Research | Open Access | Volume 9 (2): Article 72 | Published: 05 May 2026
Increasing incidence of malaria in pregnancy in Uganda, 2015–2023
Menu, Tables and Figures
Navigate this article
Tables
| Table 1: Regional trends of malaria in pregnancy in Uganda, 2015–2023 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Region | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | 2023 | Sens slope | (95% CI) |
| Acholi | 276 | 335 | 307 | 216 | 393 | 390 | 296 | 342 | 301 | 2.8 | (-23–27) |
| Ankole | 92 | 106 | 95 | 39 | 36 | 46 | 42 | 54 | 52 | -5.2 | (-13–3.5) |
| Bugisu | 126 | 109 | 125 | 73 | 99 | 88 | 112 | 140 | 122 | 1.3 | (-7.6–11) |
| Bukedi | 162 | 132 | 119 | 70 | 68 | 81 | 180 | 369 | 300 | 14 | (-21–56) |
| Bunyoro | 112 | 131 | 160 | 96 | 152 | 165 | 160 | 220 | 185 | 9.6 | (4–21) |
| Busoga | 183 | 213 | 226 | 184 | 230 | 281 | 321 | 367 | 396 | 27 | (18–43) |
| Kampala | 65 | 45 | 38 | 41 | 43 | 48 | 40 | 44 | 44 | -0.15 | (-3–1.2) |
| Karamoja | 231 | 185 | 166 | 143 | 131 | 166 | 161 | 160 | 149 | -5.1 | (-17–5) |
| Kigezi | 47 | 50 | 35 | 16 | 27 | 28 | 27 | 34 | 28 | -1.9 | (-5–2.3) |
| Lango | 248 | 283 | 232 | 158 | 285 | 289 | 249 | 378 | 355 | 15 | (-6.8–31) |
| North Central | 124 | 129 | 141 | 91 | 135 | 143 | 127 | 166 | 139 | 3.1 | (-1.3–9.6) |
| SouthCentral | 108 | 108 | 110 | 70 | 75 | 87 | 62 | 89 | 87 | -2.9 | (-8.3–3) |
| Teso | 299 | 314 | 311 | 251 | 278 | 325 | 354 | 399 | 419 | 15 | (4.7–34) |
| Tooro | 106 | 126 | 116 | 72 | 92 | 117 | 91 | 112 | 103 | -1.5 | (-7–6.2) |
| West Nile | 208 | 223 | 237 | 236 | 315 | 321 | 321 | 345 | 304 | 15 | (6.5–25) |
Table 1: Regional trends of malaria in pregnancy in Uganda, 2015–2023
Figures

Keywords
- Malaria in Pregnancy
- Incidence
- Patterns
- Distribution
- Uganda
Charity Mutesi1,&, Richard Migisha1, Patrick Kwizera1, Hildah Tendo Nansikombi1, John Rek1,2, Gerald Rukundo2, Alex Riolexus Ario1
1Uganda Public Health Fellowship Program, Uganda National Institute of Public Health, Kampala, Uganda, 2National Malaria Elimination Division, Ministry of Health, Kampala, Uganda
&Corresponding author: Charity Mutesi, Uganda Public Health Fellowship Program, Uganda National Institute of Public Health, Kampala, Uganda, Email: charitymutesi@uniph.go.ug; ORCID: https://orcid.org/0009-0003-5986-2211
Received: 20 Oct 2025, Accepted: 02 May 2026, Published: 05 May 2026
Domain: Infectious Disease Epidemiology
Keywords: Malaria in pregnancy, incidence, patterns, distribution, Uganda
©Charity Mutesi et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Charity Mutesi et al., Increasing incidence of malaria in pregnancy in Uganda, 2015–2023. Journal of Interventional Epidemiology and Public Health. 2026; 9(2):72. https://doi.org/10.37432/jieph-d-25-00247
Abstract
Introduction: Malaria in pregnancy (MiP) is associated with increased maternal and fetal health risks. In Uganda, despite widespread implementation of preventive strategies, malaria upsurges, including the 2022 epidemic, have raised concerns about the impact on MiP cases, admissions, and deaths. We described the trends and distribution of MiP incidence, as well as related admissions and fatalities in Uganda from 2015 to 2023.
Methods: We conducted a retrospective descriptive study using routinely reported MiP data in Uganda, from 2015 to 2023, obtained through the District Health Information System 2 (DHIS2). MiP incidence was defined as confirmed malaria cases per 1,000 pregnant women. Malaria test positivity rate, admissions, and deaths were also analyzed. Trends were assessed using the Mann–Kendall test and Sen’s slope, and stratified by age and region. Statistical analyses were conducted using Stata version 17, and the spatial distribution was mapped using Quantum Geographic Information System.
Results: A total of 2,808,426 MiP cases were reported among 16,462,878 pregnant women with an average annual incidence of 170 cases per 1,000 pregnant women. There was an increase of approximately 7 cases per year (Sen’s slope: 6.7 cases per year, 95%CI=1.3–17), with the highest increase observed in 2022. Regionally, significant increases were observed in Busoga (27 cases per year, 95%CI: 18–43), Teso (15 per year, 95%CI: 4.7–34), Bunyoro (10 per year, 95%CI: 4–21), and West Nile (15 per year, 95%CI: 6.5–25). Age-stratified analyses showed MiP among adolescents (10–19 years) increased from 236 per 1,000 in 2020 to 351 per 1,000 in 2023 (p=0.013), compared to adults ≥20 years (121 per 1,000 to 187 per 1,000, p=0.017). No significant change in trends was observed in MiP admissions or mortality.
Conclusion: There was an overall increase in MiP. MiP increased in both adolescent and older women, with a disproportionately higher burden among adolescents and in high-malaria-burden regions. Strengthening targeted strategies, including adolescent-focused interventions, enhanced regional surveillance, and improved access to preventive and curative services, is critical to reducing the burden of MiP in Uganda.
Introduction
Malaria continues to pose a significant public health threat, particularly for pregnant women and children under five years of age [1]. Malaria in Pregnancy (MiP) is particularly common among adolescent girls, who are at higher risk due to physiological and socio-economic factors such as lack of adequate protective immunity and limited access to healthcare services [2]. Malaria infection during pregnancy can cause severe disease and death in pregnant women, with placental sequestration of the parasite affecting fetal development and leading to poor fetal growth and low birth weight [3]. Maternal anemia, primarily caused by the destruction of red blood cells by the malaria parasites, also increases the risk of maternal death during and after childbirth, as well as contributing to stillbirths and preterm births [4, 5]. This placental infection can have long-term impacts on child growth and cognitive development and is a major risk factor for perinatal, neonatal, and infant mortality [5, 6, 7].
Globally, in 2022, there were approximately 36 million pregnancies in 33 countries with moderate to high malaria transmission, of which 12.7 million (36%) were affected by MiP [8]. In Africa, more than 30 million women become pregnant in malaria-endemic regions, where they face an increased risk of malaria infection compared to their non-pregnant counterparts [1, 9]. In Uganda, different studies have shown prevalence estimates of MiP ranging from <10% in low-transmission settings to over 25% in high-burden regions [10, 11]. The distribution of MiP in Uganda is influenced by several factors, including individual, geographic location, seasonality, and socio-economic conditions [12].
To avert the consequences of MiP, the World Health Organization (WHO) recommends a combination of interventions that are integrated as part of the routine antenatal care services. These include provision of Long-Lasting Insecticide-treated bed nets (LLINs) at first ANC visit (ANC1) and promotion of their use at all subsequent visits, effective treatment of malaria and anaemia in malaria-endemic areas, and use of intermittent preventive treatment in pregnancy using sulphadoxine-pyrimethamine (IPTp) [13]. Uganda has made notable progress in implementing MiP interventions, including the provision of LLINs to pregnant women both during ANC visits and mass campaigns, IPTp, indoor residual spraying (IRS) in areas of high malaria transmission, such as in the West Nile region, and prompt diagnosis and case management through the test and treat policy [14].
In response to malaria upsurges in 2019, Uganda revised its guidelines for malaria in pregnancy [15]. These guidelines mandate that every pregnant woman attending her first antenatal (ANC1) undergo malaria testing to enable timely diagnosis and treatment. According to the Uganda Malaria Indicator Survey (UMIS) 2018/2019, 95% attended ANC1, 65% of the pregnant women slept under an ITN, while 41% received IPTP3, still below the set targets of 100% ANC attendance for all mothers and 85% uptake of MiP intervention [16]. The proportion of pregnant women attending ANC1 is approximately 98-99%, based on the 2022 Uganda Demographic Health Survey report, implying that almost every pregnant woman receives at least one ANC visit [17]. While ANC provides an opportunity for early malaria screening and initiation of preventive interventions, factors such as long distances to ANC clinics and transport costs, stock-outs of medication (IPTp) at health facilities, and insufficient information provided by health workers may influence timely attendance and uptake of subsequent preventive services [18]. In 2022, Uganda experienced a resurgence of malaria, with some areas reporting increases of over 30% in confirmed cases, and outbreaks were documented in more than 75 districts [8, 19]. Despite progress in implementing MiP prevention strategies, the effect of the malaria resurgence and the extent of regional variations in MiP remain poorly characterized. Understanding temporal and spatial trends in MiP incidence and outcomes is essential for refining prevention strategies and supporting progress toward national malaria reduction and elimination targets. We described the trends and distribution of MiP incidence, as well as MiP admissions and fatalities, in Uganda, 2015–2023.
Methods
Study setting
Uganda is divided into 15 health regions, with the majority of the regions being malaria-endemic. Pregnant women in these settings are particularly vulnerable to malaria due to reduced immunity, especially among primigravidae. Additional risk factors include low socioeconomic status, suboptimal use of insecticide-treated nets, and incomplete uptake of intermittent preventive treatment in pregnancy (IPTp). Two regions, i.e., West Nile and Acholi, are classified as areas of high malaria transmission, characterized by an annual parasite index (API) greater than 450 cases per 1,000 population. Five regions (Lango, Karamoja, Teso, Bukedi, and Busoga) fall under moderate malaria transmission zones, with an API ranging from 251 to 450 cases per 1,000 population. Six regions (South Central, North Central, Kampala, Ankole, Tooro, Bugisu, and Bunyoro) are categorized as low malaria transmission areas, with an API of 101 to 250 cases per 1,000 population. The Kigezi Region is designated as having very low malaria transmission, with an API of 100 or fewer cases per 1,000 population annually. Kigezi is also a target area for malaria elimination under Uganda’s National Malaria Strategic Plan 2025 [20].
Uganda’s health system is decentralized and varies by both the facility type and level, with both public and private sectors. The public health sector consists of a tiered arrangement from the community-level Village Health Teams (VHTs), followed by Health Centers (HC) II’s that provide outpatient care, including antenatal services, HC III’s facilities offering outpatient, inpatient, and maternity care, HC IV’s with surgical capacity, and general hospitals providing comprehensive services. Beyond these, regional and national referral hospitals provide specialized and tertiary care [21, 22]. The private health system consists of accredited private not-for-profit and private-for-profit facilities that also deliver health services and report into the DHIS-2.
Malaria prevention and treatment are integrated into routine antenatal care at various healthcare levels, including primary health centers (HC II’s, HC III’s, and HC IV’s), district hospitals, and regional referral hospitals. These services are provided at government-owned facilities with free care or private facilities where mothers pay a fee.
Study design, data source, and variables
We conducted a retrospective descriptive study using routinely reported data from government health facilities and accredited non-government/private ones from 2015 to 2023. The design was well-suited for our study objectives of describing trends and the spatial distribution of MiP using existing data. The data is summarized through the Health Management Information System (HMIS) monthly forms, for outpatient and inpatient records, and is then uploaded into the District Health Information System version 2 (DHIS2). The HMIS forms for outpatient and inpatient records include reports on monthly outpatient and inpatient department visits, diagnoses, maternal and child health (MCH), HIV/AIDS services, laboratory results, and stock levels of essential medications, among other information. Health facilities are expected to submit complete monthly HMIS reports through DHIS2 by the 15th day of the subsequent month, in line with national reporting timelines [23]. We extracted data on annual first antenatal visits (ANC1), annual malaria in pregnancy outpatient and inpatient cases, and deaths, as well as malaria positivity rates. We further extracted data on reporting rates by the health facilities.
Inclusion and exclusion criteria
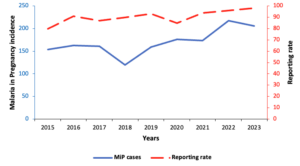
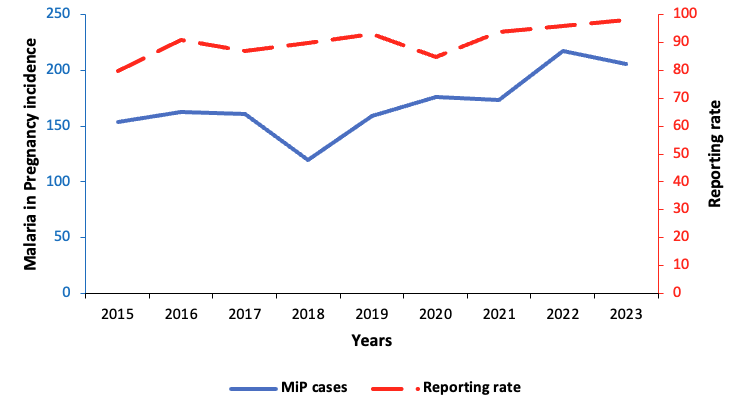
We included all health facilities in Uganda that reported malaria in pregnancy (MiP)-related variables of interest (ANC1 attendance, MiP cases, admissions, and deaths) for ≥80% of the months during 2015–2023. Health facilities that did not consistently report the variables of interest, defined as reporting data for <80% of the months during the study period, were excluded from the analysis. This threshold was selected to ensure stability and reliability of temporal trend estimates. Based on the inclusion criteria, 7228 facilities were included in the final analysis, while 1518/8746 (17%) were excluded due to incomplete reporting on the malaria indicators in the study. For facilities included in the study, reporting rates ranged from 80% in 2015 to 98% in 2023 (Figure 1).
Data cleaning
The DHIS2 has built-in validation checks, such as range limits at the point of data entry. During data cleaning, outliers were identified and cross-checked against aggregated HMIS forms, and where discrepancies persisted, verification was conducted with the respective health facilities. To ensure data quality, reporting completeness was defined as facility-level reporting of key MiP indicators (ANC1 attendance, MiP cases, admissions, and deaths) for at least 80% of the months between 2015 and 2023.
Data analysis and management
MiP was defined as the number of confirmed malaria cases among pregnant women per 1,000 ANC1 visits. First antenatal care visits were used as a proxy for the pregnant population, because ANC is nearly universal in Uganda, with approximately 98-99% of pregnant women attending ANC1 [17].
Malaria test positivity rate was defined as the number of laboratory-confirmed malaria cases among pregnant women divided by the number of pregnant women tested for malaria × 100.
MiP admission and death rates were calculated as the proportions of cases that were admitted and those that resulted in death. Age was categorized into two groups: 10-19 (adolescents) and ≥20 years (adults), as aggregated by DHIS2 antenatal data. Trends were analyzed using line graphs, the Mann-Kendall test to assess statistical significance, and Sen’s slope to estimate the magnitude of change over time [24]. The direction and magnitude of trends were assessed using the Mann-Kendall tau and Sen’s slope estimates. A significance level of p<0.05 indicated a statistically significant trend, while p-values equal to or greater than 0.05 were considered non-significant. We used the non-parametric Mann-Kendall test and Sen’s slope because they do not assume normality, are robust to outliers, and can detect monotonic changes over time, making them well-suited for routine surveillance data that may be skewed or irregular, unlike linear regression, which requires normally distributed residuals and linearity [24]. Trends were stratified by age and region. The age-specific analysis covered the period 2020–2023, when the age variable for MiP first became available in DHIS2. Quarterly data were used to capture short-term temporal patterns in MiP, as the Mann-Kendall test requires at least 10 data points [24]. Statistical analysis was conducted using Stata version 17, and choropleth maps showing MiP distribution in Uganda were created using Quantum Geographic Information System (QGIS) version 3.34.3.
Ethical consideration
Our study utilized secondary, anonymized data routinely collected through the District Health Information System 2 (DHIS2) managed by the Uganda Ministry of Health. The study did not involve direct contact with human participants, nor was any identifiable personal data used. The Uganda Public Health Fellowship Program is part of the National Rapid Response Team and has been granted permission to access and analyse surveillance data in the DHIS2 and other data, such as survey and field investigation data, to inform decision-making in the control and prevention of outbreaks and public health programming. Additionally, the Ministry of Health has also granted the program permission to disseminate the information through scientific publications. We stored the abstracted dataset in a password-protected computer and only shared it with the investigation team. In addition, the Office of the Associate Director for Science, U.S. Centres for Disease Control (CDC) and Prevention, determined that this study was not human subjects research with the primary intent of improving the use of surveillance data to guide public health planning and practice. This activity was reviewed by the CDC and was conducted consistently with applicable federal law and CDC policy (§ 149 §See e.g., 45 C.F.R. part 46, 21 C.F.R. part 56; 42 U.S.C. §241(d); 5 U.S.C. §552a; 44 U.S.C. §3501 et seq). Individual informed consent was not required based on the fact that the analysis used secondary programmatic data collected as part of routine public health surveillance, consistent with the Uganda Public Health Act (Cap. 281), which permits the use of such data for disease surveillance and response without individual consent.
Results
Trends of incidence of malaria in pregnancy, Uganda, 2015–2023
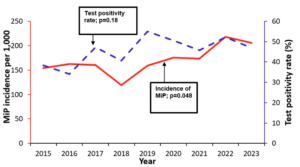
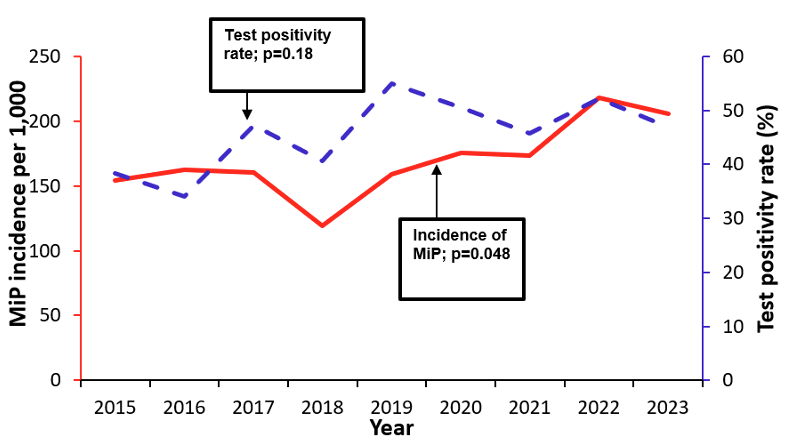
A total of 2,808,426 cases of MiP were recorded among 16,462,878 pregnant women who attended antenatal care. The average annual proportion of ANC1 attendees diagnosed with malaria during the study period was 170 cases per 1,000 pregnant women in Uganda. Malaria in pregnancy rose from 154 per 1,000 pregnant women in 2015 to 205 per 1,000 in 2023 (p=0.02), while the malaria test positivity rate did not change significantly during the study period (p=0.18). The lowest in MiP incidence was reported in 2018 as 119 per 1,000 pregnant women, while 2022 recorded the highest MiP incidence with 218 per 1,000 pregnant women (Figure 2). The Mann-Kendall test indicated a significant upward trend in MiP incidence between 2015–2023 (p=0.048), with a Sen’s slope of 6.7 cases per year (95%CI: 1.3–17).
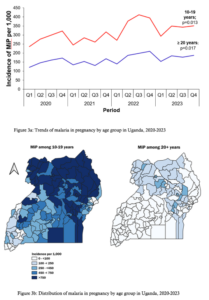
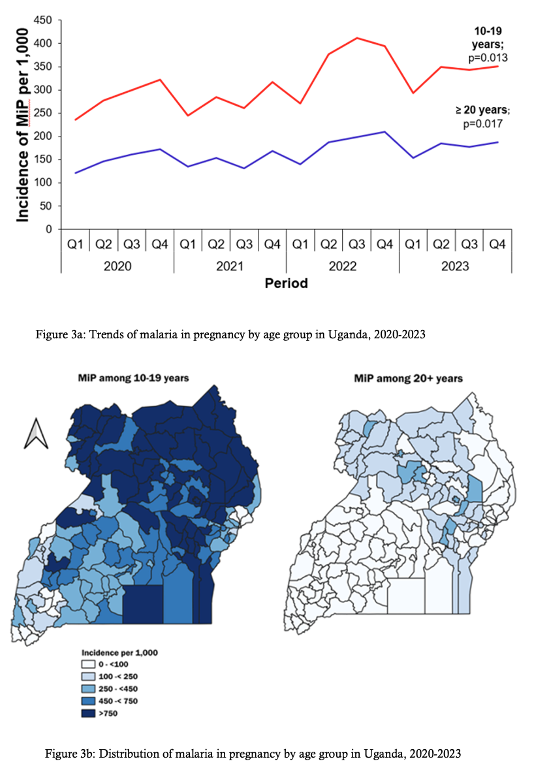
Age group-specific trends and distribution of malaria in pregnancy in Uganda, 2020–2023
Pregnant women in both age groups showed rising trends in MiP. Among women aged 10-19, MiP rose from 236 per 1,000 in the first quarter of 2020 to 351 per 1,000 (p=0.013) in the final quarter of 2023. Similarly, among women aged ≥20 years, MiP rose from 121 per 1,000 in the first quarter of 2020 to 187 per 1,000 (p=0.017) in the final quarter of 2023 (Figure 3a). Even though the MiP was rising across the two age categories, adolescent girls were the most affected. The geographical distribution of MiP showed that MiP was higher among adolescent girls, as indicated in Figure 3b. On the left is a map of MiP among women aged 10-19, and on the right is a map showing the MiP among mothers ≥20 years, with the darker colour indicating a higher MiP. More than half of the country reported high MiP among the adolescent girls (Figure 3b).
Regional trends and trends in malaria in pregnancy admissions and deaths, 2015–2023
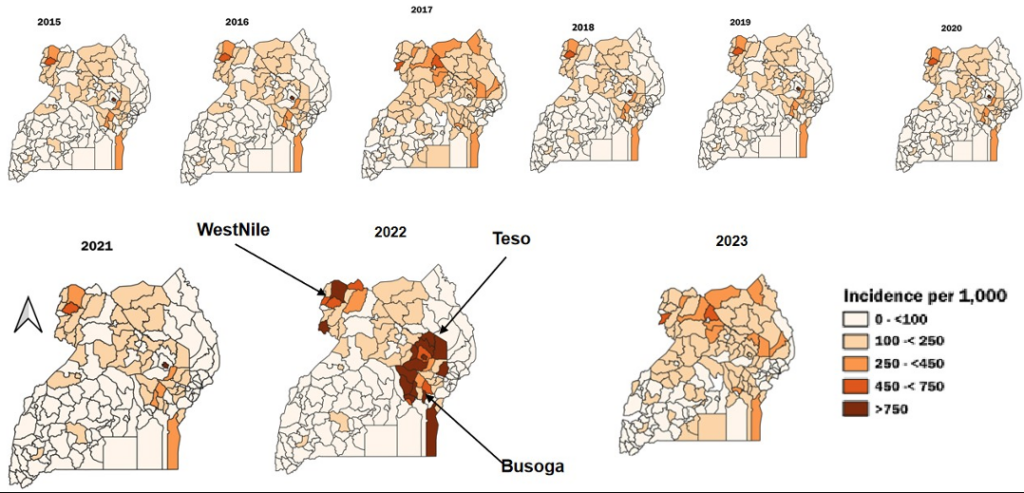
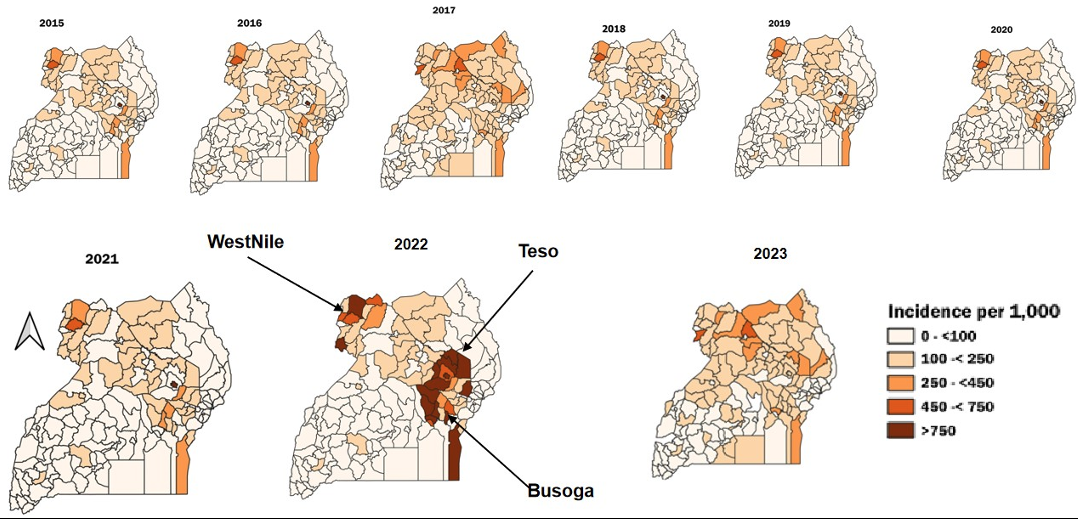
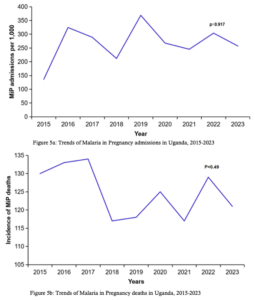
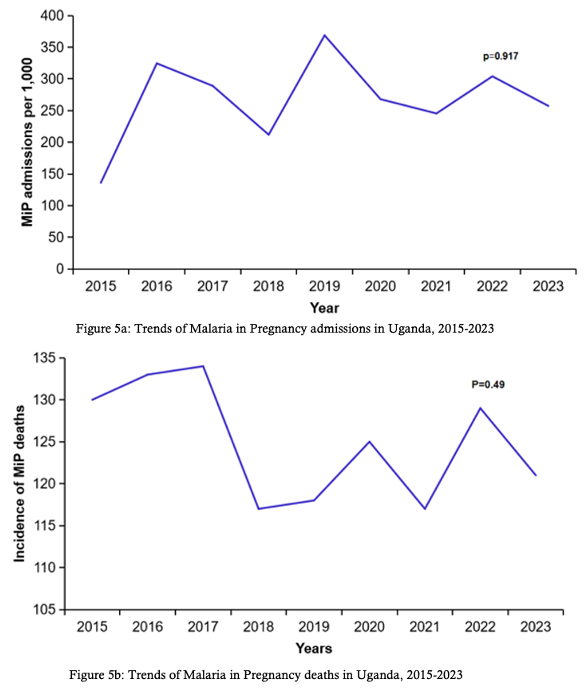
Significant increases in MiP were observed in Bunyoro with approximately 10 cases per year (95% CI: 4–21), Busoga (27 cases per year, 95%CI: 18–43), Teso (15 cases per year, 95%CI: 4.7–34), and West Nile (15 cases per year, 95%CI: 6.5–25) (Table 1, Figure 4). Throughout the study period, MiP-related admissions and deaths fluctuated. The highest MiP-associated admissions were reported in 2019 (369 per 1,000), and the lowest were recorded in 2015 (136 per 1,000). Similarly, the highest MiP-related deaths were recorded in 2019 (10 per 1,000), and the lowest occurred in 2023 (6 per 1,000). However, the trends of MiP-related admissions (p=0.92) (Figure 5a) and deaths (p=0.49) (Figure 5b) were not statistically significant.
Discussion
This study revealed an overall increase in the MiP from 2015 to 2023, with adolescent girls aged 10–19 being more affected than those aged ≥20 years. Our study also revealed regional differences in MiP trends, with the Bunyoro, Busoga, Teso, and West Nile regions reporting increases. However, hospital admissions and mortality due to MiP remained stable throughout the study period.
The proportion of ANC attendees diagnosed with MiP in our study showed an upward trend, which was consistent with findings from a similar study conducted in neighboring Tanzania, highlighting regional patterns in East Africa [25]. According to the Uganda Malaria Indicator Survey (UMIS 2018), 65% of pregnant women slept under ITN and only 41% received IPTp3, which was below the recommended coverage of 85%, highlighting a lower level of protection for pregnant women against malaria [20, 26]. Furthermore, the observed increase in MiP could partly reflect expanded malaria testing at health facilities rather than a true rise in incidence—following the 2019 Ministry of Health guidelines mandating malaria testing for all pregnant women at their first antenatal visit (ANC1) [15]. This interpretation is supported by the fact that the test positivity rate did not change significantly during the study period. Additionally, the observed trend could reflect improvements in surveillance and reporting rates of MiP during the study period. Furthermore, it’s also important to note that MiP peaked in 2022, coinciding with a nationwide rebound malaria epidemic in Uganda following disruptions associated with the COVID-19 pandemic in 2020–2021 [19]. This surge highlights the need to strengthen malaria prevention and treatment programs for pregnant women, particularly during periods of health system disruptions or epidemics.
Both age groups of pregnant women exhibited an increasing trend in MiP, with adolescents being most affected. Our findings are consistent with other studies, which indicated that adolescents were most vulnerable to malaria infections [27, 28, 29]. Furthermore, research indicates that adolescent mothers often have lower access to health information and services, making them a critical demographic for prevention intervention [30]. These findings highlight the need to strengthen malaria prevention efforts across all age groups of pregnant women.
Our findings noted regional disparities in MiP, with some regions, such as Bunyoro, Busoga, Teso, and West Nile, showing significant increases. Regional disparities could be explained by differences in malaria transmission intensity, intervention coverage, and contextual factors. High malaria burden regions of Teso and West Nile showed sustained increases in MiP consistent with high transmission malaria intensity in the region [31, 32, 33]. Moderate transmission regions, such as Busoga, also had increasing MiP, while the Bunyoro region, classified as a low transmission region, showed an increase in MiP, suggesting factors beyond overall transmission intensity. Regional variations observed in MiP incidence and outcomes may also be influenced by broader contextual factors, such as climate variability, particularly rainfall and temperature, that have been shown to affect malaria transmission intensity in Uganda [34, 35, 36, 37]. Additionally, coverage and timing of vector control interventions such as LLIN distribution and IRS implementation differ across regions, which could also explain regional malaria incidence variations [38]. Furthermore, variations in health-seeking behavior and access to antenatal care may contribute to differences in reported MiP cases. Our findings suggest that both high and low transmission settings remain at risk of increasing MiP, emphasizing the need for adaptive and region-specific malaria control strategies to ensure targeted actions in areas experiencing rising MiP burden.
Despite the observed increase in MiP in our study, the absence of a corresponding rise in admissions or deaths could be attributed to several factors. First, improved case management likely played a role in preventing a corresponding increase in severity and mortality. Evidence from Uganda indicates that interventions such as enhanced provider training, adherence to national malaria treatment guidelines, and community-based delivery of preventive and therapeutic services have contributed to better health outcomes among pregnant women [39, 40, 41]. Secondly, gaps in mortality reporting could also be a plausible explanation for non-significant malaria deaths. In Uganda, a substantial proportion of deaths occur outside health facilities and are often not documented, leading to underestimation of malaria mortality [42, 43]. Sustaining malaria case management alongside expanded antenatal care coverage and other malaria in pregnancy preventive interventions could further reduce malaria morbidity and mortality.
Study limitations
Some healthcare facilities providing MiP services, especially privately owned ones, may either not be registered to report through DHIS2 or fail to report promptly, potentially leading to an underestimation of the true MiP burden [44]. Although ANC1 attendance is nearly universal in Uganda (approximately 98-99%), our study did not examine potential systematic differences between women who attended ANC and those who did not. Women who do not attend ANC could differ systematically from attendees in age, rural versus urban residence, socioeconomic status, or access to health services. This study may be subject to selection bias due to the exclusion of health facilities that did not meet the predefined reporting completeness threshold of at least 80% of monthly reports between 2015 and 2023. This may limit the representativeness of the findings, particularly for under-reporting facilities.
Conclusion
There was an overall increase in the incidence of malaria in pregnancy from 2015 to 2023. Malaria in pregnancy increased among both adolescents and older women, with the highest incidence among adolescents. Additionally, regional variations were observed, with an increase in the Bunyoro, Teso, Busoga, and West Nile regions. Despite this increase, MiP hospital admissions and deaths did not change significantly, suggesting that effective case management may have played a crucial role in mitigating severe outcomes. These findings highlight the need for region-specific and age-targeted interventions, including prioritizing community-level interventions for adolescents and enhancing regional surveillance of malaria in pregnancy (MiP). Further studies could explore the underlying factors contributing to the vulnerability of adolescents to MiP and investigate the drivers of regional disparities.
What is already known about the topic
- Malaria in Pregnancy is a risk to maternal and fetal health, which could result in death
- Uganda has implemented MiP prevention strategies as recommended by the World Health Organization, including intermittent preventive treatment during pregnancy (IPTp), use of insecticide-treated nets (ITNs), and routine malaria screening during antenatal visits
- Despite these interventions, malaria upsurges in Uganda during the study period raise concerns about the continued burden of MiP and the effectiveness of the prevention strategies.
What this study adds
- The study provides trends in MiP, admissions, and fatalities across Uganda from 2015–2023
- Incidence of MiP increased from 2015 to 2023, with higher rates among adolescents (10-19 years) compared to older women (≥20 years)
- Incidence of MiP increased in the regions of Bunyoro, Busoga, Teso, and West Nile
- Despite the increase in MiP, MiP-related admissions and deaths did not change significantly
- Our findings highlight the need for targeted adolescent-focused and region-specific MiP interventions
Funding
The project was supported by the President’s Emergency Plan for AIDS Relief (PEPFAR) through the United States Centers for Disease Control and Prevention Cooperative Agreement number GH001353-01 through Makerere University School of Public Health to the Uganda Public Health Fellowship Program, Ministry of Health. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of the US Centers for Disease Control and Prevention, the Department of Health and Human Services, Makerere University School of Public Health, or the Uganda Ministry of Health. The funders had no role in the study design, data collection, data analysis, decision to publish or preparation of the manuscript
Authors´ contributions
CM: Participated in the conception, design, analysis, and interpretation of the study and wrote the draft manuscript; RM reviewed the drafts of the manuscript for intellectual content and made multiple edits to the draft manuscript; RM, PK, HTN, JR, GR, and ARA reviewed the manuscript to ensure intellectual content and scientific integrity. All authors read and approved of the final manuscript.
Availability of data and materials
The datasets upon which our findings are based belong to the Uganda Public Health Fellowship Program. For confidentiality reasons, the datasets are not publicly available. However, the datasets can be made available upon reasonable request from the corresponding author with permission from the Uganda Public Health Fellowship Program.
| Table 1: Regional trends of malaria in pregnancy in Uganda, 2015–2023 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Region | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | 2023 | Sens slope | (95% CI) |
| Acholi | 276 | 335 | 307 | 216 | 393 | 390 | 296 | 342 | 301 | 2.8 | (-23–27) |
| Ankole | 92 | 106 | 95 | 39 | 36 | 46 | 42 | 54 | 52 | -5.2 | (-13–3.5) |
| Bugisu | 126 | 109 | 125 | 73 | 99 | 88 | 112 | 140 | 122 | 1.3 | (-7.6–11) |
| Bukedi | 162 | 132 | 119 | 70 | 68 | 81 | 180 | 369 | 300 | 14 | (-21–56) |
| Bunyoro | 112 | 131 | 160 | 96 | 152 | 165 | 160 | 220 | 185 | 9.6 | (4–21) |
| Busoga | 183 | 213 | 226 | 184 | 230 | 281 | 321 | 367 | 396 | 27 | (18–43) |
| Kampala | 65 | 45 | 38 | 41 | 43 | 48 | 40 | 44 | 44 | -0.15 | (-3–1.2) |
| Karamoja | 231 | 185 | 166 | 143 | 131 | 166 | 161 | 160 | 149 | -5.1 | (-17–5) |
| Kigezi | 47 | 50 | 35 | 16 | 27 | 28 | 27 | 34 | 28 | -1.9 | (-5–2.3) |
| Lango | 248 | 283 | 232 | 158 | 285 | 289 | 249 | 378 | 355 | 15 | (-6.8–31) |
| North Central | 124 | 129 | 141 | 91 | 135 | 143 | 127 | 166 | 139 | 3.1 | (-1.3–9.6) |
| SouthCentral | 108 | 108 | 110 | 70 | 75 | 87 | 62 | 89 | 87 | -2.9 | (-8.3–3) |
| Teso | 299 | 314 | 311 | 251 | 278 | 325 | 354 | 399 | 419 | 15 | (4.7–34) |
| Tooro | 106 | 126 | 116 | 72 | 92 | 117 | 91 | 112 | 103 | -1.5 | (-7–6.2) |
| West Nile | 208 | 223 | 237 | 236 | 315 | 321 | 321 | 345 | 304 | 15 | (6.5–25) |




References
- World Health Organization. WHO policy brief for the implementation of intermittent preventive treatment of malaria in pregnancy using sulfadoxine-pyrimethamine (IPTp-SP) [Internet]. Geneva (Switzerland): WHO; 2013 Apr [cited 2026 May 05]; 13 p. Ref no.: WHO/HTM/GMP/2014.4. Available from: https://iris.who.int/server/api/core/bitstreams/1cc5110f-4ca9-47e3-80b1-467e598311f1/content
- Bedu-Addo G, Alicke M, Boakye-Appiah JK, Abdul-Jalil I, Van Der Giet M, Schulze MB, Mockenhaupt FP, Danquah I. In utero exposure to malaria is associated with metabolic traits in adolescence: The Agogo 2000 birth cohort study. Journal of Infection [Internet]. 2017 Oct [cited 2026 May 05];75(5):455–63. doi:10.1016/j.jinf.2017.08.010. Available from: https://www.sciencedirect.com/science/article/pii/S0163445317302670
- Bauserman M, Conroy AL, North K, Patterson J, Bose C, Meshnick S. An overview of malaria in pregnancy. Seminars in Perinatology [Internet]. 2019 Aug [cited 2026 May 05];43(5):282–90. doi:10.1053/j.semperi.2019.03.018. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0146000519300436?via%3Dihub
- Saito M, Briand V, Min AM, McGready R. Deleterious effects of malaria in pregnancy on the developing fetus: a review on prevention and treatment with antimalarial drugs. The Lancet Child & Adolescent Health [Internet]. 2020 Oct [cited 2026 May 05];4(10):761–74. doi:10.1016/S2352-4642(20)30099-7. Available from: https://www.sciencedirect.com/science/article/abs/pii/S2352464220300997
- Omer SA, Idress HE, Adam I, Abdelrahim M, Noureldein AN, Abdelrazig AM, Elhassan MO, Sulaiman SM. Placental malaria and its effect on pregnancy outcomes in Sudanese women from Blue Nile State. Malar J [Internet]. 2017 Sep 16 [cited 2026 May 05];16(1):374. doi:10.1186/s12936-017-2028-0. Available from: https://link.springer.com/article/10.1186/s12936-017-2028-0
- Mlugu EM. Optimization of intermittent preventive therapy for malaria during pregnancy: effectiveness of dihydroartemisinin-piperaquine versus sulfadoxine-pyrimethamine [thesis] [Internet]. Karolinska Institutet; 2021 [cited 2026 May 05]. Available from: https://openarchive.ki.se/articles/thesis/Optimization_of_intermittent_preventive_therapy_for_malaria_during_pregnancy_effectiveness_of_dihydroartemisinin-piperaquine_versus_sulfadoxine-pyrimethamine/26917651/1
- Heng S, O’Meara WP, Simmons RA, Small DS. Relationship between changing malaria burden and low birth weight in sub-Saharan Africa: A difference-in-differences study via a pair-of-pairs approach. eLife [Internet]. 2021 Jul 14 [cited 2026 May 05];10:e65133. doi:10.7554/elife.65133. Available from: https://elifesciences.org/articles/65133
- World Health Organization (WHO). World malaria report 2023 [Internet]. Geneva (Switzerland): WHO; 2023 Nov 30 [cited 2026 May 05]. Available from: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2023
- Barber BE, Rajahram GS, Grigg MJ, William T, Anstey NM. World Malaria Report: time to acknowledge Plasmodium knowlesi malaria. Malar J [Internet]. 2017 Mar 31 [cited 2026 May 05];16(1):135. doi:10.1186/s12936-017-1787-y. Available from: https://link.springer.com/article/10.1186/s12936-017-1787-y
- Namusoke F, Rasti N, Kironde F, Wahlgren M, Mirembe F. Malaria burden in pregnancy at Mulago National Referral Hospital in Kampala, Uganda. Malaria Research and Treatment [Internet]. 2010 Nov 7 [cited 2026 May 05];2010(1). doi:10.4061/2010/913857. Available from: https://onlinelibrary.wiley.com/doi/full/10.4061/2010/913857
- Mangusho C, Mwebesa E, Izudi J, Aleni M, Dricile R, Ayiasi RM, Legason ID. High prevalence of malaria in pregnancy among women attending antenatal care at a large referral hospital in northwestern Uganda: A cross-sectional study. PLoS ONE [Internet]. 2023 Apr 5 [cited 2026 May 05];18(4):e0283755. doi:10.1371/journal.pone.0283755. Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0283755
- Ssempiira J, Nambuusi B, Kissa J, Agaba B, Makumbi F, Kasasa S, Vounatsou P. Geostatistical modelling of malaria indicator survey data to assess the effects of interventions on the geographical distribution of malaria prevalence in children less than 5 years in Uganda. PLoS ONE [Internet]. 2017 Apr 4 [cited 2026 May 05];12(4):e0174948. doi:10.1371/journal.pone.0174948. Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0174948
- World Health Organization (WHO). WHO guidelines for malaria [Internet]. Geneva (Switzerland): WHO; 2022 Feb 18 [cited 2026 May 05]; 240 p. Available from: https://apps.who.int/iris/bitstream/handle/10665/351995/WHO-UCN-GMP-2022.01-eng.pdf
- Yeka A, Gasasira A, Mpimbaza A, Achan J, Nankabirwa J, Nsobya S, Staedke SG, Donnelly MJ, Wabwire-Mangen F, Talisuna A, Dorsey G, Kamya MR, Rosenthal PJ. Malaria in Uganda: challenges to control on the long road to elimination. Acta Tropica [Internet]. 2011 Mar 21 [cited 2026 May 05];121(3):184–95. doi:10.1016/j.actatropica.2011.03.004. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC3156969/
- Ministry of Health (MoH) Uganda. Integrated management of malaria training health worker’s manual [Internet]. Kampala (Uganda): MoH; 2012 Mar [cited 2026 May 05]; 129 p. Available from: https://library.health.go.ug/communicable-disease/malaria/integrated-management-malaria-training-health-workers-manual
- The World Bank, Uganda. Uganda malaria indicator survey 2018–2019 [Internet]. Kampala (Uganda): MOH; 2021 May 19 [cited 2026 May 05]; [about 14 screens]. Ref no.: WBG_UGA_2018_MIS_v01_M. Available from: https://microdata.unhcr.org/index.php/catalog/428
- Uganda Bureau of Statistics (UBoS). Uganda demographic and health survey 2022 main report [Internet]. Kampala (Uganda): UBoS; 2023 Nov [cited 2026 May 05]. Available from: https://www.ubos.org/wp-content/uploads/publications/UDHS-2022-Report.pdf
- Wafula ST, Mendoza H, Nalugya A, Musoke D, Waiswa P. Determinants of uptake of malaria preventive interventions among pregnant women in eastern Uganda. Malar J [Internet]. 2021 Jan 3 [cited 2026 May 05];20(1):5. doi:10.1186/s12936-020-03558-1. Available from: https://link.springer.com/article/10.1186/s12936-020-03558-1
- Epstein A, Maiteki-Sebuguzi C, Namuganga JF, Nankabirwa JI, Gonahasa S, Opigo J, Staedke SG, Rutazaana D, Arinaitwe E, Kamya MR, Bhatt S, Rodríguez-Barraquer I, Greenhouse B, Donnelly MJ, Dorsey G. Resurgence of malaria in Uganda despite sustained indoor residual spraying and repeated long lasting insecticidal net distributions. PLOS Glob Public Health [Internet]. 2022 Sep 7 [cited 2026 May 05];2(9):e0000676. doi:10.1371/journal.pgph.0000676. Available from: https://journals.plos.org/globalpublichealth/article?id=10.1371/journal.pgph.0000676
- Ministry of Health (MoH) Uganda. Uganda malaria reduction strategic plan 2014–2020 [Internet]. Kampala (Uganda): Ministry of Health, Uganda; 2014 May [cited 2026 May 05]. Available from: https://www.severemalaria.org/sites/default/files/content/document/The%20Uganda%20Malaria%20Reduction%20Strategic%20Plan%202014-2020%20%283%29.pdf
- Chen J, Ssennyonjo A, Wabwire-Mangen F, Kim JH, Bell G, Hirschhorn L. Does decentralization of health systems translate into decentralization of authority? A decision space analysis of Ugandan healthcare facilities. Health Policy and Planning [Internet]. 2021 Jun 24 [cited 2026 May 05];36(9):1408–17. doi:10.1093/heapol/czab074. Available from: https://academic.oup.com/heapol/article/36/9/1408/6308752
- Munabi-Babigumira S, Nabudere H, Asiimwe D, Fretheim A, Sandberg K. Implementing the skilled birth attendance strategy in Uganda: a policy analysis. BMC Health Serv Res [Internet]. 2019 Sep 10 [cited 2026 May 05];19(1):655. doi:10.1186/s12913-019-4503-5. Available from: https://link.springer.com/article/10.1186/s12913-019-4503-5
- Ministry of Health (MoH) Uganda. EMR system implementation guidelines [Internet]. Kampala (Uganda): MoH Uganda; 2024 Jan [cited 2026 May 05]. Available from: https://library.health.go.ug/health-information-systems/digital-health/guidelines-implementation-electronic-medical-records
- Sen PK. Estimates of the regression coefficient based on Kendall’s Tau. Journal of the American Statistical Association [Internet]. 1968 Dec [cited 2026 May 05];63(324):1379–89. doi:10.2307/2285891. Available from: https://www.tandfonline.com/doi/abs/10.1080/01621459.1968.10480934
- Fried M, Duffy PE. Malaria during pregnancy. Cold Spring Harb Perspect Med [Internet]. 2017 Feb 17 [cited 2026 May 05];7(6):a025551. doi:10.1101/cshperspect.a025551. Available from: https://perspectivesinmedicine.cshlp.org/content/7/6/a025551
- Uganda Bureau of Statistics (UBOS), ICF International. Uganda malaria indicator survey report; 2018–19 [Internet]. Kampala (Uganda), Rockville (MA): UBoS and ICF International; 2020 Mar [cited 2026 May 05]; 163 p. Available from: https://microdata.unhcr.org/index.php/catalog/428
- Orish VN, Onyeabor OS, Boampong JN, Aforakwah R, Nwaefuna E, Iriemenam NC. Adolescent pregnancy and the risk of Plasmodium falciparum malaria and anaemia—a pilot study from Sekondi-Takoradi metropolis, Ghana. Acta Tropica [Internet]. 2012 Jun [cited 2026 May 05];123(3):244–8. doi:10.1016/j.actatropica.2012.05.014. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0001706X12002240?via%3Dihub
- Mombo-Ngoma G, Mackanga JR, González R, Ouedraogo S, Kakolwa MA, Manego RZ, Basra A, Rupérez M, Cot M, Kabanywany AM, Matsiegui PB, Agnandji ST, Vala A, Massougbodji A, Abdulla S, Adegnika AA, Sevene E, Macete E, Yazdanbakhsh M, Kremsner PG, Aponte JJ, Menéndez C, Ramharter M. Young adolescent girls are at high risk for adverse pregnancy outcomes in sub-Saharan Africa: an observational multicountry study. BMJ Open [Internet]. 2016 Jun 29 [cited 2026 May 05];6(6):e011783. doi:10.1136/bmjopen-2016-011783. Available from: https://bmjopen.bmj.com/content/6/6/e011783
- Pons-Duran C, Mombo-Ngoma G, Macete E, Desai M, Kakolwa MA, Zoleko-Manego R, Ouédragou S, Briand V, Valá A, Kabanywanyi AM, Ouma P, Massougbodji A, Sevene E, Cot M, Aponte JJ, Mayor A, Slutsker L, Ramharter M, Menéndez C, González R. Burden of malaria in pregnancy among adolescent girls compared to adult women in 5 sub-Saharan African countries: A secondary individual participant data meta-analysis of 2 clinical trials. PLoS Med [Internet]. 2022 Sep 2 [cited 2026 May 05];19(9):e1004084. doi:10.1371/journal.pmed.1004084. Available from: https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1004084
- Mangeli M, Rayyani M, Cheraghi MA, Tirgari B. Exploring the challenges of adolescent mothers from their life experiences in the transition to motherhood: a qualitative study. J Family Reprod Health [Internet]. 2017 Sep [cited 2026 May 05];11(3):165–73. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC6045691/
- Zalwango MG, Bulage L, Zalwango JF, Migisha R, Agaba BB, Kadobera D, Kwesiga B, Opigo J, Ario AR. Trends and distribution of severe malaria cases, Uganda, 2017–2021: analysis of health management information system data. Uganda National Institute of Public Health Quarterly Epidemiological Bulletin [Internet]. 2023 Apr–Jun [cited 2026 May 05];8(2):2. Available from: https://uniph.go.ug/trends-and-distribution-of-severe-malaria-cases-uganda-2017-2021-analysis-of-health-management-information-system-data/
- Wasswa R, Kananura RM, Muhanguzi H, Waiswa P. Spatial variation and attributable risk factors of anaemia among young children in Uganda: evidence from a nationally representative survey. PLOS Glob Public Health [Internet]. 2023 May 17 [cited 2026 May 05];3(5):e0001899. doi:10.1371/journal.pgph.0001899. Available from: https://journals.plos.org/globalpublichealth/article?id=10.1371/journal.pgph.0001899
- Odokonyero T, Ahaibwe G, Ssengooba F. Financing indoor residual spraying for malaria prevention in Uganda: options for cost minimization [Internet]. Kampala (Uganda): Economic Policy Research Centre; 2019 Mar [cited 2026 May 05]; 38 p. Available from: https://eprcug.org/publication/financing-indoor-residual-spraying-for-malaria-prevention-in-uganda-options-for-cost-minimization/
- Mwaura MF. Regional variations in climate and malaria transmission in Uganda. EEJBS [Internet]. 2024 [cited 2026 May 05];5(3):50–54. Available from: https://smartie.kiu.ac.ug/public/assets/publications/754a5b0722dfea91e5b23dc3429ea04362ce2b27.pdf
- Okiring J, Routledge I, Epstein A, Namuganga JF, Kamya EV, Obeng-Amoako GO, Sebuguzi CM, Rutazaana D, Kalyango JN, Kamya MR, Dorsey G, Wesonga R, Kiwuwa SM, Nankabirwa JI. Associations between environmental covariates and temporal changes in malaria incidence in high transmission settings of Uganda: a distributed lag nonlinear analysis. BMC Public Health [Internet]. 2021 Oct 30 [cited 2026 May 05];21(1):1962. doi:10.1186/s12889-021-11949-5. Available from: https://link.springer.com/article/10.1186/s12889-021-11949-5
- Beloconi A, Nyawanda BO, Bigogo G, Khagayi S, Obor D, Danquah I, Kariuki S, Munga S, Vounatsou P. Malaria, climate variability, and interventions: modelling transmission dynamics. Sci Rep [Internet]. 2023 May 5 [cited 2026 May 05];13(1):7367. doi:10.1038/s41598-023-33868-8. Available from: https://www.nature.com/articles/s41598-023-33868-8
- Kiragga A, Otieno F, Nabukeera B, Ng’etich M, Tozan Y, Kajungu D. Climate-driven malaria mortality among children in malaria-endemic areas of Uganda. BMC Public Health [Internet]. 2025 Aug 18 [cited 2026 May 05];25(1):2825. doi:10.1186/s12889-025-23678-0. Available from: https://link.springer.com/article/10.1186/s12889-025-23678-0
- Awine T, Silal SP. Accounting for regional transmission variability and the impact of malaria control interventions in Ghana: a population level mathematical modelling approach. Malar J [Internet]. 2020 Nov 23 [cited 2026 May 05];19(1):423. doi:10.1186/s12936-020-03496-y. Available from: https://link.springer.com/article/10.1186/s12936-020-03496-y
- Wafula ST, Kananura RM, Pande G, Kizito F, Namutamba S, Kyobe B, Agiraembabazi G, Ekirapa-Kiracho E, Waiswa P. Effect of community–facility linked interventions on maternal health service utilization and newborn care in rural low-resource settings in Eastern Uganda. BMC Pregnancy Childbirth [Internet]. 2024 Oct 22 [cited 2026 May 05];24(1):692. doi:10.1186/s12884-024-06883-4. Available from: https://link.springer.com/article/10.1186/s12884-024-06883-4
- Koita K, Kayentao K, Worrall E, Van Eijk AM, Hill J. Community-based strategies to increase coverage of intermittent preventive treatment of malaria in pregnancy with sulfadoxine–pyrimethamine in sub-Saharan Africa: a systematic review, meta-analysis, meta-ethnography, and economic assessment. The Lancet Global Health [Internet]. 2024 Sep [cited 2026 May 05];12(9):e1456–69. doi:10.1016/S2214-109X(24)00228-6. Available from: https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(24)00228-6/fulltext
- Faculty of Clinical Medicine and Dentistry Kampala International University, Kanobere H. Enhancing IPTp program implementation: provider practices, challenges, and malaria prevalence among pregnant women in Uganda. IDOSR-JST [Internet]. 2024 Mar 13 [cited 2026 May 05];10(1):43–50. doi:10.59298/IDOSR/JST/24/101.234350. Available from: https://www.idosr.org/wp-content/uploads/2024/04/IDOSR-JST-10143-50-2024.pdf
- Zalwango MG, Migisha R, Kwesiga B, Bulage L, Kadobera D, Ario AR. Using a quality improvement approach to improve reporting of malaria deaths in Namutumba District, Eastern Uganda, 2022–2023 [Internet]. 2024 May 21 [cited 2026 May 05]. doi:10.1101/2024.05.21.24307657. Available from: http://medrxiv.org/lookup/doi/10.1101/2024.05.21.24307657
- Zalwango MG, Kyozira C, Nambuya M, Bulamu M, Muruta A. A rapid assessment of mortality surveillance in Uganda. PLOS Glob Public Health [Internet]. 2025 Jul 15 [cited 2026 May 05];5(7):e0003722. doi:10.1371/journal.pgph.0003722. Available from: https://journals.plos.org/globalpublichealth/article?id=10.1371/journal.pgph.0003722
- Birabwa C, Banke-Thomas A, Semaan A, Van Olmen J, Kananura RM, Arinaitwe ES, Waiswa P, Beňová L. The quality of routine data for measuring facility-based maternal mortality in public and private health facilities in Kampala City, Uganda. Popul Health Metrics [Internet]. 2024 Aug 23 [cited 2026 May 05];22(1):22. doi:10.1186/s12963-024-00343-z. Available from: https://link.springer.com/article/10.1186/s12963-024-00343-z
