Research | Open Access | Volume 9 (2): Article 68 | Published: 28 Apr 2026
Evaluation of the national HIV surveillance system for exposed children in Togo, 2021
Menu, Tables and Figures
Navigate this article
Tables
| Indicators | Calculation / Data Input | Indicator value | Data source | Collection techniques | Evaluation criteria |
|---|---|---|---|---|---|
| Utility | |||||
| Proportion of surveillance stakeholders who find the HIV surveillance system useful | Number of stakeholders who answered yes / total surveyed | 64/64 (100%; 95% CI: 94.4–100) | Stakeholders involved | Data extraction | ≥80%, Good performance |
| Proportion of newborns of HIV+ mothers who received ARV prophylaxis | Number receiving ARV / expected newborns | 81.5% | PNLS-HV-IST 2020 report | Annual report | ≥80%, Good performance |
| Proportion of HIV-positive pregnant women who initiated ART | HIV+ pregnant women on ART / total HIV+ pregnant women | 3627/3759 (96.5%) | ≥80%, Good performance | ||
| Utility performance | 92.6% | ≥80%, Good performance | |||
| Simplicity | |||||
| Clarity of case definition | Officers answering yes / total | 58/64 (90.6%) | Stakeholders | Interview | ≥80%, Good performance |
| Ease of sending samples | Agents answering yes / total | 56/64 (87.5%) | Stakeholders | Interview | ≥80%, Good performance |
| Simplicity of notification form | Agents recognizing simplicity / total | 62/64 (96.9%) | Stakeholders | Interview | ≥80%, Good performance |
| Simplicity performance | 91.6% | ≥80%, Good performance | |||
| Acceptability | |||||
| Perception of number of variables | Respondents saying not excessive / total | 62/64 (96.9%) | Stakeholders | Interview | ≥80%, Good performance |
| Completeness of reporting | Reported sites / total sites | 768/768 (100%) | DHIS2 database | Data extraction | ≥80%, Good performance |
| Time spent completing form | Agents responding yes / total | 64/64 (100%) | Stakeholders | Interview | ≥80%, Good performance |
| Stakeholder commitment | Committed stakeholders / total | 64/64 (100%) | Stakeholders | Data extraction | ≥80%, Good performance |
| Acceptability performance | 99.2% | ≥80%, Good performance | |||
| Data Quality | |||||
| PCR variable completeness | Complete PCR values / total forms | 2059/2087 (98.6%) | DHIS2 forms | Data extraction | ≥80%, Good performance |
| Delivery mode completeness | Complete delivery values / total forms | 543/2087 (26.3%) | DHIS2 forms | <60%, Low performance | |
| Lab arrival date completeness | Complete values / total forms | 907/2087 (43.4%) | DHIS2 forms | Data extraction | <60%, Low performance |
| Date of birth completeness | Complete DOB values / total forms | 2077/2087 (99.5%) | DHIS2 forms | Data extraction | ≥80%, Good performance |
| Data quality performance | 66.9% | (60–79%), Moderate performance | |||
Table 1: Indicators of simplicity, acceptability and quality of data used to evaluate the HIV surveillance system for exposed children in Togo
| Occupation | Frequency | % |
|---|---|---|
| State-certified midwife | 29 | 42.7% |
| Medical Assistant | 26 | 38.2% |
| Doctor | 2 | 2.9% |
| Biolim Managers | 2 | 2.9% |
| Laboratory Technicians | 1 | 1.5% |
| Midwife assistant | 1 | 1.5% |
| PMTCT Manager | 1 | 1.5% |
| PNLS Monitoring and Evaluation Officer | 1 | 1.5% |
Table 2: Distribution of the respondents by occupation, Togo, 2021
Figures

Keywords
- HIV
- Surveillance system
- Early infant diagnosis
- PMTCT
- Togo
Labaguibe Gangak1, Fifonsi Gbeasor2, Rebecca Kinde2,3, Anoumou Dagnra4, Gmakouba Wankpaouyare5, Péléké Hilim6, Adodo Yao Sadji7
1National Programme for the Fight Against AIDS and STIs (PNLS-HV-IST), Lomé, Togo, 2Faculty of Health Sciences, University of Lomé, Lomé, Togo, 3FETP Togo, Centre for Training and Research in Public Health, Lomé, Togo, 4National Programme to Combat AIDS, Viral Hepatitis and STIs, Lomé, Togo, 5Maternal and Child Health Division, Lomé, Togo, 6Prefectural Directorate of Health, Tône, Togo, 7Laboratory Biosafety and Biosecurity, National Institute of Hygiene, Lomé, Togo
&Corresponding author: Labaguibe Gangak, National Programme for the Control of AIDS and STIs (PNLS-HV-IST), Lomé, Togo, Email: gangaklabguibe@gmail.com, ORCID: https://orcid.org/0009-0007-9479-7952
Received: 29 Oct 2025, Accepted: 24 Apr 2026, Published: 28 Apr 2026
Domain: HIV Epidemiology
Keywords: HIV, surveillance system, early infant diagnosis, PMTCT, Togo
©Labaguibe Gangak et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Labaguibe Gangak et al., Evaluation of the national HIV surveillance system for exposed children in Togo, 2021. Journal of Interventional Epidemiology and Public Health. 2026; 9(2):68. https://doi.org/10.37432/jieph-d-25-00259
Abstract
Introduction: In Togo, early infant diagnosis (EID) coverage remains low despite the expansion of PMTCT services. This study evaluated the national HIV surveillance system for exposed children.
Methods: A descriptive cross-sectional evaluation was conducted from September to October 2021 across all six health regions of Togo. The evaluation process followed the Centres for Disease Control and Prevention (CDC) guidelines for surveillance system evaluation. Data were collected from key stakeholders in HIV surveillance through interviews using a structured questionnaire, and the national database was analysed to assess system attributes, including utility, simplicity, acceptability, and data quality. Proportions were estimated with 95% confidence intervals (CI).
Results: A total of 68 HIV exposed children surveillance actors participated with a response rate of 87.2% (68/78). The surveillance system demonstrated good performance across key attributes based on predefined evaluation criteria: utility (92.6%; 95%CI: 94.4-100), simplicity (91.6%; 95% CI: 82.5-96.8), and acceptability (99.2%; 95% CI: 95.6-99.9). However, substantial underreporting was observed, as only 993 cases were reported out of 5,700 expected cases. Data quality was estimated at 66.9% (95% CI: 64.9-68.9). Training gaps and logistical constraints were identified as major barriers.
Conclusion: The HIV surveillance system among exposed children in Togo is operational, useful, and highly accepted, but suffers from underreporting and data quality challenges, which may hinder national targets.
Introduction
Human immunodeficiency virus (HIV) remains a major global public health concern, with 37.7 million people living with HIV in 2020, including 1.7 million children under 14 years [1]. In children, HIV transmission occurs mainly through vertical transmission during pregnancy, delivery, or breastfeeding [2,3]. Prevention of mother-to-child transmission (PMTCT) programs play a critical role in reducing this burden [4,5].
In Togo, the HIV prevalence declined from 2.5% in 2014 to 2.1% in 2019 [6], yet paediatric testing and treatment remain insufficient. Coverage of the first virological test dropped from 48.8% in 2019 to 17.4% in 2020 [7]. Despite the expansion of early infant diagnosis and PMTCT services in many sub-Saharan African countries, limited data are available on the performance of HIV surveillance systems supporting PMTCT objectives, particularly in low-resource settings. Evaluation of surveillance systems is essential to monitor HIV infection trends, guide programs, and address gaps. In Togo, no comprehensive evaluation of the surveillance system for HIV-exposed infants using standardised system attributes has yet been conducted.
This study fills this gap by providing the first systematic assessment of the HIV surveillance system for early diagnosis in infants in Togo, focusing on key attributes such as utility, simplicity, acceptability, data quality, and timeliness. The results provide context-specific data that can be used to strengthen the system and support national PMTCT surveillance and validation efforts.
Methods
Study design and setting
A descriptive cross-sectional study was conducted between September and October 2021 across regions of all six health regions of Togo. The study evaluated the national HIV surveillance system for exposed children, in accordance with the Centers for Disease Control and Prevention (CDC) framework for evaluation of public health surveillance systems.
Study population
The study population consisted of stakeholders involved in the coordination, supervision, monitoring, and reporting of early infant diagnosis (EID) HIV surveillance data at district, regional, and central levels. Participants were included if they were directly involved in EID surveillance activities at the facility, district, and national levels. Eligible participants had at least six months of experience in activities related to case notification, data recording, reporting, or data use within the surveillance system. Health workers who were temporarily assigned to services unrelated to HIV surveillance, those with less than six months of relevant experience, and individuals who were unavailable during the data collection period were excluded from the study.
Sampling
A purposive non-probability sampling method was used to select district PMTCT and HIV focal points and regional and central program managers based on their technical roles within the surveillance system. This method ensured the inclusion of respondents with sufficient knowledge of data flow, reporting procedures, and system performance.
Data collection
Interviews, document review, and national database extraction were used. Tools included questionnaires, data extraction forms, and interview guides. Primary data were collected through structured interviewer-administered questionnaires. Although some open-ended comments were documented, the study was not designed as a formal qualitative inquiry with verbatim quotations or thematic coding, while secondary data were obtained from national surveillance reports and the DHIS2 database. Surveillance stakeholders were asked to assess the availability, clarity, and ease of application of these case definitions, as well as the simplicity of completing notification forms and reporting procedures. These elements were measured using structured questionnaire items and contributed to the overall simplicity score of the surveillance system.
Variables and indicators
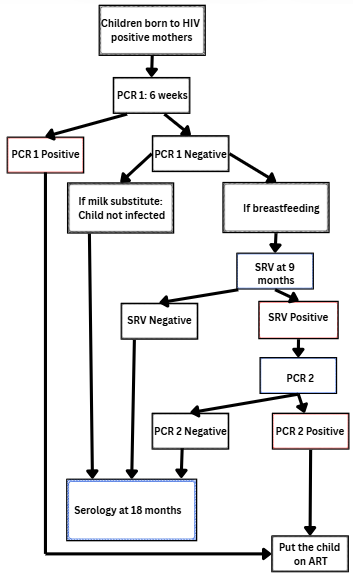
Attributes evaluated included utility, simplicity, acceptability, and data quality. The indicators used to assess each surveillance attribute, and their corresponding definitions, are summarised in Table 1. Each attribute was operationalised using predefined indicators derived from questionnaire items and database variables, with performance thresholds defined a priori (≥80% considered good performance), in accordance with the CDC updated guidelines for evaluating public health surveillance systems. Utility was assessed using both perceived usefulness (based on structured questionnaire responses) and objective PMTCT programmatic indicators, including ART coverage among HIV-positive pregnant women and ARV prophylaxis among HIV-exposed newborns. Simplicity and acceptability were assessed through respondents’ perceptions using standardized Likert -scale questionnaire items addressing clarity of case definitions, ease of completing notification forms, reporting procedures, and perceived workload. For each attribute, a composite score was calculated as the proportion of positive responses. An attribute was classified as “good” if the score was ≥80%, in line with standard surveillance system evaluation guidelines. Specifically, the following case definitions were applied in routine surveillance: A suspected case of HIV infection in an infant referred to any HIV-exposed infant presenting for early infant diagnosis testing, typically between six weeks and 18 months of age, in accordance with national guidelines. A confirmed HIV-infected infant was defined as an HIV-exposed infant with a positive virological test (PCR) result. The early infant diagnosis algorithm used in Togo is presented in Figure 1.
These standardised case definitions are used to guide notification, reporting, and laboratory testing procedures. Respondents were asked to assess the clarity, ease of understanding, and practical applicability of these definitions, as well as the associated notification tools, reporting procedures, and perceived workload, to evaluate the simplicity and acceptability of the surveillance system. Data quality was assessed using indicators of completeness and validity of key variables, including PCR type, age, breastfeeding status, and laboratory turnaround times.
Data analysis
Descriptive statistics were computed. Frequencies, proportions, and 95% confidence intervals were calculated. Descriptive statistical analyses were performed using Stata version 16.0. Categorical variables were summarised using frequencies and proportions, while continuous variables were summarised using medians and ranges. For key surveillance indicators and system attributes (utility, simplicity, acceptability, and data quality), proportions were calculated, and 95% confidence intervals (95% CIs) were estimated using the exact binomial method, which is appropriate for proportion estimates derived from surveillance data. Surveillance system performance was classified as low (<60%), moderate (60 -79%), or good (≥80%), in accordance with the Centres for Disease Control and Prevention (CDC) guidelines. Exact binomial confidence intervals were preferred over normal approximations due to the bounded nature of proportions and the sample size of some indicators
Ethical considerations
Ethical approval for this study was obtained from the Institutional Review Board (IRB) of the Centre for Public Health Training and Research (CFRSP) in Togo. As part of the Field Epidemiology Training Program (FETP), student protocols are routinely reviewed by the Institutional Review Board through an established institutional process. The protocol was examined and received approval from the committee; however, no formal reference number was issued. Administrative authorisation was granted by the National HIV Program. Participation was voluntary, written informed consent was obtained from all respondents, and confidentiality and anonymity were strictly ensured.
Results
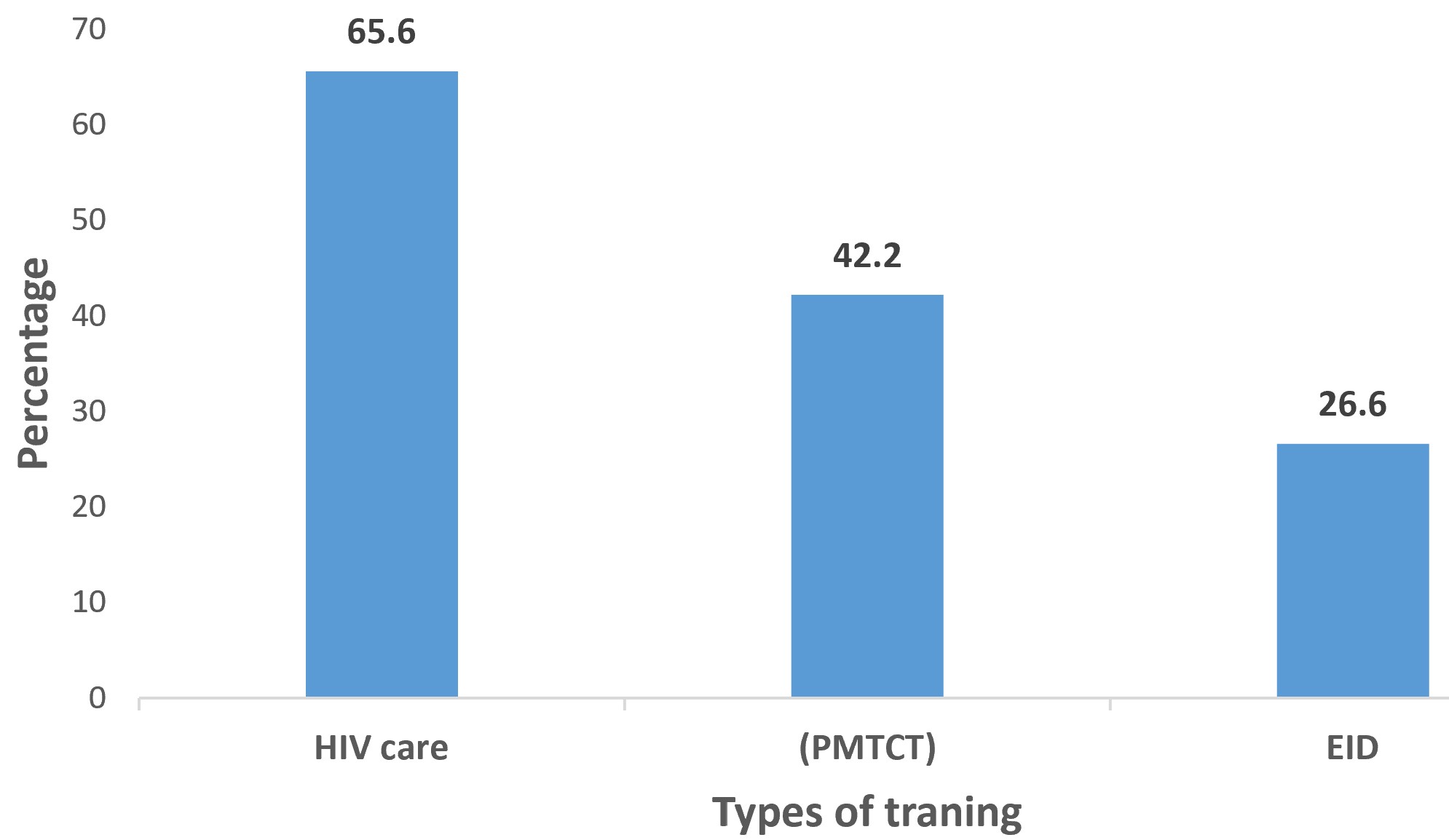
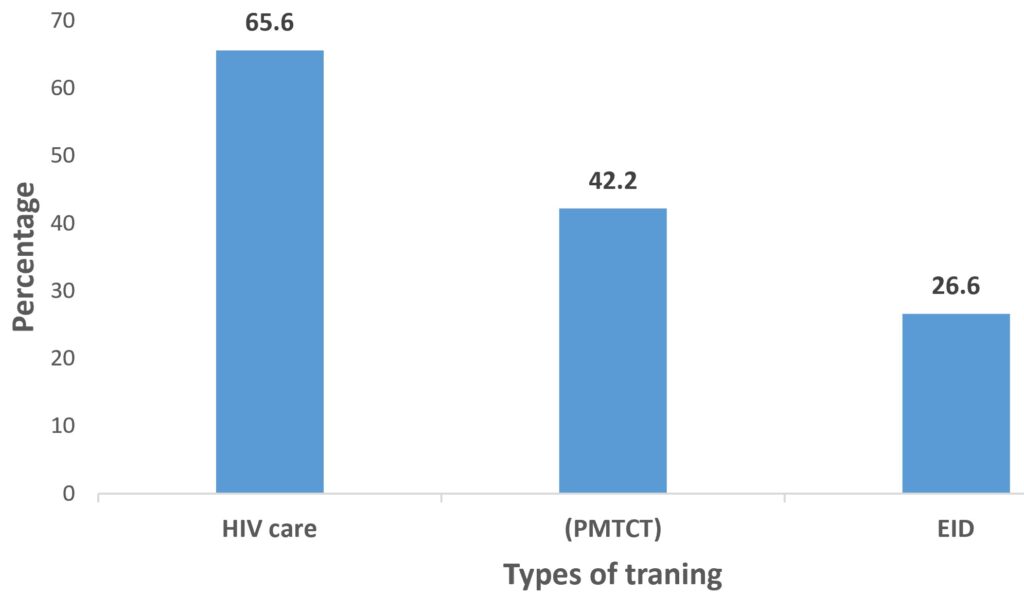
A total of 68 HIV infant surveillance actors completed the assessment, representing an overall response rate of 87.2% (68/78). The median age of respondents was 38 years (range: 32–53), with 42.6% (29/68) male and 57.4% (39/68) female. Respondents included key actors involved in the implementation and monitoring of the HIV children surveillance system at different levels of the health system. They comprised healthcare providers, data managers, and program officers engaged in surveillance activities. Nurses constituted 51.5% of participants, followed by physicians (27.9%) and laboratory technicians (20.6%). The distribution of respondents by occupation is presented in Table 2. Overall, 65% of respondents had more than five years of experience in HIV surveillance, but only 26.6% reported having received formal training on early infant diagnosis (EID). The distribution of respondents according to training received is shown in Figure 2.
The reporting chain was hierarchical and functional, but stock-outs of test kits and delays in sample transportation were reported as barriers. These findings were consistent with stakeholder comments describing limited refresher training, logistical constraints, and reporting challenges.
The surveillance system demonstrated good utility, with an overall utility score of 92.6%. All surveillance actors interviewed reported that the system was useful (100%; 95% CI: 94.4-100). Programmatic indicators further supported this finding, including a high proportion of HIV-positive pregnant women initiated on antiretroviral therapy (96.5%) and a substantial coverage of antiretroviral prophylaxis among newborns exposed to HIV (81.5%).
Simplicity of the surveillance system was rated as good (91.6%; 95% CI: 82.5-96.8), based on respondents’ perception of clarity of case definitions and ease of completing notification forms. No objective time-motion or process-based assessment of simplicity was conducted.
Acceptability was very high (99.2% 95% CI: 95.6-99.9), as nearly all stakeholders expressed willingness to continue participating in surveillance activities. Completeness reached 66.9% (95% CI: 64.9- 68.9), based on the review of 993 surveillance records. Demographic variables were largely complete (>80%), whereas key variables such as breastfeeding status (45%) and PCR turnaround times (38%) were poorly documented, indicating gaps in data quality.
Discussion
Overall, the findings indicate that the HIV surveillance system for exposed children in Togo demonstrates strong performance in utility, simplicity, and acceptability, but faces important challenges related to underreporting and data completeness. This is the first systematic evaluation of the HIV surveillance system for exposed children in Togo.
The strong utility observed is supported not only by positive perceptions of surveillance actors but also by objective programmatic indicators, including high antiretroviral therapy coverage among HIV-positive pregnant women and substantial prophylaxis coverage among HIV-exposed newborns. Despite these strengths, moderate data quality and marked underreporting limit the system’s capacity to accurately monitor early infant HIV infection.
The system benefits from strong health worker engagement, with 99.2% acceptability, similar to Zimbabwe [7]. Its simplicity score is consistent with Mozambique [8] and Yemen [9], where reporting tools were also user-friendly. The structured reporting chain is another strength.
The system had two limitations, including significant underreporting of cases and insufficient data quality in the reported records. Underreporting was evidenced by the significant discrepancy between expected and reported cases (993 reported cases out of 5,700 expected), indicating gaps in case notification and reporting coverage.
In contrast, data quality issues resulted in a low completeness rate for key variables in the reported records (e.g., breastfeeding status and time to PCR test results), with the overall completeness rate estimated at 66.9%. Data quality gaps, particularly regarding breastfeeding status and EID turnaround time, are consistent with deficiencies reported in similar surveillance systems in Kenya and Uganda [10, 11]. Training gaps were striking, with only 26.6% had EID training, compared to higher coverage in Botswana [12]. The low level of training among surveillance actors (26.6%) may contribute to both underreporting and incomplete data in reported records.
Severe underreporting threatens the ability of Togo to reach the 95-95-95 targets. Reliable surveillance data are essential for planning and monitoring PMTCT programs. Health worker motivation represents an opportunity to strengthen the system if adequate training, supervision, and supplies are ensured.
In line with recent World Health Organization recommendations, global strategies have evolved from the prevention of mother-to-child transmission (PMTCT) towards the broader framework of elimination of mother-to-child transmission (EMTCT) of HIV, syphilis, and hepatitis B. EMTCT goes beyond service coverage indicators and emphasizes sustained impact, data quality, surveillance performance, and verification of elimination targets.
In Togo, EMTCT initiatives are being progressively implemented through the integration of early infant diagnosis, maternal viral load monitoring, and routine surveillance within the national health information system. The country has strengthened antenatal screening, expanded access to antiretroviral therapy for pregnant and breastfeeding women, and reinforced follow-up mechanisms for HIV-exposed infants. These efforts are supported by routine program monitoring and periodic evaluations of surveillance system performance, as reflected in this study.
However, the transition from PMTCT to EMTCT requires robust surveillance systems capable of producing high-quality, timely, and complete data. Findings from this evaluation highlight persistent challenges related to data completeness, timeliness, and case definition clarity, which may limit the country’s ability to document progress toward EMTCT validation. Addressing these gaps is critical to ensure that surveillance data can effectively support EMTCT verification processes and guide evidence-based decision-making.
Limitations
The unavailability of individual-level data prevented the calculation of additional dispersion measures such as the interquartile range (Q1- Q3). In addition, exclusion of some peripheral sites, self-reported responses may have introduced social desirability and recall biases. Because the study was not designed as a formal qualitative inquiry, detailed quotations and thematic analysis were not available. Retrospective data analysis may also limit findings. Although the response rate was high (87.2%), the possibility of non-response bias cannot be excluded. Finally, some system performance indicators were assessed based on available reports and respondent perceptions, which may affect the precision of the findings. Despite these limitations, the study provides useful insights into the functioning of the HIV children surveillance system in Togo.
Conclusion
The national HIV infant surveillance system in Togo is operational, useful, and highly acceptable. However, substantial underreporting and data quality gaps, particularly in key variables such as breastfeeding status and EID turnaround time, limit its effectiveness. Strengthening the training of surveillance actors and improving data management practices are essential to enhance system performance and support progress toward national and global HIV targets.
Recommendations
National program: Strengthen routine training of surveillance actors, as only 26.6 % reported having received training on early infant diagnosis (EID), which may contribute to both underreporting and poor data completeness. Improve data quality monitoring systems, particularly for key variables such as breastfeeding status and PCR turnaround time, which were found to be poorly documented. Strengthen case reporting mechanisms to reduce the gap between expected and reported cases, as substantial underreporting was observed. Further exploration of the integration of EID surveillance into electronic health information systems, such as DHIS2, may be considered to improve reporting efficiency, although this was not directly assessed in the present study. Strengthening feedback mechanisms to reporting sites could be beneficial, but this aspect was not directly evaluated in the current study.
Service delivery: Ensure uninterrupted supply of test kits, improve DBS sample transport, and assign dedicated data focal points.
Laboratories: Enhance PCR processing capacity, systematically document turnaround times, and adopt electronic result tracking.
Partners: Support digitalisation, provide technical assistance for data analysis, and mobilise resources for training, supervision, and logistics.
What is already known about the topic
- Early infant diagnosis coverage in sub-Saharan Africa remains below WHO targets.
- Surveillance systems are essential for monitoring pediatric HIV.
- Data quality challenges limit reliability of surveillance in many African countries.
What this study adds
- Provides the first evaluation of the HIV infant surveillance system in Togo.
- Demonstrates strong simplicity and acceptability.
- Identifies underreporting and poor data completeness as critical weaknesses.
| Indicators | Calculation / Data Input | Indicator value | Data source | Collection techniques | Evaluation criteria |
|---|---|---|---|---|---|
| Utility | |||||
| Proportion of surveillance stakeholders who find the HIV surveillance system useful | Number of stakeholders who answered yes / total surveyed | 64/64 (100%; 95% CI: 94.4–100) | Stakeholders involved | Data extraction | ≥80%, Good performance |
| Proportion of newborns of HIV+ mothers who received ARV prophylaxis | Number receiving ARV / expected newborns | 81.5% | PNLS-HV-IST 2020 report | Annual report | ≥80%, Good performance |
| Proportion of HIV-positive pregnant women who initiated ART | HIV+ pregnant women on ART / total HIV+ pregnant women | 3627/3759 (96.5%) | ≥80%, Good performance | ||
| Utility performance | 92.6% | ≥80%, Good performance | |||
| Simplicity | |||||
| Clarity of case definition | Officers answering yes / total | 58/64 (90.6%) | Stakeholders | Interview | ≥80%, Good performance |
| Ease of sending samples | Agents answering yes / total | 56/64 (87.5%) | Stakeholders | Interview | ≥80%, Good performance |
| Simplicity of notification form | Agents recognizing simplicity / total | 62/64 (96.9%) | Stakeholders | Interview | ≥80%, Good performance |
| Simplicity performance | 91.6% | ≥80%, Good performance | |||
| Acceptability | |||||
| Perception of number of variables | Respondents saying not excessive / total | 62/64 (96.9%) | Stakeholders | Interview | ≥80%, Good performance |
| Completeness of reporting | Reported sites / total sites | 768/768 (100%) | DHIS2 database | Data extraction | ≥80%, Good performance |
| Time spent completing form | Agents responding yes / total | 64/64 (100%) | Stakeholders | Interview | ≥80%, Good performance |
| Stakeholder commitment | Committed stakeholders / total | 64/64 (100%) | Stakeholders | Data extraction | ≥80%, Good performance |
| Acceptability performance | 99.2% | ≥80%, Good performance | |||
| Data Quality | |||||
| PCR variable completeness | Complete PCR values / total forms | 2059/2087 (98.6%) | DHIS2 forms | Data extraction | ≥80%, Good performance |
| Delivery mode completeness | Complete delivery values / total forms | 543/2087 (26.3%) | DHIS2 forms | <60%, Low performance | |
| Lab arrival date completeness | Complete values / total forms | 907/2087 (43.4%) | DHIS2 forms | Data extraction | <60%, Low performance |
| Date of birth completeness | Complete DOB values / total forms | 2077/2087 (99.5%) | DHIS2 forms | Data extraction | ≥80%, Good performance |
| Data quality performance | 66.9% | (60–79%), Moderate performance | |||
| Occupation | Frequency | % |
|---|---|---|
| State-certified midwife | 29 | 42.7% |
| Medical Assistant | 26 | 38.2% |
| Doctor | 2 | 2.9% |
| Biolim Managers | 2 | 2.9% |
| Laboratory Technicians | 1 | 1.5% |
| Midwife assistant | 1 | 1.5% |
| PMTCT Manager | 1 | 1.5% |
| PNLS Monitoring and Evaluation Officer | 1 | 1.5% |

References
- Joint United Nations Programme on HIV/AIDS. UNAIDS data 2021 [Internet]. Geneva (Switzerland): Joint United Nations Programme on HIV/AIDS; 2021 [cited 2026 Apr 28]. 464 p. Available from: https://www.unaids.org/sites/default/files/media_asset/JC3032_AIDS_Data_book_2021_En.pdf
- Ngwej DT, Mukuku O, Mudekereza R, Karaj E, Fwamba EB, Luboya ON, Kakoma JBS, Wembonyama SO. Etude de facteurs de risque de la transmission du VIH de la mère à l’enfant dans la stratégie « option A » à Lubumbashi, République Démocratique du Congo [Study of risk factors for HIV transmission from mother to child in the strategy «option A» in Lubumbashi, Democratic Republic of Congo] [Internet]. Pan Afr Med J. 2015;22. [cited 2026 Apr 28]. doi:10.11604/pamj.2015.22.18.7480 French.
- World Health Organization. HIV diagnosis and ARV use in HIV-exposed infants: a programmatic update [Internet]. Geneva (Switzerland): World Health Organization; 2018 Jul 1 [cited 2026 Apr 28]. 24 p. Available from: https://www.who.int/publications/i/item/9789241550125
- World Health Organization. Antiretroviral therapy for HIV infection in infants and children – 2010 [Internet]. Geneva (Switzerland): World Health Organization; 2021 Jan 1 [cited 2026 Apr 28]. 228 p. Available from: https://www.who.int/publications/i/item/9789241599801
- Ankrah AK, Dako-Gyeke P. Factors influencing the delivery and uptake of early infant diagnosis of HIV services in Greater Accra, Ghana: a qualitative study [Internet]. PLoS ONE. 2021 Feb 17 [cited 2026 Apr 28];16(2):e0246876. doi:10.1371/journal.pone.0246876
- Ministère de la Santé (Togo). Rapport annuel 2020 des activités du PNLS-HV-IST [2020 Annual Report of PNLS-HV-IST Activities] [Internet]. Lomé (Togo): Ministère de la Santé (Togo); 2020 [cited 2026 Apr 28]. 49 p. French.
- Nsubuga P, Mabaya S, Apollo T, Sithole N, Komtenza B, Matare T, Chimwaza A, Takarinda K, Moyo B, Mbano L, Choto R, Moyo T, Lowrance D, Low-Beer D, Mugurungi O, Gasasira A. Evaluation of the Zimbabwe HIV case surveillance pilot project, 2019 [Internet]. Pan Afr Med J. 2020 Dec 17 [cited 2026 Apr 28];37. doi:10.11604/pamj.2020.37.353.25600
- Banze AR, Homo BP, Mussá TN, Baltazar CS, Boothe MS. Evaluation of prevention of mother-to-child transmission national health information system for HIV/AIDS, in southern region of Mozambique, April to November 2016 [Internet]. Pan Afr Med J. 2021 Jan 12 [cited 2026 Apr 28];38. doi:10.11604/pamj.2021.38.26.24255
- Abdulrazzak MH, Alsahybi AH, Assabri A, Khader Y. Evaluation of HIV reporting form in Sana’a City, Yemen, 2016 [Internet]. INQUIRY. 2019 May 7 [cited 2026 Apr 28];56:0046958019847020. doi:10.1177/0046958019847020
- Welty S, Motoku J, Muriithi C, Rice B, De Wit M, Ashanda B, Waruiru W, Mirjahangir J, Kingwara L, Bauer R, Njoroge D, Karimi J, Njoroge A, Rutherford GW. Brief report: recent HIV infection surveillance in routine HIV testing in Nairobi, Kenya: a feasibility study [Internet]. JAIDS J Acquir Immune Defic Syndr. 2020 May 1 [cited 2026 Apr 28];84(1):5–9. doi:10.1097/QAI.0000000000002317
- Migamba SM, Nyombi TN, Nsubuga EJ, Kwiringira A, Delaney A, Kabwama SN, Nakafeero M, Kwesiga B, Kadobera D, Monalisa-Mayambala P, Bulage L, Ario AR, Harris JR. Rapid antiretroviral therapy initiation following rollout of point-of-care early infant diagnosis testing, Uganda, 2018–2021 [Internet]. AIDS Res Ther. 2024 May 15 [cited 2026 Apr 28];21(1):31. doi:10.1186/s12981-024-00613-8
- Batisani K, Simusokwe R, Lubemba H, Chimanga B, Kwanga P, Mununga K. Botswana’s 95–95–95 achievement: pathways to HIV epidemic control [Internet]. Health Sci Rep. 2026 Feb 23 [cited 2026 Apr 28];9(2):e71904. doi:10.1002/hsr2.71904
