Research | Open Access | Volume 9 (Suppl 12): Article 2 | Published: 20 Apr 2026
Improving infection prevention and control during a Lassa fever outbreak: Experience from a military hospital in Nigeria
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Table 1: Selected IPC infrastructure and staffing subcomponents before and after intervention | ||
|---|---|---|
| Variable | Before intervention | After intervention |
| Built environment and infrastructure | ||
| Isolation ward fenced and cordoned off crowded areas of the hospital | Yes | Yes |
| Well ventilated | Yes | Yes |
| Regular supply of electricity and functional back-up power sources? | Yes | Yes |
| Water services available at all times and of sufficient quantity for all uses | Yes | Yes |
| Patient care (high-risk) area clearly separated from the general/administrative (low-risk) area | No | Yes |
| Separate entrances and exits for staff and patients | No | Yes |
| Dedicated space for staff to don PPE before entry into high-risk area | No | Yes |
| Dedicated area for doffing PPE with hand hygiene facility in place at the high-risk area | No | Yes |
| Any identified unidirectional workflow to prevent cross-contamination | No | Yes |
| Distance between two beds in the cohort bays more than 1 meter | Yes | Yes |
| Separate rooms for males and females | Yes | Yes |
| Staffing | ||
| Trained staff in the isolation ward | No | Yes |
| Dedicated staff assigned to preparing chlorine solution for disinfection | No | Yes |
| Dedicated cleaning staff | No | Yes |
| Dedicated IPC focal person in the ward | No | Yes |
| Daily IPC training for staff in the isolation ward | No | Yes |
| Staff trained to perform hand hygiene appropriately | No | Yes |
| Staff trained on the appropriate use of PPE | No | Yes |
| Staff trained on cleaning and disinfection of the isolation ward | Partially | Yes |
| Staff trained on healthcare waste management | Partially | Yes |
| Staff trained on safe sample packaging and transport | No | Yes |
Table 1: Selected IPC infrastructure and staffing subcomponents before and after intervention
| Table 2: Selected IPC equipment, practices and guidelines before and after intervention | ||
|---|---|---|
| Variable | Before intervention | After intervention |
| IPC equipment and materials | ||
| Functional hand hygiene stations at all points of care | No | Yes |
| Functional hand hygiene stations in general (low-risk) area | No | Yes |
| Patient-care equipment available and individualized | No | Yes |
| PPE available in different sizes and in sufficient quantity for clinical and support staff | Yes | Yes |
| Alcohol based hand rub available and adequate | Yes | Yes |
| Chlorine (powder/bleach) available and in sufficient quantity | Yes | Yes |
| Functional waste collection bins for segregation of waste at all waste generation points | No | Yes |
| Sharp boxes available at all points of care and not more than ¾ full | Yes | Yes |
| Incinerator available | Yes | Yes |
| IPC practices | ||
| Chlorine solution freshly prepared daily with the recommended formula | No | Yes |
| All surfaces in the patient care area are cleaned and disinfected twice daily | No | Yes |
| Staff wear full PPE to enter the patient care area | No | Yes |
| Waste segregated, transported and disposed of appropriately | No | Yes |
| IPC guidelines and SOPs | ||
| Clinical guidelines for the management of Lassa fever patients are available | No | No |
| SOP for Hand hygiene is available | No | No |
| SOP for cleaning and disinfection is available | No | No |
| SOP for Health care worker protection and safety is available | No | No |
| SOP for Waste management is available | No | No |
| SOP for donning and doffing PPE | No | No |
Table 2: Selected IPC equipment, practices and guidelines before and after intervention
| Table 3: IPC performance scores before and after IPC intervention | |||
|---|---|---|---|
| IPC Components | Before Intervention Score (%) | After Intervention Score (%) | Change (%) |
| Built Environment and Infrastructure | 53.2 | 86.8 | 33.6 |
| IPC Equipment and Materials | 44.7 | 89.5 | 44.8 |
| Staffing and Training | 35.0 | 91.7 | 56.7 |
| IPC guidelines/SOPs | 0.0 | 0.0 | 0.0 |
| Overall Score | 39.0 | 73.5 | 34.5 |
Table 3: IPC performance scores before and after IPC intervention
Figures
Keywords
- Lassa fever
- Infection Prevention and Control
- Outbreak
- Healthcare Worker
Ola Chikerendu Egbuta1&, Joseph Okoeguale2,3, Mojeed Olaitan Rafiu2,4,5, Christian Ehigbor Erohubie2,4,5, Pristar Omogbai2, Pokyes Hosea Daburum2, Jacqueline Agbukor2, Thomas Olokor2, Rita Omoafeba Esumeh2, Winifred Ambrose2, Emmanuel Illonah2, Reuben Agbons Eifediyi2,3
1Department of Community Medicine, Irrua Specialist Teaching Hospital, Irrua, Edo State, Nigeria, 2Institute of Viral and Emergent Pathogens, Control and Research, Irrua Specialist Teaching Hospital, Edo State, Nigeria, 3Department of Obstetrics and Gynaecology, Ambrose Alli University, Ekpoma, Edo State, Nigeria, 4Department of Internal Medicine, Irrua Specialist Teaching Hospital, Irrua, Edo State, Nigeria, 5Department of Medicine, Ambrose Alli University, Ekpoma, Edo State, Nigeria.
&Corresponding author: Ola Chikerendu Egbuta, Irrua Specialist Teaching Hospital, Irrua, Edo State, Nigeria; Email: olaegbutac@gmail.com ORCID: https://orcid.org/0000-0001-6520-3245
Received: 24 Nov 2025, Accepted: 16 Apr 2026, Published: 20 Apr 2026
Domain: Infectious Disease Epidemiology
Keywords: Lassa fever, Infection Prevention and Control, Outbreak, Healthcare Worker
©Ola Chikerendu Egbuta et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Ola Chikerendu Egbuta et al., Improving infection prevention and control during a Lassa fever outbreak: Experience from a military hospital in Nigeria. Journal of Interventional Epidemiology and Public Health. 2026; 9(Suppl 12):2. https://doi.org/10.37432/jieph-d-25-00301
Abstract
Introduction: Lassa fever remains a serious public health threat in Nigeria, with annual outbreaks affecting healthcare workers due to inadequate Infection Prevention and Control (IPC) measures. As of March 2024, the country had reported 806 confirmed cases and 150 deaths, including 32 infections among healthcare workers. An outbreak at a Nigerian military hospital resulted in four deaths, three of whom were healthcare workers. In response, a multidisciplinary emergency team from the Irrua Specialist Teaching Hospital (ISTH) was deployed to assess existing IPC facilities and implement rapid IPC interventions.
Method: A targeted intervention was conducted from 27th February to 29th March 2024. IPC facilities and practices within the isolation ward were assessed using a checklist adapted from the World Health Organisation 2017 IPC Assessment Framework and the Nigeria Centre for Disease Control and Prevention Guidelines on infection prevention and control for viral haemorrhagic fevers. Structural modifications were undertaken to reduce transmission risks and facilitate effective barrier nursing. Comprehensive training sessions were provided to staff, focusing on Lassa fever transmission, hand hygiene, appropriate use of personal protective equipment, environmental disinfection, waste disposal and chlorine preparation.
Result: Initial assessment revealed that the isolation ward scored 39.0%, which improved to 73.5% post-intervention. Key improvements included the demarcation of high and low risk zones, the establishment of donning and doffing areas, and the installation of hand hygiene stations at strategic points. Three confirmed cases were admitted, treated with intravenous ribavirin and supportive care, and subsequently discharged without further deaths. No additional health worker infections occurred.
Conclusion: The IPC intervention implemented was associated with a reduction in the risk of hospital-based transmission of Lassa fever at the Military hospital. Sustained investment in IPC infrastructure and personnel training is essential in ensuring effective responses to future outbreaks.
Introduction
Lassa fever is an acute viral haemorrhagic disease caused by the Lassa virus, an arenavirus endemic in West Africa[1,2] . Transmission occurs primarily through contact with food or household items contaminated by the excreta, urine or saliva of Mastomys natalensis rats. Secondary human-to-human transmission involving direct contact with blood or bodily fluids of infected persons may occur, especially within healthcare settings [2,3]. The disease poses a significant public-health burden, with an estimated 100000 – 300000 infections and approximately 5000 deaths occurring annually in the West African region [4,5]. Nigeria accounts for a substantial proportion of this burden, with year-round transmission and seasonal outbreaks occurring during the dry season, from November to March [6,7].
Healthcare facilities are often significantly affected during these outbreaks, as healthcare workers face heightened risks of infection, especially in facilities where infection prevention and control (IPC) measures are inadequate [8–10]. Factors such as limited access to personal protective equipment (PPE), poor adherence to standard precautions, and substandard infrastructure contribute to preventable disease transmission [8]. Effective IPC measures encompassing correct hand hygiene techniques, environmental cleaning and disinfection, the appropriate use of PPE, waste management, regular training, and proper patient screening, placement, and isolation are essential for interrupting transmission of viral haemorrhagic fevers and protecting healthcare workers [11]. Purpose-built isolation units for managing highly infectious diseases such as Lassa fever are a cornerstone of IPC. These units feature clearly defined high- and low-risk zones, secure access controls, separate entry and exit points for staff and patients, dedicated staff areas, well-ventilated rooms, reliable water supply, easy-to-clean surfaces, and capacity for expansion, all designed to minimise transmission risks to healthcare workers and the wider community [11]. However, many healthcare facilities in Nigeria lack such infrastructure and struggle to maintain effective IPC systems, particularly during outbreaks, due to resource constraints and limited institutional capacity [12–14].
As of March 2024, Nigeria had reported 806 confirmed cases of Lassa fever and 150 associated deaths, including 32 infections among healthcare workers [15]. Among the affected health institutions was a Nigerian military hospital that experienced an outbreak that resulted in four deaths, three of which occurred amongst healthcare workers. In response, a multidisciplinary emergency team from Irrua Specialist Teaching Hospital (ISTH), Edo State, was deployed to provide technical support. The team assessed existing IPC facilities, implemented rapid IPC interventions, and supported clinical case management within the isolation ward of the hospital. This article describes the IPC interventions implemented and the impact on IPC performance of the isolation ward of the affected military hospital.
Methods
The Irrua Specialist Teaching Hospital emergency team conducted a rapid IPC intervention in the isolation ward of a Nigerian military hospital between 27th February and 29th March 2024 during a Lassa fever outbreak.
Study setting
The hospital is a 600-bed tertiary healthcare facility providing clinical, laboratory and radiological services to military personnel and members of neighbouring communities. The isolation ward is a 25-bed facility comprising 5 separate bays designed for patient cohorting. It is fenced and situated away from the rest of the hospital to limit access and reduce the risk of disease transmission. Before intervention, the ward lacked clearly defined high-risk (patient care) and low-risk (nursing office/general) areas, and there were no designated areas for donning and doffing PPEs. These deficiencies increased the risk of cross-contamination between patients and staff.
IPC assessment and scoring
The baseline and post-intervention IPC assessments were conducted using an observational checklist adapted from the World Health Organisation IPC Assessment Framework (IPCAF) and the Nigeria Centre for Disease Control and Prevention (NCDC) Guidelines on Infection Prevention and Control for Viral Haemorrhagic Fevers [11,16]. Four key components were assessed, including the built environment and infrastructure, IPC equipment and materials, staffing and training and IPC guidelines/SOPs for the isolation ward. A total of 53 indicators were used to assess IPC performance. Each indicator was scored as 5 (meets requirement), 2.5 (partially meets requirement) and 0 (does not meet requirement). The component and overall scores were calculated as percentages using the formula:
\[
\frac{\text{Total Points Achieved}}{\text{Total Possible Points}} \times 100
\]
and categorised as inadequate (0 – 24%), basic (25 – 49%), intermediate (50 – 74%) and advanced (> 74%). The baseline assessment was conducted within the first two days of deployment, while the post-intervention assessment was conducted over one week before the end of the emergency response. To ensure scoring consistency, both assessments were conducted by the same assessment team.
The intervention
Key interventions were designed and implemented based on gaps identified during the baseline assessment, including:
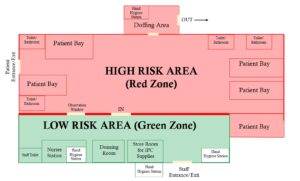
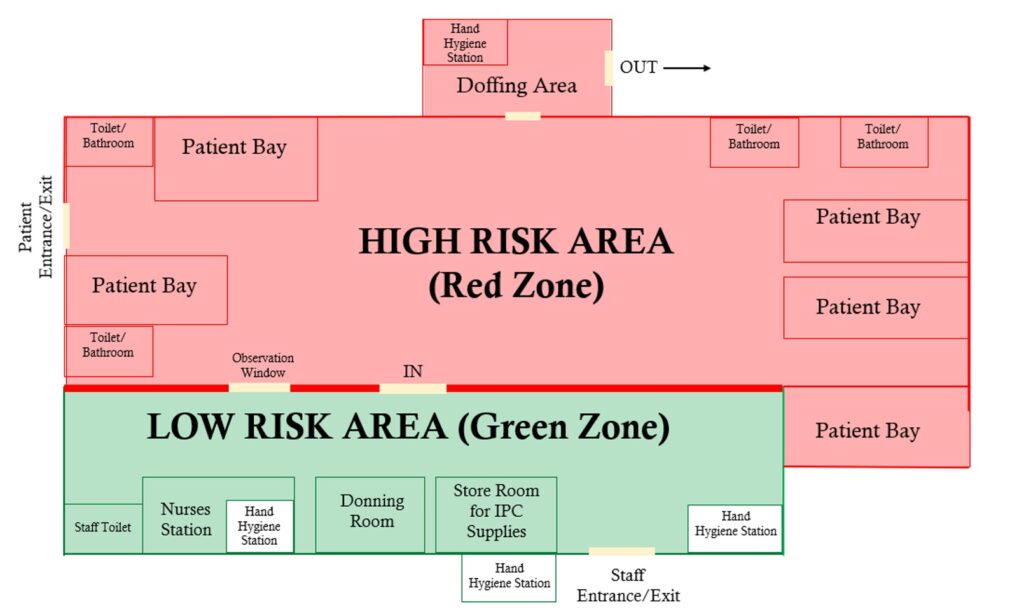
- Structural modification of the isolation ward: The ward was restructured to establish a clear separation between high-risk (patient care) and low-risk (nursing/general) zones (Figure 1). This involved the installation of a floor-to-ceiling wooden partition equipped with an observation window. In addition, a red demarcation line was clearly marked to reinforce restricted access into the high-risk zone and minimise the risk of cross-contamination.
- Creation of dedicated access routes: Separate entry and exit points were established for staff and patients (Figure 1).
- Establishment of designated PPE donning and doffing areas: A dedicated donning room was established within the low-risk area, while a doffing area equipped with a hand hygiene station and an infectious waste container for used PPE was set up in the high-risk zone before the exit (Figure 1).
- Installation of hand hygiene stations: Handwashing facilities and alcohol-based hand rubs were installed at points of care in the isolation ward.
- Provision of real-time Lassa PCR testing: The ISTH mobile laboratory provided real-time Lassa PCR testing, enabling prompt confirmation and management of cases.
- Comprehensive training sessions for healthcare workers: All healthcare workers received training on IPC measures, including hand hygiene, appropriate use of personal protective equipment, environmental disinfection, waste disposal and chlorine preparation. Staff assigned to the isolation ward participated in daily refresher training during the intervention period.
- Implementation of transmission-based precautions in the isolation ward: The ward was supplied with PPE daily to enable availability and easy access. Healthcare workers were regularly monitored and supervised on the use of PPE.
- Daily cleaning and disinfection of the ward: The cleaning staff were regularly trained and supervised to ensure twice daily cleaning and disinfection of the ward.
Data on patient outcomes were extracted from clinical case notes.
Ethical consideration
This study was conducted as part of a formally designated emergency outbreak response activity by the Irrua Specialist Teaching Hospital, Edo State, under the authority of the Federal Ministry of Health. Since the intervention was a response to a public health emergency, no ethical approval was sought from an Institutional Review Board or Ethics Committee. Permission to use anonymised clinical data was obtained verbally from the management of the military hospital. Informed consent was obtained from all patients at discharge for the use of their clinical data for reporting purposes, and confidentiality was maintained for all patient data.
Results
Baseline findings
Table 1 shows selected IPC infrastructure and staffing sub-components before and after IPC intervention. At baseline (before intervention), the isolation ward was fenced and well-ventilated with water services available at all times. However, major deficiencies across key areas were identified. The built environment lacked functional demarcation between the patient care area and general/administrative area; there were no dedicated donning and doffing areas, trained staff were not available in the isolation ward, and there was no dedicated IPC focal person (Table 1). Table 1 also showed that before intervention, staff were not appropriately trained on hand hygiene, the appropriate use of PPE and safe sample packaging and transport. Table 2 shows selected IPC equipment, practices and guidelines before and after intervention. At baseline, PPE, alcohol-based hand rub, and chlorine were available in sufficient quantities. However, there were no functional hand hygiene stations at points of care or in the general area, and staff did not wear full PPE to enter the patient care area. There were no available guidelines on the management of Lassa fever patients, hand hygiene, donning and doffing of PPE and cleaning and disinfection of the isolation ward.
Post-intervention findings
Following implementation of the interventions, the overall IPC performance score improved from 39.0% (basic) at baseline to 73.5% (Intermediate), with a 34.5% improvement in percentage score (Table 3). The built environment and infrastructure improved from 53.2% (intermediate) to 86.8% (advanced) after structural modifications, while staffing and training improved from 35.0% (basic) to 91.7% (advanced). However, the percentage score of the IPC guidelines/SOP component remained at 0.0% (inadequate) before and after intervention, as no written ward-specific IPC guidelines or SOPs were developed during the intervention period, despite the introduction and implementation of practical IPC measures. Specific improvements in IPC included:
- Clear demarcation of the ward into a high-risk (patient care) and a low-risk (general/administrative) area with improved unidirectional workflow.
- Establishment of dedicated donning and doffing areas.
- Installation of hand hygiene stations at strategic points of care.
- Regular cleaning and disinfection of the ward twice daily by supervised cleaning staff.
- Daily training of isolation ward staff on IPC
- Improved waste segregation, transport and disposal method
Case management and outcomes
During the intervention period, three confirmed cases of Lassa fever aged 30 -55 years were admitted into the isolation ward. All received intravenous ribavirin and supportive therapy and were discharged alive. No new health worker infections were recorded during the post-intervention phase.
Challenges
The response was constrained by limited financial resources, the unavailability of written IPC guidelines, and staff shortages. Despite these challenges, multidisciplinary collaboration and continuous supervision sustained IPC compliance throughout the response.
Discussion
This study demonstrates that rapid, well-coordinated infection prevention and control interventions can significantly improve the preparedness and response capacity of a military hospital isolation ward during a Lassa fever outbreak. The baseline assessment revealed substantial gaps across infrastructure, staffing, IPC equipment, and practices, consistent with findings from other Nigerian healthcare facilities, where there is weak IPC governance, poor IPC practices, and inadequate isolation facility design [10,17–19]. These gaps pose a major public health risk by increasing the occupational exposure of healthcare workers to Lassa fever and amplifying outbreaks within communities.
The improvement in the overall IPC performance of the isolation ward from 39% to 73.5% shows that rapid improvements can be achieved in resource-constrained settings when interventions are well-coordinated and supported by strong technical leadership. This experience confirmed that targeted environmental modifications, such as establishing clearly defined high-risk and low-risk zones and dedicated donning and doffing areas, are essential for reducing cross-infection and improving staff safety in suboptimal isolation units. Furthermore, improvements in hand hygiene and barrier nursing practices were associated with a reduced risk of exposure, as demonstrated by the absence of new healthcare worker infections during active case management.
A key lesson from this intervention is the critical role of continuous hands-on training. The increase in staffing and training domain from 35% to 91.7% illustrates how regular IPC drills, mentorship and close supervision are associated with improved staff competence, confidence and adherence to IPC standards. This finding is consistent with previous studies, which demonstrate that frequent practical training significantly improved adherence to hand hygiene, PPE use, and waste management [20–22].
Despite these gains, the absence of written IPC guidelines/SOPs in the isolation ward before and after the intervention was a significant gap which poses a risk to long-term sustainability. Written IPC guidelines are recognised as a core component of effective IPC programmes, ensuring standardised patient care, establishing clear procedures for IPC practices, and serving as essential tools for staff training and monitoring [23]. Without formal written guidelines or SOPs in the ward, improvements achieved during the emergency response may diminish once external support ceases or staffing changes occur. Adapting existing NCDC Lassa fever case management and IPC guidelines into ward-level SOPs would present a practical and cost-effective approach to addressing this gap.
Sustaining these IPC gains will further require strengthened governance structures and reliable resource allocation. Establishing a functional IPC team and appointing ward focal persons to oversee the implementation and periodic review of SOPs, as well as monitoring and evaluation of IPC practices, will enhance accountability [24,25]. Continuous training, particularly short hands-on refresher training during routine clinical activities and regular supportive supervision, will strengthen IPC compliance. Given the challenge of limited funding, incorporating IPC supplies and maintenance of IPC infrastructure into the recurrent budget of the hospital may be necessary to ensure uninterrupted availability of essential IPC materials and their maintenance.
The deployment of the Irrua Specialist Teaching Hospital outbreak response team under the authority of the Nigerian Federal Ministry of Health provided essential technical expertise, onsite training, supervision, and mobile diagnostic support. This collaboration strengthened the effectiveness and coordination of the response and improved the preparedness of the military healthcare facility for future outbreaks.
Study limitations
This study was conducted as part of an emergency outbreak response in a single military hospital, which limits the generalisability of the findings to other settings with different capacities or organisational structures. The assessment relied primarily on observation of the IPC system, which may introduce observer bias. This bias was minimised by the use of a standardised checklist adapted from the WHO and NCDC guidelines for infection prevention and control, and findings were cross-validated by an assessment team. Furthermore, because the study focused on rapid outbreak response, improvements were measured immediately after the intervention; therefore, the long-term sustainability of these gains could not be assessed.
Conclusion
The targeted IPC interventions, including infrastructural modification, regular staff training and strengthened routine IPC practices, were associated with rapid improvements in isolation ward performance, a reduction in occupational exposure and the absence of healthcare worker infections during a Lassa fever outbreak in a military hospital. However, sustaining these improvements beyond the outbreak period requires long-term institutional commitment. Continued investment in functional IPC infrastructure, development of ward-specific SOPs, establishment of functional IPC team and ward focal persons, regular personnel training, and uninterrupted supply of essential IPC materials are important in ensuring effective responses to future outbreaks. Strengthening collaboration between hospitals alongside dedicated government funding will further enhance national outbreak preparedness for Lassa fever and other highly infectious diseases.
What is already known about the topic
- Healthcare workers are at risk of Lassa fever infections during outbreaks.
- Strengthening IPC is essential for preventing hospital-based transmission of viral haemorrhagic fevers.
What this study adds
- This study demonstrates that rapid, targeted IPC interventions is associated with significant improvement in the IPC performance of a military healthcare facility during an active Lassa fever outbreak.
- It shows that IPC interventions such as infrastructural modification of an isolation unit and continuous, hands-on training can increase adherence to IPC practices and prevent healthcare worker infections.
- The experience highlights the value of collaboration between healthcare facilities in outbreak response.
Authors´ contributions
OCE, JO and RAE conceptualised the study. OCE, PO, PHD, JA, TO, ROE, WA and EI conducted the IPC assessment and interventions. JO, MOR and CEE supervised the IPC assessment and interventions. OCE prepared the first draft of the manuscript, and JO, MOR, CEE participated in manuscript writing and review to ensure intellectual content. All authors read and approved the final version of the manuscript.
| Table 1: Selected IPC infrastructure and staffing subcomponents before and after intervention | ||
|---|---|---|
| Variable | Before intervention | After intervention |
| Built environment and infrastructure | ||
| Isolation ward fenced and cordoned off crowded areas of the hospital | Yes | Yes |
| Well ventilated | Yes | Yes |
| Regular supply of electricity and functional back-up power sources? | Yes | Yes |
| Water services available at all times and of sufficient quantity for all uses | Yes | Yes |
| Patient care (high-risk) area clearly separated from the general/administrative (low-risk) area | No | Yes |
| Separate entrances and exits for staff and patients | No | Yes |
| Dedicated space for staff to don PPE before entry into high-risk area | No | Yes |
| Dedicated area for doffing PPE with hand hygiene facility in place at the high-risk area | No | Yes |
| Any identified unidirectional workflow to prevent cross-contamination | No | Yes |
| Distance between two beds in the cohort bays more than 1 meter | Yes | Yes |
| Separate rooms for males and females | Yes | Yes |
| Staffing | ||
| Trained staff in the isolation ward | No | Yes |
| Dedicated staff assigned to preparing chlorine solution for disinfection | No | Yes |
| Dedicated cleaning staff | No | Yes |
| Dedicated IPC focal person in the ward | No | Yes |
| Daily IPC training for staff in the isolation ward | No | Yes |
| Staff trained to perform hand hygiene appropriately | No | Yes |
| Staff trained on the appropriate use of PPE | No | Yes |
| Staff trained on cleaning and disinfection of the isolation ward | Partially | Yes |
| Staff trained on healthcare waste management | Partially | Yes |
| Staff trained on safe sample packaging and transport | No | Yes |
| Table 2: Selected IPC equipment, practices and guidelines before and after intervention | ||
|---|---|---|
| Variable | Before intervention | After intervention |
| IPC equipment and materials | ||
| Functional hand hygiene stations at all points of care | No | Yes |
| Functional hand hygiene stations in general (low-risk) area | No | Yes |
| Patient-care equipment available and individualized | No | Yes |
| PPE available in different sizes and in sufficient quantity for clinical and support staff | Yes | Yes |
| Alcohol based hand rub available and adequate | Yes | Yes |
| Chlorine (powder/bleach) available and in sufficient quantity | Yes | Yes |
| Functional waste collection bins for segregation of waste at all waste generation points | No | Yes |
| Sharp boxes available at all points of care and not more than ¾ full | Yes | Yes |
| Incinerator available | Yes | Yes |
| IPC practices | ||
| Chlorine solution freshly prepared daily with the recommended formula | No | Yes |
| All surfaces in the patient care area are cleaned and disinfected twice daily | No | Yes |
| Staff wear full PPE to enter the patient care area | No | Yes |
| Waste segregated, transported and disposed of appropriately | No | Yes |
| IPC guidelines and SOPs | ||
| Clinical guidelines for the management of Lassa fever patients are available | No | No |
| SOP for Hand hygiene is available | No | No |
| SOP for cleaning and disinfection is available | No | No |
| SOP for Health care worker protection and safety is available | No | No |
| SOP for Waste management is available | No | No |
| SOP for donning and doffing PPE | No | No |
| Table 3: IPC performance scores before and after IPC intervention | |||
|---|---|---|---|
| IPC Components | Before Intervention Score (%) | After Intervention Score (%) | Change (%) |
| Built Environment and Infrastructure | 53.2 | 86.8 | 33.6 |
| IPC Equipment and Materials | 44.7 | 89.5 | 44.8 |
| Staffing and Training | 35.0 | 91.7 | 56.7 |
| IPC guidelines/SOPs | 0.0 | 0.0 | 0.0 |
| Overall Score | 39.0 | 73.5 | 34.5 |

References
- World Health Organization. Lassa fever [Internet]. Geneva (Switzerland): WHO; 2024 Dec 5 [cited 2026 Apr 20]. Available from: https://www.who.int/news-room/fact-sheets/detail/lassa-fever
- Asogun DA, Günther S, Akpede GO, Ihekweazu C, Zumla A. Lassa Fever: Epidemiology, Clinical Features, Diagnosis, Management and Prevention. Infect Dis Clin North Am [Internet]. 2019 Dec [cited 2026 Apr 20];33(4):933–51. doi: 10.1016/j.idc.2019.08.002. Available from: https://doi.org/10.1016/j.idc.2019.08.002
- World Health Organization. Lassa Fever [Internet]. Geneva (Switzerland): WHO; 2024 Dec 5 [cited 2026 Apr 20]; [about 7 screens]. Available from: https://www.who.int/news-room/fact-sheets/detail/lassa-fever
- Sogoba N, Feldmann H, Safronetz D. Lassa Fever in West Africa: Evidence for an Expanded Region of Endemicity. Zoonoses Public Health [Internet]. 2012 Sep 7 [cited 2026 Apr 20];59(s2):43–7. doi: 10.1111/j.1863-2378.2012.01469.x. Available from: https://doi.org/10.1111/j.1863-2378.2012.01469.x
- Dwalu E, Jetoh RW, Shobayo BI, Pewu I, Taweh F, Wilson-Sesay HW, Akpan GE, Shannon F, Joseph BO, Umeokonkwo CD, Adewuyi P, Amo-Addae M, Nagbe TK, Gilayeneh J, MaCauley JA. Trend of Lassa fever cases and factors associated with mortality in Liberia, 2016 – 2021: a secondary data analysis. Pan Afr Med J [Internet]. 2024 Jan 19 [cited 2026 Apr 20];47:22. doi: 10.11604/pamj.2024.47.22.42156. Available from: https://doi.org/10.11604/pamj.2024.47.22.42156
- Asogun DA, Adomeh DI, Ehimuan J, Odia I, Hass M, Gabriel M, Ölschläger S, Becker-Ziaja B, Folarin O, Phelan E, Ehiane PE, Ifeh VE, Uyigue EA, Oladapo YT, Muoebonam EB, Osunde O, Dongo A, Okokhere PO, Okogbenin SA, Momoh M, Alikah SO, Akhuemokhan OC, Imomeh P, Odike MAC, Gire S, Andersen K, Sabeti PC, Happi CT, Akpede GO, Günther S. Molecular Diagnostics for Lassa Fever at Irrua Specialist Teaching Hospital, Nigeria: Lessons Learnt from Two Years of Laboratory Operation. Bausch DG, editor. PLoS Negl Trop Dis [Internet]. 2012 Sep 27 [cited 2026 Apr 20];6(9):e1839. doi: 10.1371/journal.pntd.0001839. Available from: https://doi.org/10.1371/journal.pntd.0001839
- Asogun D, Arogundade B, Unuabonah F, Olugbenro O, Asogun J, Aluede F, Ehichioya D. A Review of the Epidemiology of Lassa Fever in Nigeria. Microorganisms [Internet]. 2025 Jun 18 [cited 2026 Apr 20];13(6):1419. Available from: https://www.mdpi.com/2076-2607/13/6/1419
- Okwor T, Ndu A, Okeke T, Arinze-Onyia S, Chinawa A, Kassy W, Ochie C. A review of Lassa fever outbreaks in Nigeria from 1969 to 2017: Epidemiologic profile, determinants and public health response. Niger J Med [Internet]. 2018 Jul-Sep [cited 2026 Apr 20];27(3):219. doi: 10.4103/1115-2613.278784. Available from: https://doi.org/10.4103/1115-2613.278784
- Hamblion EL, Raftery P, Wendland A, Dweh E, Williams GS, George RNC, Soro L, Katawera V, Clement P, Gasasira AN, Musa E, Nagbe TK. The challenges of detecting and responding to a Lassa fever outbreak in an Ebola-affected setting. Int J Infect Dis [Internet]. 2017 Nov 11 [cited 2026 Apr 20];66:65–73. doi: 10.1016/j.ijid.2017.11.007. Available from: https://doi.org/10.1016/j.ijid.2017.11.007
- Chikwe I, Muhammad S, Oladipupo I, Winifred U, Sandra, Chukwuyem A, Chioma DN, Elsie I, Adunola O, Usman A, Moses T, John O, Lateefat A, Adebola O, Lois O, Osahogie E, Ibrahim M, Patrick E, Damian N. Exposure Incidents and Outcome of Lassa Fever Virus (LASV) Infection among Healthcare Workers in Nigeria, 2019. J Infect Dis Epidemiol [Internet]. 2020 Oct 16 [cited 2026 Apr 20];6(5). doi: 10.23937/2474-3658/1510168. Available from: https://doi.org/10.23937/2474-3658/1510168
- Nigeria Centre for Disease Control and Prevention (NCDC). National Guidelines on Infection Prevention and Control of Viral Haemorrhagic Fevers [Internet]. Abuja (Nigeria): NCDC; 2017 Apr [cited 2026 Apr 20]; 59 p. Available from: https://www.ncdc.gov.ng/themes/common/docs/protocols/18_1501495944.pdf
- Falana ROA, Ogidan OC, Fajemilehin BR. Barriers to infection prevention and control implementation in selected healthcare facilities in Nigeria. Infect Dis Now [Internet]. 2024 Feb [cited 2026 Apr 20];54(3):104877. doi: 10.1016/j.idnow.2024.104877. Available from: https://doi.org/10.1016/j.idnow.2024.104877
- Eneh SC, Obi CG, Ephraim Ikpongifono U, Dauda Z, Udoewah SA, Anokwuru CC, Onukansi FO, Ikhuoria OV, Ojo TO, Madukaku CU, Orabueze IN, Chizoba AF. The resurgence of Lassa fever in Nigeria: economic impact, challenges, and strategic public health interventions. Front Public Health [Internet]. 2025 Jul 16 [cited 2026 Apr 20];13:1574459. doi: 10.3389/fpubh.2025.1574459. Available from: https://doi.org/10.3389/fpubh.2025.1574459
- Denue B, Stephen M, Dauvoux I. The unending threat of Lassa fever in Nigeria, what can be done; what should be done. Port Harcourt Med J [Internet]. 2017 Dec 30 [cited 2026 Apr 20];11(3):113. doi: 10.60787/phmj.v11i3.132. Available from: https://doi.org/10.60787/phmj.v11i3.132
- Nigeria Centre for Disease Control and Prevention (NCDC). NCDC Lassa Fever Situation Report Epi Week 13: 25th – 31st March 2024 [Internet]. Abuja (Nigeria): NCDC; 2024 Apr 11 [cited 2026 Apr 20]. Available from: https://ncdc.gov.ng/themes/common/files/sitreps/1aabc83ea46f825a772894b4a53ab870.pdf
- World Health Organization (WHO). Infection Prevention and Control Assessment Framework at the Facility Level [Internet]. Geneva (Switzerland): WHO; 2018 Nov 6 [cited 2026 Apr 20]; 15 p. Available from: https://www.who.int/publications/i/item/WHO-HIS-SDS-2018.9
- Obionu IM, Ochu CL, Ukponu W, Okwor T, Dan-Nwafor C, Ilori E, Ihekweazu C. Evaluation of infection prevention and control practices in Lassa fever treatment centers in north-central Nigeria during an ongoing Lassa fever outbreak. J Infect Prev [Internet]. 2021 Aug 28 [cited 2026 Apr 20];22(6):275–82. doi: 10.1177/17571774211035838. Available from: https://doi.org/10.1177/17571774211035838
- Ijarotimi I, Ilesanmi O, Aderinwale A, Abiodun-Adewusi O, Okon I. Knowledge of Lassa fever and use of infection prevention and control facilities among health care workers during Lassa fever outbreak in Ondo State, Nigeria. Pan Afr Med J [Internet]. 2018 May 24 [cited 2026 Apr 20];30:56. doi: 10.11604/pamj.2018.30.56.13125. Available from: https://doi.org/10.11604/pamj.2018.30.56.13125
- Odegbemi OB, Umeokonkwo CD, Nwachukwu W, Nwaekpe CN, Oladejo J, Azuogu B, Ojide K, Agboeze J, Ilori E, Balogun MS, Nguku P, Ihekweazu C. Infection prevention and control in a treatment centre during a Lassa fever outbreak in southeastern Nigeria – January, 2018. Glob Biosecurity [Internet]. 2019 Nov 21 [cited 2026 Apr 20];1(3). doi: 10.31646/gbio.44. Available from: https://doi.org/10.31646/gbio.44
- Ilesanmi OS, Ayodeji OO, Bakare AA, Adedosu N, Adeagbo A, Odutayo A, Ayun FO, Bello AE. Infection prevention and control (IPC) at a Lassa fever treatment center before and after the implementation of an intensive IPC program. Jidhealth [Internet]. 2020 Oct 21 [cited 2026 Apr 20];3(3):213–6. doi: 10.47108/jidhealth.Vol3.Iss3.66. Available from: https://doi.org/10.47108/jidhealth.Vol3.Iss3.66
- Ousman K, Kabego L, Talisuna A, Diaz J, Mbuyi J, Houndjo B, Ngandu JP, Omba G, Aruna A, Mossoko M, Djingarey MH, Balde T, Abok P, Diallo B, Dovlo D, Yao M, Fortin A, Formenty P, Fall IS. The impact of Infection Prevention and control (IPC) bundle implementation on IPC compliance during the Ebola virus outbreak in Mbandaka/Democratic Republic of the Congo: a before and after design. BMJ Open [Internet]. 2019 Sep 5 [cited 2026 Apr 20];9(9):e029717. doi: 10.1136/bmjopen-2019-029717. Available from: https://doi.org/10.1136/bmjopen-2019-029717
- Kinyenje E, Hokororo J, Ngowi R, Kiremeji M, Mnunga E, Samwel A, Sylvanus E, Mnken E, Yango M, Mtalika M, Mmbaga V, Saitoti N, Malecha A, Kundy F, Rwabilimbo M, Kaniki I, Mwisomba G, Charles E, Mughanga P, Kitambi M, Paul R, Richard E, Musyani A, Rabiel I, Haule G, Marandu L, Mwakapasa E, Manasseh G, Sindato C, Beyanga M, Kapyolo E, Jacob F, Mcharo J, Mayige M, Msemwa F, Saguti G, Kauki G, Masuma J, Mrema G, Kohi M, Yoti Z, Habtu M, Mwengee W, Mukurasi K, Gatei W, Ruggajo P, Kwesi E, Eliakimu E, Horumpende P, Magembe G, Nagu T. Infection prevention and control of highly infectious pathogens in resource-limited countries: an experience from Marburg viral disease outbreak in Kagera Region – Tanzania. BMC Infect Dis [Internet]. 2024 Jun 24 [cited 2026 Apr 20];24(1):628. doi: 10.1186/s12879-024-09508-5. Available from: https://doi.org/10.1186/s12879-024-09508-5
- World Health Organization (WHO). Guidelines on core components of infection prevention and control programmes at the national and acute health care facility level [Internet]. Geneva (Switzerland): WHO; 2016 Nov 1 [cited 2026 Apr 20]; 91 p. Available from: https://www.who.int/publications/i/item/9789241549929
- Keil V, Schaumburg T, Buchta F, Luz JK, Kolbe-Busch S, Chaberny IF, Von Lengerke T. Improving leadership skills of infection prevention and control teams by psychological empowerment: study protocol for a cluster randomised controlled trial (IP-POWER). BMJ Open [Internet]. 2024 Feb 12 [cited 2026 Apr 20];14(2):e083806. doi: 10.1136/bmjopen-2023-083806. Available from: https://doi.org/10.1136/bmjopen-2023-083806
- Hale R, Powell T, Drey NS, Gould DJ. Working practices and success of infection prevention and control teams: a scoping study. J Hosp Infect [Internet]. 2014 Nov 21 [cited 2026 Apr 20];89(2):77–81. doi: 10.1016/j.jhin.2014.10.006. Available from: https://doi.org/10.1016/j.jhin.2014.10.006
