria vaccination, including occurrence, type of reaction, severity (serious or non-serious), outcome, and whether the event influenced acceptance of subsequent vaccinations.
Section VI: Willingness to accept the malaria vaccine
Willingness was measured through items assessing caregivers’ readiness to vaccinate their children with the malaria vaccine, the influence of previous childhood vaccination experiences on acceptance, and willingness to pay for the vaccine if required.
The questionnaire was adapted from previously validated malaria vaccine studies and modified to suit the local context and study objectives. A pretest was conducted among 37 caregivers in the Jasikan Municipality, after which necessary revisions were made to improve clarity, relevance, and flow of questions. One supervisor and six trained data collectors administered the electronic questionnaire following standardized procedures to ensure data quality and consistency [1,21–23]
Data management and analysis
Data collected was exported into Microsoft Excel 2016 for initial cleaning and validation. Data cleaning involved verifying the completeness, consistency, and accuracy of responses, including confirmation of malaria vaccine uptake from Child Welfare Clinic (CWC) cards. The cleaned dataset was then imported into Stata version 15 (StataCorp, College Station, TX, USA) for statistical analysis.
Descriptive statistics were computed to summarize respondents’ socio-demographic characteristics and key study variables. Categorical variables were summarized using frequencies and percentages, while results were presented in tables and figures as appropriate. Five primary outcome variables were analyzed: awareness of the malaria vaccine, attitude towards the malaria vaccine, malaria vaccine uptake, adverse events following immunization (AEFI), and willingness to accept the malaria vaccine. The reliability of the survey tool was tested in a pilot study with 37 mothers, yielding a Cronbach’s alpha coefficient indicating high internal consistency and validity.
Attitude was measured using three Likert-scale items and categorized as a positive attitude for scores of 66.7% or higher, and a negative attitude otherwise [24,25]. Vaccine uptake was assessed based on receipt of RTS, S doses verified from CWC cards. Adverse Events Following Immunization (AEFI) were categorized as non-serious or serious according to the National Expanded Programme on Immunization (EPI) policy guidelines, and willingness was defined as caregivers’ readiness to present their ward for the malaria vaccine. In addition, age-appropriate full malaria vaccination status was derived as a composite outcome. Children aged 5–11 months were considered fully vaccinated if they had received RTS, S dose 1; those aged 12–23 months if they had received RTS, S doses 1–3; and those aged 24–59 months if they had received all four RTS, S doses.
Bivariate logistic regression analyses were first conducted to examine associations between each outcome variable and independent variables, including caregiver age, sex, marital status, education level, religion, ethnicity, employment status, child sex, and child age. Crude Odds Ratios (CORs) with 95% Confidence Intervals (CIs) were estimated. Variables with a p-value < 0.20 at the bivariate level were entered into multivariable logistic regression models to identify independent predictors while controlling for potential confounders.
Multivariable logistic regression analyses were performed separately for each outcome variable, and Adjusted Odds Ratios (AORs) with corresponding 95% confidence intervals were reported. Statistical significance was set at a p-value < 0.05. Model results were presented in tables, while uptake trends and predictors of full malaria vaccination were additionally illustrated using figures where appropriate [26,27]. results presented in tables, figures, and narrative text [22,28].
Ethical approval
The study obtained ethical approval from the University of Health and Allied Sciences Review Ethics Committee (UHAS-REC), with reference number UHAS-REC B.10 22-23, dated May 22, 2023. Additional permission was granted from the District Health Management Team. Entry into the community was facilitated through engagement with local opinion leaders and communication with health facility managers and participants about the study aims. Written informed consent was secured from all caregiver participants after a thorough explanation of the study’s purpose, objectives, methods, possible risks, and benefits. Participation was entirely voluntary.
Results
Demographic characteristics
A total of 367 caregivers participated in the study. The median age of caregivers was 25 years (IQR: 9), with ages ranging from 15 to 65 years, while the median age of children was 24 months (IQR: 18), ranging from 5 to 59 months. Most caregivers were aged 17–28 years, 189/367 (51.5%), or 28–45 years, 169/367 (46.0%). Most caregivers were female 342/367, 93.2%), and most were married (302/367; 82.3%). With respect to educational attainment, 151/367 (41.1%) had completed JHS/Middle education, 82/367 (22.3%) had primary education, 47/367 (12.8%) had tertiary education, while 47/367 (12.8%) reported no formal education. Most respondents were employed (268/367; 73.0%). Christianity was the predominant religion 205/367 (55.9%), followed by Islam 162/367 (44.1%). Ethnically, caregivers were mainly Ewe 128/367 (34.9%) and Kotokoli 121/367 (33.0%), with smaller proportions being Others (46/367; 12.5%), Akan/Twi (33/367; 9.0%), Basare 26/367 (7.1%), and Hausa 13/367 (3.5%). Among the children assessed, 202/367 (55.0%) were male, and 165/367 (45.0%) were female. Nearly half of the children were aged 24–60 months, 177/367 (48.2%), while 112/367 (30.5%) were aged 12-23 months, and 78/367 (21.3%) were aged 5–11 months. [Table 1].
Factors associated with awareness of malaria vaccine among caregivers
Overall, 264/367 (71.9%) of caregivers demonstrated good awareness of the malaria vaccine. In multivariable logistic regression analysis, caregiver education level was independently associated with awareness. Compared with caregivers with no formal education, those with primary education (AOR = 3.96; 95% CI: 1.74–9.01), JHS/Middle education (AOR = 2.41; 95% CI: 1.18–4.92), and SHS/Vocational education (AOR = 2.62; 95% CI: 1.01–6.80) had significantly higher odds of good awareness. In addition, caregivers belonging to other ethnic groups were more likely to have good awareness compared with Akan/Twi caregivers (AOR = 3.91; 95% CI: 1.18–12.9). No other variables were significantly associated with awareness after adjustment. [Table 2].
Caregivers’ attitude toward the malaria vaccine
Attitudes toward malaria vaccination were generally favourable. Most caregivers agreed or strongly agreed that the malaria vaccine can reduce serious and life-threatening malaria episodes in children under five years (309/367; 84.2%). However, perceptions regarding the broader impact of the vaccine were mixed. While 201/367 (54.5%) correctly indicated that malaria vaccination alone cannot eliminate malaria in Ghana, 166/367 (45.5%) believed that vaccination could eliminate malaria. In addition, 246/367 (66.7%) agreed or strongly agreed that the malaria vaccine provides lifelong protection, suggesting optimistic but inaccurate expectations regarding duration of protection. Encouragingly, 295/367 (80.4%) disagreed or strongly disagreed with the statement that individuals who are not immunized with the malaria vaccine should be socially avoided, indicating low levels of stigmatizing attitudes.
Based on the composite attitude score, 309/367 (84.2%) of caregivers were categorized as having a positive attitude toward malaria vaccination. In multivariable logistic regression analysis, caregiver awareness was a strong independent predictor of attitude. Caregivers with good awareness were significantly more likely to have a positive attitude compared with those with poor awareness (AOR = 3.98; 95% CI: 2.05–7.71). Educational attainment was also independently associated with attitude; caregivers with JHS/Middle education (AOR = 3.91; 95% CI: 1.75–8.72) and tertiary education (AOR = 6.98; 95% CI: 1.79–27.20) had higher odds of positive attitude compared with those with no formal education. Additionally, caregivers of children aged 24–60 months were more likely to demonstrate a positive attitude than those with children aged 5–11 months (AOR = 2.31; 95% CI: 1.14–4.68). [Table 3].
Caregivers’ willingness to accept the malaria vaccine
Willingness to accept malaria vaccination was high among caregivers. Overall, 344/367 (93.7%) reported willingness to vaccinate their children, while 23/367 (6.3%) were not willing. In multivariable logistic regression analysis, caregiver awareness and attitude were independently associated with willingness. Caregivers with good awareness were more likely to be willing to accept the malaria vaccine compared with those with poor awareness (AOR = 3.21; 95% CI: 1.18–8.72). Similarly, caregivers with a positive attitude had higher odds of willingness compared with those with a negative attitude (AOR = 4.12; 95% CI: 1.71–9.92). In contrast, caregivers who reported that their child experienced an adverse event following immunization were significantly less likely to be willing to accept the malaria vaccine (AOR = 0.39; 95% CI: 0.16–0.94). No other socio-demographic or child-related variables remained statistically significant after adjustment. [Table 4].
Malaria vaccine uptake
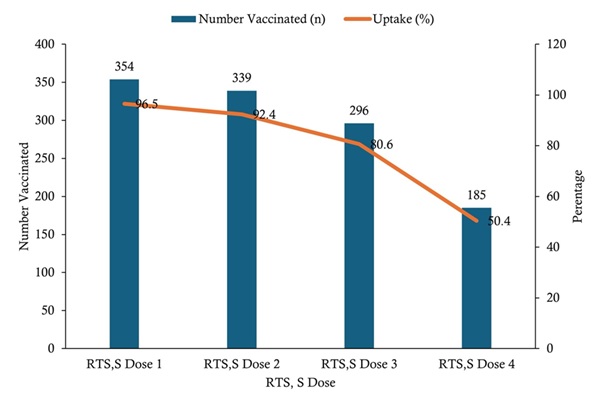
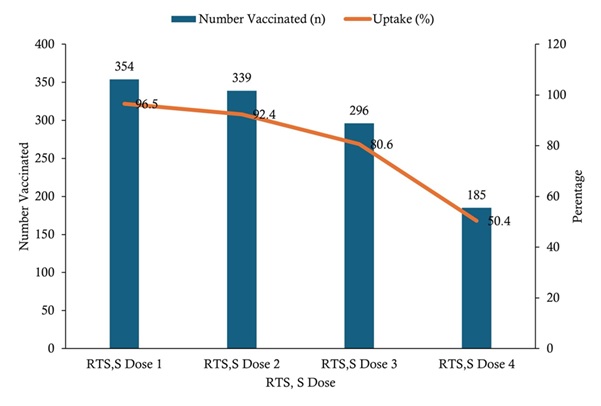
Uptake of RTS, S malaria vaccine doses among children aged 6–59 months was high for the initial doses but declined with subsequent doses. Coverage was highest for RTS, S dose 1 (354/367; 96.5%), followed by dose 2 (339/367; 92.4%) and dose 3 (296/367; 80.6%), while the lowest uptake was observed for dose 4 (185/367; 50.4%) (Figure 2).
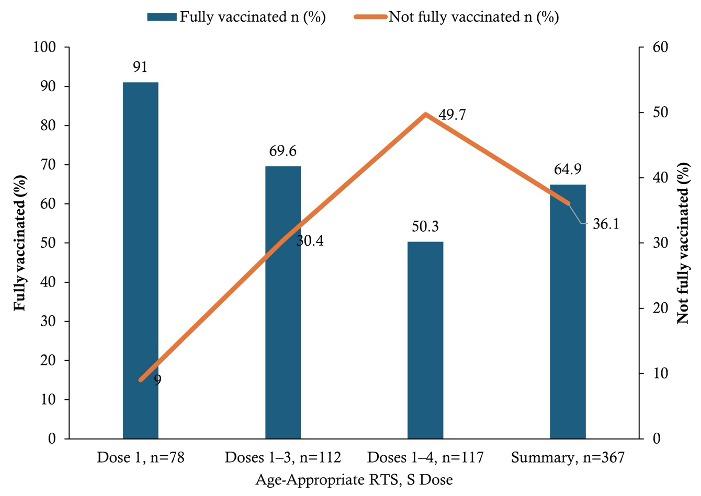
Based on age-specific eligibility, 238/367 (64.9%) of children were fully vaccinated for their age, while 129/367 (35.1%) were not fully vaccinated (Figure 3). In multivariable logistic regression analysis, child age, caregiver attitude, caregiver education, and experience of adverse events following immunization (AEFI) were independently associated with age-appropriate full malaria vaccination. Compared with children aged 5–11 months, those aged 12–23 months (AOR = 0.26; 95% CI: 0.11–0.60) and 24–59 months (AOR = 0.14; 95% CI: 0.06–0.31) had significantly lower odds of being fully vaccinated. Caregivers with a positive attitude toward malaria vaccination were more likely to have fully vaccinated children compared with those with a negative attitude (AOR = 2.74; 95% CI: 1.47–5.12). Similarly, caregivers with JHS/Middle education had higher odds of full vaccination compared with those with no formal education (AOR = 2.21; 95% CI: 1.11–4.39). In contrast, children who experienced AEFI had lower odds of full vaccination compared with those who did not (AOR = 0.41; 95% CI: 0.25–0.67). [Table 5].
Adverse event following immunization (AEFI) of malaria vaccine implementation
One hundred and sixty-one of 367 (43.9%) caregivers reported that their child experienced at least one adverse event following RTS, S malaria vaccination. Among caregivers who reported an adverse event following immunization, fever was the most reported event, accounting for 147/161 (91.3%) of all reported adverse events. Other reported events included swelling at the injection site 9/161 (5.6%), vomiting 3/161 (1.9%), and other symptoms 2/161 (1.2%). Most reported adverse events were non-serious, with 145/161 (90.1%). All children who experienced an adverse event had fully recovered 161/161 (100%). Regarding the effect of adverse events on subsequent vaccination behaviour, 125/161 (77.6%) of caregivers indicated that the adverse event did not affect acceptance of other vaccines, whereas 36/161 (22.4%) reported that the adverse event influenced their acceptance of subsequent vaccinations [Table 6].
Discussion
One of the numerous malaria elimination initiatives being implemented in Ghana is the novel vaccine known as RTS, S/AS01, which was piloted in April 2017 in Ghana, Kenya, and Malawi. This study provides implementation-phase evidence on caregivers’ awareness, attitudes, willingness, uptake, and adverse events following immunization (AEFI) related to the RTS, S malaria vaccine in the Kadjebi District of Ghana’s Oti Region. By integrating caregiver perceptions with verified vaccination records, the findings offer insight into both behavioural and health-system factors influencing malaria vaccine performance in a real-world setting.
Caregivers’ awareness of the malaria vaccine
Caregivers in this study demonstrated high awareness of malaria vaccination, with the majority reporting that they had heard of a malaria vaccine and a substantial proportion specifically aware of the RTS, S vaccine. This level of awareness suggests that community sensitization and routine immunization messaging within the district have achieved broad population reach. Similar patterns have been reported in both pre-introduction and post-implementation studies of malaria vaccines, where high levels of awareness were observed following targeted communication campaigns [28][29]. Despite this high awareness, important misconceptions were evident, particularly regarding the belief that malaria vaccination alone could eliminate malaria and that the vaccine provides lifelong protection. These findings indicate that while communication efforts have successfully promoted exposure to information about the vaccine, they may have been less effective in clarifying the vaccine’s partial efficacy and its role as a complementary intervention alongside other malaria control strategies, such as Long-Lasting Insecticide-Treated Nets (LLINs) and seasonal malaria chemoprevention. Given that healthcare providers were the predominant source of information, strengthening provider-led counselling and emphasizing clear, consistent messaging about the scope and limitations of RTS, S may further improve the quality of caregiver understanding. Tailored health education strategies, particularly for caregivers with lower levels of formal education and those in rural settings, remain essential to ensure that awareness translates into informed decision-making and sustained adherence to the vaccination schedule.
Attitudes toward the malaria vaccine
Attitudes toward malaria vaccination were predominantly positive, reflecting widespread recognition of malaria as a serious and potentially fatal disease within the study population. This finding is consistent with earlier acceptability studies conducted before the rollout of RTS, S, as well as more recent post-introduction evaluations in sub-Saharan Africa, which similarly reported favourable caregiver perceptions of the malaria vaccine [30,31]. In this study, positive attitudes were significantly associated with higher educational attainment, suggesting that formal education may enhance confidence in vaccine effectiveness and trust in health interventions. This aligns with behavioural frameworks that emphasize the role of perceived benefits, trust in health systems, and confidence in vaccine safety as key drivers of favourable vaccine attitudes [32]. However, positive attitudes coexisted with certain misconceptions, particularly regarding lifelong protection and the vaccine’s ability to eliminate malaria. This indicates that favourable perceptions do not necessarily equate to a comprehensive understanding of the vaccine’s limitations. Such optimism, if not appropriately addressed, may affect long-term adherence to vaccination schedules and the continued use of complementary malaria prevention strategies such as LLINs and seasonal malaria chemoprevention.
Willingness to accept RTS, S and vaccine confidence
Willingness to vaccinate children with RTS, S was notably high in this study, consistent with findings from other settings across sub-Saharan Africa that report strong caregiver readiness to adopt the malaria vaccine following its introduction [29,1]. This high level of willingness, despite the presence of certain misconceptions, supports existing evidence that trust in health systems and the perceived severity of malaria often play a more decisive role in vaccine acceptance than detailed biomedical understanding alone [33][34]. However, the observed reduction in willingness among caregivers who reported previous adverse events following immunization (AEFI) underscores the fragility of vaccine confidence when shaped by personal experience. Although RTS, S associated AEFIs are predominantly mild and transient, as demonstrated in long-term clinical trials and post-marketing evaluations [10,29], even non-serious adverse events may negatively influence subsequent vaccination decisions if caregivers are not adequately prepared for or counselled about expected reactions. These findings highlight the critical importance of anticipatory guidance, transparent risk communication, and timely reassurance by healthcare providers. Strengthening communication around the normalcy and transient nature of common post-vaccination symptoms may help sustain vaccine confidence and prevent avoidable drop-out across subsequent doses.
Vaccine uptake, completion, and programmatic challenges
High uptake of the initial RTS, S doses reflects strong early acceptance and adequate access to immunization services within the district. However, the progressive decline in coverage for subsequent doses mirrors a well-documented challenge in multi-dose vaccine delivery, observed both for RTS, S and other routine childhood vaccines [31,35]. This pattern underscores the difficulty of sustaining engagement beyond early infancy within routine immunization systems. Reduced contact with child welfare services as children grow older may partly explain the lower completion rates observed. Importantly, positive caregiver attitudes and higher educational attainment were independently associated with improved age-appropriate full vaccination, highlighting the interplay between psychosocial determinants and service utilization. These findings align with accumulating evidence suggesting that drop-out across later RTS, S doses are more frequently attributable to health-system constraints such as inadequate follow-up, weak reminder mechanisms, and limited defaulter tracing rather than active caregiver refusal [18,36].
Adverse events following immunization
Most reported adverse events following immunization (AEFI) were non-serious, with fever being the most frequently observed reaction, and all affected children reportedly recovered fully. This finding is consistent with the well-established safety profile of RTS, S demonstrated in both long-term clinical trials and post-introduction surveillance studies [10,29]. However, evidence from behavioural and vaccine confidence research suggests that even mild and transient adverse events may influence caregiver perceptions and subsequent vaccination decisions, particularly when risk communication is insufficient or poorly framed [37]. In contexts where personal experience strongly shapes health behaviour, unmanaged expectations regarding post-vaccination symptoms can undermine confidence in otherwise safe and effective interventions. These findings underscore the importance of proactive AEFI counselling, clear anticipatory guidance regarding expected side effects, and responsive feedback systems within routine immunization services. Strengthening these components is critical to maintaining public trust and preventing avoidable drop-out from subsequent vaccine doses.
Implications for policy and practice
Despite the limitations of this study, the findings offer important programmatic and policy-relevant insights. The high levels of awareness, positive attitudes, and willingness observed among caregivers provide a favourable foundation for sustained malaria vaccine scale-up within the district. However, the marked decline in completion of later RTS, S doses indicates that maintaining coverage beyond initial uptake requires targeted system-level interventions.
Strengthening reminder systems, structured defaulter tracing mechanisms, and integration of malaria vaccine follow-up into routine child welfare services are critical to improving age-appropriate full vaccination. In addition, reinforcing provider-led communication that clearly explains the vaccine’s partial efficacy, the necessity of completing all recommended doses, and the expected nature of mild adverse events will be essential to preventing misconceptions from undermining long-term program performance. From a broader policy perspective, aligning malaria vaccine messaging with existing malaria control strategies, such as LLIN use and seasonal malaria chemoprevention, may further reinforce the understanding that RTS, S functions as a complementary intervention rather than a standalone solution. Investment in continuous community engagement, supportive supervision, and health worker capacity building will therefore be central to sustaining vaccine confidence and optimizing public health impact.
Limitations
Several limitations should be considered when interpreting the findings of this study. First, the cross-sectional design precludes causal inference between caregiver characteristics and malaria vaccine-related outcomes, as exposures and outcomes were assessed at a single point in time.
In addition, information on awareness, attitudes, willingness, and adverse events following immunization (AEFI) was self-reported and may therefore be subject to recall bias and social desirability bias, particularly within a context where vaccination is strongly promoted.
Although vaccination status was verified using Child Welfare Clinic (CWC) cards, incomplete, missing, or inconsistently documented records may have introduced some degree of misclassification, particularly regarding dose timing and completion status.
Furthermore, the study was conducted in selected sub-districts within the Oti Region, which may limit the generalizability of the findings to other districts or regions with differing sociodemographic or health system characteristics.
Finally, the analysis did not incorporate district-level implementation variables such as timing of vaccine rollout, intensity of community sensitization, health worker training, or facility-level service delivery differences—which may have influenced caregiver perceptions and vaccine uptake patterns. Future research incorporating health system performance indicators and longitudinal designs would provide a more comprehensive understanding of determinants of malaria vaccine completion.
Conclusion
This study demonstrates that caregivers in the Kadjebi District exhibit high awareness, predominantly positive attitudes, and strong willingness to accept the RTS, S malaria vaccine. Despite this favourable context, completion of later vaccine doses remains suboptimal, resulting in moderate age-appropriate full vaccination coverage.
Caregiver awareness and educational attainment were key determinants of positive attitudes, while a positive caregiver attitude was a significant predictor of full vaccination. Conversely, older child age and prior experience of adverse events following immunization (AEFI) were associated with reduced likelihood of completing the vaccination schedule. Although reported AEFIs were largely mild and self-limiting, their influence on caregiver perceptions underscores the importance of effective communication.
These findings highlight that awareness alone is insufficient to ensure completion and that sustained engagement strategies are required. Strengthening provider-led communication, Evidence for Action (E4A) strategies, improving caregiver follow-up systems, and reinforcing defaulter tracing and reminder mechanisms are critical to improving completion of later RTS, S doses. Integrating clear messaging on vaccine expectations and safety within routine immunization services will be essential to maximizing the public health impact of the malaria vaccine in endemic settings.
What is already known about the topic
- The RTS, S/AS01 malaria vaccine (Mosquirix™) was developed to target the early stages of Plasmodium falciparum and has been pilot implemented in Ghana, Kenya, and Malawi since 2019.
- Early trials and pilot implementation demonstrated a meaningful reduction in severe malaria cases and hospitalizations among vaccinated children.
- Despite its promise, vaccine uptake has varied across regions due to challenges such as cold chain logistics, financial constraints, and caregiver hesitancy.
- In Ghana, particularly Kadjebi District in the Oti Region, RTS, S vaccine coverage for doses has been below the national target of 90%, with challenges in sustaining coverage for subsequent doses.
- Adverse events following immunization (AEFI) with RTS, S are generally not serious, with fever being the most common side effect.
- Caregiver knowledge and attitudes are crucial to the vaccine’s acceptance and success.
- Previous studies indicate that caregivers’ knowledge, attitudes, and vaccine acceptance rates vary across regions but are generally positive where awareness is high.
What this study adds
- This study provides specific data on caregivers’ awareness, attitudes, uptake, AEFI, and willingness to accept the malaria vaccine in a pilot district in Ghana’s Oti Region.
- It reports high awareness (88% heard of the malaria vaccine) and positive attitudes (84.2% positive) towards malaria vaccination among caregivers.
- Uptake rates for RTS, S doses 1 and 2 met the national target (96.5% and 92.4%), but doses 3 and 4 had lower uptake (80.6% and 50.4%), showing a declining trend.
- AEFIs were reported by 43.9% of caregivers, mostly mild and non-serious, with fever as the predominant symptom.
- The study found high willingness (93.5%) among caregivers to vaccinate their children, and over half were willing to pay for the vaccine.
- It highlights the effectiveness of social mobilization strategies and the role of healthcare providers as the main source of vaccine information.
- The findings emphasize the need for targeted health promotion and routine mop-up vaccination efforts to improve coverage for later doses.
- The study also contributes local evidence supporting policy and programmatic adjustments to boost malaria vaccine uptake towards malaria control goals in endemic regions.
Acknowledgements
We acknowledge the effort of the Kadjebi District Health Management Team, data collectors and health workers, the University of Health and Allied Sciences, Fred N. Binka School of Public Health, Hohoe, Departmental Head of Epidemiological and Biostatistics, the Ethical Review Committee, Mr Mawuse, and Supervisor Dr. Wisdom Kwami Takramah for his relentless service to us.
Authors´ contributions
John Sonnyinado Duako Baffoe – Conception of design, Discussion, and drafting of the Manuscript
Joseph Abdul-Latif – Literature Review and Data Collector
Samuel Agyiri – Literature Review and Data Collector
Vida Dabaloro – Literature Review and Data Collector
Elijah Wamale Nlamba – Literature Review and Data Collector
Gregory Amenuvegbe – Editing and revising the manuscript
Joy Sefakor Mawusi – Data Analysis and Interpretation
Eric Takyi – Stakeholder Engagement
Rita Agyekumwah Asante – Editing and revising the manuscript
George Akowuah – Editing and revising the manuscript
Wisdom Kwame Takramah – Supervisor and revising the manuscript
Joyce Berkumwin Der – Supervisor and revising the manuscript
Donne Kofi Ameme – Editing and revising the manuscript
Austin Gideon Adobasom-Anane – Editing and revising the manuscript
George Asumah-Karikari Adu – Editing and revising the manuscript
| Variable | n | % (95% CI) |
|---|---|---|
| Caregiver characteristics | ||
| Age (years) | ||
| 15–17 | 6 | 1.6 (0.6–3.5) |
| 17–28 | 189 | 51.5 (46.3–56.7) |
| 28–45 | 169 | 46.0 (40.9–51.2) |
| 45–65 | 3 | 0.8 (0.2–2.4) |
| Sex | ||
| Female | 342 | 93.2 (90.0–95.5) |
| Male | 25 | 6.8 (4.5–10.0) |
| Marital status | ||
| Married | 302 | 82.3 (78.0–85.9) |
| Not married* | 65 | 17.7 (14.1–22.0) |
| Education level | ||
| No formal education | 47 | 12.8 (9.7–16.6) |
| Primary | 82 | 22.3 (18.3–26.9) |
| JHS/Middle | 151 | 41.1 (36.1–46.3) |
| SHS/Vocational | 40 | 10.9 (8.0–14.6) |
| Tertiary | 47 | 12.8 (9.7–16.6) |
| Employment status | ||
| Employed | 268 | 73.0 (68.2–77.4) |
| Not employed | 99 | 27.0 (22.6–31.8) |
| Religion | ||
| Christianity | 205 | 55.9 (50.7–61.0) |
| Islam | 162 | 44.1 (39.0–49.3) |
| Ethnicity | ||
| Ewe | 128 | 34.9 (30.2–39.9) |
| Kotokoli | 121 | 33.0 (28.4–37.9) |
| Akan/Twi | 33 | 9.0 (6.4–12.4) |
| Basare | 26 | 7.1 (4.9–10.1) |
| Hausa | 13 | 3.5 (2.1–6.0) |
| Other | 46 | 12.5 (9.5–16.3) |
| Child characteristics | ||
| Sex of child | ||
| Male | 202 | 55.0 (49.8–60.1) |
| Female | 165 | 45.0 (39.9–50.2) |
| Age group (months) | ||
| 5–11 | 78 | 21.3 (17.4–25.9) |
| 12–23 | 112 | 30.5 (26.0–35.4) |
| 24–59 | 177 | 48.2 (43.1–53.4) |
| Awareness of malaria vaccine | ||
| Ever heard of a malaria vaccine (Yes) | 323 | 87.5 (83.7–90.5) |
| Heard of the RTS, S vaccine (Yes) | 221 | 59.9 (54.8–64.8) |
| Sources of information† | ||
| Healthcare providers | 321 | 87.0 (83.2–90.0) |
| Friends/family | 15 | 4.1 (2.5–6.7) |
| Radio/Television | 4 | 1.1 (0.4–2.8) |
| Social media | 4 | 1.1 (0.4–2.8) |
| Other sources | 16 | 4.3 (2.7–6.9) |
| Notes: • Values are presented as frequency (percentage with 95% confidence interval) • Not married includes single, divorced/separated, and widowed • †Multiple responses allowed; percentages may not sum to 100% | ||
| Characteristics | Awareness | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Good (n = 264) | Poor (n = 103) | ||||
| Age group (years) | |||||
| 15–17 | 3 (50.0) | 3 (50.0) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 128 (67.7) | 61 (32.3) | 189 (100) | 2.10 (0.41–10.8), 0.372 | 1.96 (0.36–10.6), 0.438 |
| 28–45 | 131 (77.5) | 38 (22.5) | 169 (100) | 3.45 (0.67–17.7), 0.139 | 3.12 (0.58–16.8), 0.186 |
| 45–65 | 2 (66.7) | 1 (33.3) | 3 (100) | 2.00 (0.12–33.6), 0.620 | 1.88 (0.10–34.1), 0.650 |
| Sex | |||||
| Female | 248 (72.5) | 94 (27.5) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 16 (64.0) | 9 (36.0) | 25 (100) | 0.67 (0.29–1.57), 0.355 | 0.71 (0.29–1.75), 0.458 |
| Marital status | |||||
| Not married | 41 (63.1) | 24 (36.9) | 65 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Married | 223 (73.8) | 79 (26.2) | 302 (100) | 1.66 (0.95–2.89), 0.074 | 1.58 (0.87–2.85), 0.129 |
| Religion | |||||
| Christianity | 155 (75.6) | 50 (24.4) | 205 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Islam | 109 (67.3) | 53 (32.7) | 162 (100) | 0.66 (0.43–1.02), 0.061 | 0.71 (0.45–1.12), 0.142 |
| Ethnicity | |||||
| Akan/Twi | 20 (60.6) | 13 (39.4) | 33 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Kotokoli | 82 (67.8) | 39 (32.2) | 121 (100) | 1.37 (0.60–3.15), 0.454 | 1.31 (0.55–3.12), 0.545 |
| Ewe | 98 (76.6) | 30 (23.4) | 128 (100) | 2.13 (0.93–4.86), 0.072 | 1.98 (0.84–4.69), 0.118 |
| Hausa | 7 (53.8) | 6 (46.2) | 13 (100) | 0.76 (0.21–2.73), 0.679 | 0.81 (0.22–3.02), 0.754 |
| Basare | 17 (65.4) | 9 (34.6) | 26 (100) | 1.23 (0.42–3.60), 0.706 | 1.19 (0.39–3.62), 0.758 |
| Others | 40 (87.0) | 6 (13.0) | 46 (100) | 4.33 (1.39–13.5), 0.012 | 3.91 (1.18–12.9), 0.026 |
| Education level | |||||
| No education | 24 (51.1) | 23 (48.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 67 (81.7) | 15 (18.3) | 82 (100) | 4.28 (1.94–9.44), <0.001 | 3.96 (1.74–9.01), 0.001 |
| JHS/Middle | 111 (73.5) | 40 (26.5) | 151 (100) | 2.66 (1.36–5.19), 0.004 | 2.41 (1.18–4.92), 0.016 |
| SHS/Vocational | 30 (75.0) | 10 (25.0) | 40 (100) | 2.88 (1.16–7.17), 0.023 | 2.62 (1.01–6.80), 0.047 |
| Tertiary | 32 (68.1) | 15 (31.9) | 47 (100) | 2.04 (0.94–4.45), 0.071 | 1.88 (0.83–4.24), 0.130 |
| Employment status | |||||
| Not employed | 63 (63.6) | 36 (36.4) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 201 (75.0) | 67 (25.0) | 268 (100) | 1.71 (1.04–2.82), 0.034 | 1.62 (0.94–2.79), 0.081 |
| Sex of child | |||||
| Female | 113 (68.5) | 52 (31.5) | 165 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 151 (74.8) | 51 (25.2) | 202 (100) | 1.36 (0.88–2.10), 0.170 | 1.29 (0.81–2.05), 0.287 |
| Age of child (months) | |||||
| 5–11 | 49 (62.8) | 29 (37.2) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 | 78 (69.6) | 34 (30.4) | 112 (100) | 1.36 (0.73–2.54), 0.334 | 1.29 (0.67–2.48), 0.448 |
| 24–60 | 137 (77.4) | 40 (22.6) | 177 (100) | 2.02 (1.11–3.67), 0.022 | 1.86 (0.98–3.51), 0.056 |
| Characteristics | Attitude | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Good (n = 309) | Poor (n = 58) | ||||
| Awareness | |||||
| Poor | 63 (61.2) | 40 (38.8) | 103 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Good | 246 (93.2) | 18 (6.8) | 264 (100) | 8.73 (4.66–16.4), <0.001 | 3.98 (2.05–7.71), <0.001 |
| Age group (years) | |||||
| 15–17 | 4 (66.7) | 2 (33.3) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 150 (79.4) | 39 (20.6) | 189 (100) | 1.92 (0.35–10.6), 0.453 | 1.78 (0.31–10.1), 0.514 |
| 28–45 | 152 (89.9) | 17 (10.1) | 169 (100) | 4.47 (0.79–25.4), 0.090 | 3.91 (0.66–23.1), 0.137 |
| 45–65 | 3 (100) | 0 (0.0) | 3 (100) | — | — |
| Sex | |||||
| Female | 289 (84.5) | 53 (15.5) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 20 (80.0) | 5 (20.0) | 25 (100) | 0.73 (0.26–2.06), 0.553 | 0.78 (0.26–2.33), 0.655 |
| Marital status | |||||
| Not married | 49 (75.4) | 16 (24.6) | 65 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Married | 260 (86.1) | 42 (13.9) | 302 (100) | 2.02 (1.05–3.89), 0.036 | 1.83 (0.91–3.67), 0.089 |
| Religion | |||||
| Christianity | 178 (86.8) | 27 (13.2) | 205 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Islam | 131 (80.9) | 31 (19.1) | 162 (100) | 0.64 (0.37–1.12), 0.119 | 0.71 (0.39–1.29), 0.262 |
| Ethnicity | |||||
| Akan/Twi | 25 (75.8) | 8 (24.2) | 33 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Kotokoli | 101 (83.5) | 20 (16.5) | 121 (100) | 1.62 (0.64–4.09), 0.307 | 1.49 (0.57–3.89), 0.418 |
| Ewe | 113 (88.3) | 15 (11.7) | 128 (100) | 2.41 (0.93–6.23), 0.069 | 2.12 (0.79–5.67), 0.135 |
| Hausa | 9 (69.2) | 4 (30.8) | 13 (100) | 0.72 (0.17–3.04), 0.655 | 0.77 (0.17–3.49), 0.735 |
| Basare | 21 (80.8) | 5 (19.2) | 26 (100) | 1.34 (0.37–4.83), 0.656 | 1.29 (0.35–4.75), 0.700 |
| Others | 40 (87.0) | 6 (13.0) | 46 (100) | 2.13 (0.63–7.17), 0.221 | 1.98 (0.57–6.86), 0.283 |
| Education level | |||||
| No education | 30 (63.8) | 17 (36.2) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 67 (81.7) | 15 (18.3) | 82 (100) | 2.54 (1.14–5.64), 0.022 | 2.31 (0.99–5.41), 0.053 |
| JHS/Middle | 134 (88.7) | 17 (11.3) | 151 (100) | 4.46 (2.09–9.52), <0.001 | 3.91 (1.75–8.72), 0.001 |
| SHS/Vocational | 34 (85.0) | 6 (15.0) | 40 (100) | 3.21 (1.11–9.26), 0.031 | 2.84 (0.94–8.61), 0.064 |
| Tertiary | 44 (93.6) | 3 (6.4) | 47 (100) | 8.31 (2.25–30.7), 0.002 | 6.98 (1.79–27.2), 0.005 |
| Employment status | |||||
| Not employed | 78 (78.8) | 21 (21.2) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 231 (86.2) | 37 (13.8) | 268 (100) | 1.68 (0.94–3.01), 0.079 | 1.52 (0.82–2.82), 0.184 |
| Sex of child | |||||
| Female | 135 (81.8) | 30 (18.2) | 165 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 174 (86.1) | 28 (13.9) | 202 (100) | 1.38 (0.79–2.40), 0.254 | 1.29 (0.72–2.31), 0.392 |
| Age of child (months) | |||||
| 5–11 | 60 (76.9) | 18 (23.1) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 | 90 (80.4) | 22 (19.6) | 112 (100) | 1.23 (0.63–2.40), 0.546 | 1.16 (0.58–2.32), 0.672 |
| 24–60 | 159 (89.8) | 18 (10.2) | 177 (100) | 2.65 (1.36–5.15), 0.004 | 2.31 (1.14–4.68), 0.020 |
| Characteristics | Willingness | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Willing (n = 344) | Not willing (n = 23) | ||||
| Awareness | |||||
| Poor | 88 (85.4) | 15 (14.6) | 103 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Good | 256 (97.0) | 8 (3.0) | 264 (100) | 5.45 (2.19–13.6), <0.001 | 3.21 (1.18–8.72), 0.023 |
| Attitude | |||||
| Negative | 47 (81.0) | 11 (19.0) | 58 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Positive | 297 (96.1) | 12 (3.9) | 309 (100) | 5.80 (2.55–13.2), <0.001 | 4.12 (1.71–9.92), 0.002 |
| Age group (years) | |||||
| 15–17 | 5 (83.3) | 1 (16.7) | 6 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 17–28 | 177 (93.7) | 12 (6.3) | 189 (100) | 2.95 (0.32–27.3), 0.342 | 2.61 (0.27–25.2), 0.403 |
| 28–45 | 160 (94.7) | 9 (5.3) | 169 (100) | 3.56 (0.38–33.4), 0.263 | 3.01 (0.31–29.5), 0.340 |
| 45–65 | 2 (66.7) | 1 (33.3) | 3 (100) | 0.40 (0.02–7.02), 0.530 | 0.36 (0.02–6.54), 0.493 |
| Sex | |||||
| Female | 321 (93.9) | 21 (6.1) | 342 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Male | 23 (92.0) | 2 (8.0) | 25 (100) | 0.75 (0.16–3.48), 0.713 | 0.81 (0.17–3.89), 0.793 |
| Education level | |||||
| No education | 40 (85.1) | 7 (14.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 77 (93.9) | 5 (6.1) | 82 (100) | 2.69 (0.84–8.64), 0.095 | 2.11 (0.61–7.25), 0.238 |
| JHS/Middle | 144 (95.4) | 7 (4.6) | 151 (100) | 3.60 (1.18–10.9), 0.024 | 2.84 (0.87–9.23), 0.083 |
| SHS/Vocational | 38 (95.0) | 2 (5.0) | 40 (100) | 3.33 (0.68–16.3), 0.137 | 2.91 (0.55–15.4), 0.207 |
| Tertiary | 45 (95.7) | 2 (4.3) | 47 (100) | 3.94 (0.81–19.2), 0.090 | 3.12 (0.60–16.3), 0.175 |
| Employment status | |||||
| Not employed | 89 (89.9) | 10 (10.1) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 255 (95.1) | 13 (4.9) | 268 (100) | 2.21 (0.95–5.15), 0.065 | 1.87 (0.76–4.59), 0.173 |
| AEFI experienced | |||||
| No | 197 (95.6) | 9 (4.4) | 206 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Yes | 147 (91.3) | 14 (8.7) | 161 (100) | 0.48 (0.20–1.12), 0.090 | 0.39 (0.16–0.94), 0.036 |
| Characteristics | Vaccination Status | Total n (%) | COR (95% CI), p-value | AOR (95% CI), p-value | |
|---|---|---|---|---|---|
| Fully vaccinated n (%) | Not fully vaccinated n (%) | ||||
| Age of child (months) | |||||
| 5–11 (Dose 1) | 71 (91.0) | 7 (9.0) | 78 (100) | 1.00 (Ref) | 1.00 (Ref) |
| 12–23 (Doses 1–3) | 78 (69.6) | 34 (30.4) | 112 (100) | 0.23 (0.10–0.52), <0.001 | 0.26 (0.11–0.60), 0.002 |
| 24–59 (Doses 1–4) | 89 (50.3) | 88 (49.7) | 177 (100) | 0.11 (0.05–0.24), <0.001 | 0.14 (0.06–0.31), <0.001 |
| Caregiver education | |||||
| No education | 24 (51.1) | 23 (48.9) | 47 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Primary | 56 (68.3) | 26 (31.7) | 82 (100) | 2.05 (1.01–4.17), 0.046 | 1.89 (0.90–3.96), 0.093 |
| JHS/Middle | 109 (72.2) | 42 (27.8) | 151 (100) | 2.47 (1.28–4.78), 0.007 | 2.21 (1.11–4.39), 0.024 |
| SHS/Vocational | 28 (70.0) | 12 (30.0) | 40 (100) | 2.26 (0.95–5.35), 0.066 | 1.98 (0.80–4.93), 0.141 |
| Tertiary | 21 (44.7) | 26 (55.3) | 47 (100) | 0.76 (0.33–1.76), 0.524 | 0.81 (0.34–1.96), 0.642 |
| Caregiver attitude | |||||
| Negative | 24 (41.4) | 34 (58.6) | 58 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Positive | 214 (69.3) | 95 (30.7) | 309 (100) | 3.17 (1.78–5.66), <0.001 | 2.74 (1.47–5.12), 0.001 |
| AEFI experienced | |||||
| No | 156 (75.7) | 50 (24.3) | 206 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Yes | 82 (50.9) | 79 (49.1) | 161 (100) | 0.34 (0.22–0.54), <0.001 | 0.41 (0.25–0.67), <0.001 |
| Employment status | |||||
| Not employed | 58 (58.6) | 41 (41.4) | 99 (100) | 1.00 (Ref) | 1.00 (Ref) |
| Employed | 180 (67.2) | 88 (32.8) | 268 (100) | 1.44 (0.89–2.33), 0.136 | 1.29 (0.77–2.18), 0.337 |
| Overall | 238 (64.9) | 129 (35.1) | 367 (100) | — | — |
| AEFI Characteristic | Frequency (n) | Percentage (%) |
|---|---|---|
| Experienced any AEFI | 161 | 43.9 |
| No AEFI reported | 206 | 56.1 |
| Type of AEFI reported | ||
| Fever | 147 | 91.3 |
| Swelling at the injection site | 9 | 5.6 |
| Vomiting | 3 | 1.9 |
| Others | 2 | 1.2 |
| AEFI severity | ||
| Non-serious | 145 | 90.1 |
| Serious | 16 | 9.9 |
| AEFI outcome | ||
| Recovered | 161 | 100 |
| Did AEFI affect the acceptance of other vaccines? | ||
| No | 125 | 77.6 |
| Yes | 36 | 22.4 |


References
- Musa-Booth T, Enobun B, Agbomola A, Shiff C. Knowledge, attitude and willingness to accept the RTS,S malaria vaccine among mothers in Abuja, Nigeria. Ann Afr Med Res [Internet]. 2021 Sep 13 [cited 2026 Apr 17];4(1):128. doi:10.4081/aamr.2021.128 Available from: https://africa.pagepress.net/aamr/article/view/441
- World Health Organization. Malaria vaccine: WHO position paper, January 2016 – Recommendations. Vaccine [Internet]. 2017 Apr 3 [cited 2026 Apr 17];36(25):3576–7. doi:10.1016/j.vaccine.2016.10.047 Available from: https://www.sciencedirect.com/science/article/abs/pii/S0264410X16309823
- Thomas M, Kohli V, King D. Barriers to Childhood Immunization: Findings from a Needs Assessment Study. Home Health Care Serv Q [Internet]. 2004 Jun 28 [cited 2026 Apr 17];23(2):19–39. doi:10.1300/J027v23n02_02 Available from: https://www.tandfonline.com/doi/abs/10.1300/J027v23n02_02
- Odikro MA, Kenu E, Malm KL, Asiedu-Bekoe F, Noora CL, Frimpong J, Calys-Tagoe B, Koram KA. Epidemiology of COVID-19 outbreak in Ghana, 2020. Ghana Med J [Internet]. 2020 Dec 31 [cited 2026 Apr 17];54(Suppl 4):5–15. doi:10.4314/gmj.v54i4s.3 Available from: https://www.ajol.info/index.php/gmj/article/view/202974
- World Health Organization. Global technical strategy for malaria 2016-2030, 2021 update [Internet]. Geneva (Switzerland): World Health Organization; 2021 Jul 19 [cited 2026 Apr 17]. 30 p. Available from: https://www.who.int/publications/i/item/9789240031357
- Bejon P, White MT, Olotu A, Bojang K, Lusingu JPA, Salim N, Otsyula NN, Agnandji ST, Asante KP, Owusu-Agyei S, Abdulla S, Ghani AC. Efficacy of RTS,S malaria vaccines: individual-participant pooled analysis of phase 2 data. Lancet Infect Dis [Internet]. 2013 Mar 1 [cited 2026 Apr 17];13(4):319–27. doi:10.1016/S1473-3099(13)70005-7 Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(13)70005-7/fulltext
- Galactionova K, Tediosi F, Camponovo F, Smith TA, Gething PW, Penny MA. Country specific predictions of the cost-effectiveness of malaria vaccine RTS,S/AS01 in endemic Africa. Vaccine [Internet]. 2016 Nov 24 [cited 2026 Apr 17];35(1):53–60. doi:10.1016/j.vaccine.2016.11.042 Available from: https://www.sciencedirect.com/science/article/pii/S0264410X16311033?via%3Dihub
- PATH Malaria Vaccine Initiative. RTS,S (MosquirixTM) Frequently Asked Questions (FAQs). Seattle (WA): PATH Malaria Vaccine Initiative; 2016. 5 p.
- World Health Organization. Malaria vaccines (RTS,S and R21) [Internet]. Geneva (Switzerland): WHO; 2026 Feb 4 [cited 2026 Apr 17]. Available from: https://www.who.int/news-room/questions-and-answers/item/q-a-on-rts-s-malaria-vaccine
- Olotu A, Fegan G, Wambua J, Nyangweso G, Leach A, Lievens M, Kaslow DC, Njuguna P, Marsh K, Bejon P. Seven-Year Efficacy of RTS,S/AS01 Malaria Vaccine among Young African Children. N Engl J Med [Internet]. 2016 Jun 30 [cited 2026 Apr 17];374(26):2519–29. doi:10.1056/NEJMoa1515257 Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa1515257
- Silver KL, Singer PA. SDGs: start with maternal, newborn, and child health cluster. Lancet [Internet]. 2014 Sep 20 [cited 2026 Apr 17];384(9948):1093–4. doi:10.1016/S0140-6736(14)61667-6 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(14)61667-6/fulltext
- Simkus J. Cross-Sectional Study: Definition, Designs & Examples [Internet]. London (UK): Simply Psychology; 2023 Jul 31 [cited 2026 Apr 17]. Available from: https://www.simplypsychology.org/what-is-a-cross-sectional-study.html
- Setia M. Methodology series module 3: Cross-sectional studies. Indian J Dermatol [Internet]. 2016 May-Jun [cited 2026 Apr 17];61(3):261-64. doi:10.4103/0019-5154.182410 Available from: https://journals.lww.com/ijd/fulltext/2016/61030/methodology_series_module_3__cross_sectional.3.aspx
- Ghana Statistical Service (GSS). Kassena Nankana East Municipality. Accra (Ghana): GSS; 2014.
- UNICEF Ghana. Ghana Expands Malaria Vaccine Rollout [Internet]. New York (NY): UNICEF; 2024 Sep 24 [cited 2026 Apr 17]. Available from: https://www.unicef.org/ghana/press-releases/ghana-expands-malaria-vaccine-rollout
- Adjei MR, Tweneboah PO, Bonsu G, Baafi JV, Amponsa-Achiano K, Asiedu-Bekoe F, Ohene SA, Kuma-Aboagye P, Grobusch MP. An assessment of Ghana’s pilot of the RTS,S malaria vaccine implementation programme; 2019–2021: a retrospective study. Malar J [Internet]. 2024 Sep 27 [cited 2026 Apr 17];23(1):290. doi:10.1186/s12936-024-05113-8 Available from: https://link.springer.com/article/10.1186/s12936-024-05113-8
- Adeshina OO, Nyame S, Milner J, Milojevic A, Asante KP. Barriers and facilitators to nationwide implementation of the malaria vaccine in Ghana. Health Policy Plan [Internet]. 2022 Sep 9 [cited 2026 Apr 17];38(1):28–37. doi:10.1093/heapol/czac077 Available from: https://academic.oup.com/heapol/article/38/1/28/6694852
- Ampomah S. Ghana has expanded the Malaria Vaccine Exercise [Internet]. Accra (Ghana): Ministry of Health (Ghana); 2023 Feb 21 [cited 2026 Apr 17]. Available from: https://www.moh.gov.gh/ghana-has-expanded-the-malaria-vaccine-exercise/
- Naing L, Nordin RB, Abdul Rahman H, Naing YT. Sample size calculation for prevalence studies using Scalex and ScalaR calculators. BMC Med Res Methodol [Internet]. 2022 Jul 30 [cited 2026 Apr 17];22(1):209. doi:10.1186/s12874-022-01694-7 Available from: https://link.springer.com/article/10.1186/s12874-022-01694-7
- Pourhoseingholi MA, Vahedi M, Rahimzadeh M. Sample size calculation in medical studies. Gastroenterol Hepatol Bed Bench. 2013 Winter [cited 2026 Apr 17];6(1):14–7. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC4017493/
- Tabiri D, Ouédraogo JCRP, Nortey PA. Factors associated with malaria vaccine uptake in Sunyani Municipality, Ghana. Malar J [Internet]. 2021 Jul 27 [cited 2026 Apr 17];20(1):325. doi:10.1186/s12936-021-03857-1 Available from: https://link.springer.com/article/10.1186/s12936-021-03857-1
- Yeboah D, Owusu-Marfo J, Agyeman YN. Predictors of malaria vaccine uptake among children 6–24 months in the Kassena Nankana Municipality in the Upper East Region of Ghana. Malar J [Internet]. 2022 Nov 16 [cited 2026 Apr 17];21(1):339. doi:10.1186/s12936-022-04378-1 Available from: https://link.springer.com/article/10.1186/s12936-022-04378-1
- Asmare G. Willingness to accept malaria vaccine among caregivers of under-5 children in Southwest Ethiopia: a community based cross-sectional study. Malar J [Internet]. 2022 May 12 [cited 2026 Apr 17];21(1):146. doi:10.1186/s12936-022-04164-z Available from: https://link.springer.com/article/10.1186/s12936-022-04164-z
- One Community Global. Bloom’s Taxonomy Revised: An Open Source Resource Guide and Collaboration [Internet]. San Gabriel (CA): One Community Global; 2013 [cited 2026 Apr 17]. Available from: https://onecommunityglobal.org/blooms-taxonomy/
- Likert R. A technique for the measurement of attitudes. Arch Psychol. 1932;22(140):5–55.
- Harris JK. Primer on binary logistic regression. Fam Med Community Health [Internet]. 2021 Dec 23 [cited 2026 Apr 17];9(Suppl 1):e001290. doi:10.1136/fmch-2021-001290 Available from: https://fmch.bmj.com/content/9/Suppl_1/e001290
- Sperandei S. Understanding logistic regression analysis. Biochem Med [Internet]. 2014 Feb 15 [cited 2026 Apr 17];24(1):12–8. doi:10.11613/BM.2014.003 Available from: https://www.biochemia-medica.com/en/journal/24/1/10.11613/BM.2014.003
- Bingham A, Gaspar F, Lancaster K, Conjera J, Collymore Y, Ba-Nguz A, Vansadia P, Bingham A. Community perceptions of malaria and vaccines in two districts of Mozambique. Malar J [Internet]. 2012 Nov 28 [cited 2026 Apr 17];11(1):394. doi:10.1186/1475-2875-11-394 Available from: https://link.springer.com/article/10.1186/1475-2875-11-394
- Febir LG, Asante KP, Dzorgbo DBS, Senah KA, Letsa TS, Owusu-Agyei S. Community perceptions of a malaria vaccine in the Kintampo districts of Ghana. Malar J [Internet]. 2013 May 7 [cited 2026 Apr 17];12(1):156. doi:10.1186/1475-2875-12-156 Available from: https://link.springer.com/article/10.1186/1475-2875-12-156
- Ojakaa DI, Ofware P, Machira YW, Yamo E, Collymore Y, Ba-Nguz A, Vansadia P, Bingham A. Community perceptions of malaria and vaccines in the South Coast and Busia regions of Kenya. Malar J [Internet]. 2011 May 30 [cited 2026 Apr 17];10(1):147. doi:10.1186/1475-2875-10-147 Available from: https://link.springer.com/article/10.1186/1475-2875-10-147
- Saaka SA, Mohammed K, Pienaah CKA, Luginaah I. Child malaria vaccine uptake in Ghana: Factors influencing parents’ willingness to allow vaccination of their children under five (5) years. PLoS One [Internet]. 2024 Jan 19 [cited 2026 Apr 17];19(1):e0296934. doi:10.1371/journal.pone.0296934 Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0296934
- Vandoolaeghe P, Schuerman L. The RTS,S/AS01 malaria vaccine in children 5 to 17 months of age at first vaccination. Expert Rev Vaccines [Internet]. 2016 Nov 18 [cited 2026 Apr 17];15(12):1481–93. doi:10.1080/14760584.2016.1236689 Available from: https://www.tandfonline.com/doi/full/10.1080/14760584.2016.1236689
- Larson HJ, Cooper LZ, Eskola J, Katz SL, Ratzan S. Addressing the vaccine confidence gap. Lancet [Internet]. 2011 Jun 9 [cited 2026 Apr 17];378(9790):526–35. doi:10.1016/S0140-6736(11)60678-8 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(11)60678-8/abstract
- Osoro CB, Ochodo E, Kwambai TK, Otieno JA, Were L, Sagam CK, Owino EJ, Kariuki S, Ter Kuile FO, Hill J. Policy uptake and implementation of the RTS,S/AS01 malaria vaccine in sub-Saharan African countries: status 2 years following the WHO recommendation. BMJ Glob Health [Internet]. 2024 Apr 30 [cited 2026 Apr 17];9(4):e014719. doi:10.1136/bmjgh-2023-014719 Available from: https://gh.bmj.com/content/9/4/e014719
- Adjei MR, Tweneboah PO, Bonsu G, Baafi JV, Amponsa-Achiano K, Asiedu-Bekoe F, Ohene SA, Kuma-Aboagye P, Grobusch MP. Post introduction evaluation of the malaria vaccine implementation programme in Ghana, 2021. BMC Public Health [Internet]. 2023 Mar 29 [cited 2026 Apr 17];23(1):586. doi:10.1186/s12889-023-15481-6 Available from: https://link.springer.com/article/10.1186/s12889-023-15481-6
- Zacharia MM, Ziema SA, Abanga WA, Kyeremeh RA, Tamal CS, Adjei MR, Kubio C. RTS, S malaria vaccination among children aged 24–59 months in the Sunyani Municipality, Ghana; 2023. Vaccine [Internet]. 2024 Oct 31 [cited 2026 Apr 17];42(26):126490. doi:10.1016/j.vaccine.2024.126490 Available from: https://www.sciencedirect.com/science/article/pii/S0264410X24011721
- Adjei MR, Tweneboah PO, Bawa JT, Baafi JV, Kubio C, Amponsa-Achiano K, Asiedu-Bekoe F, Kuma-Aboagye P, Grobusch MP, Ohene SA. Trend of RTS,S vaccine uptake in the malaria vaccine implementing programme (MVIP) pilot regions, Ghana; 2019–2022. Heliyon [Internet]. 2024 Oct [cited 2026 Apr 17];10(19):e38858. doi:10.1016/j.heliyon.2024.e38858 Available from: https://www.sciencedirect.com/science/article/pii/S2405844024123456
