Research | Open Access | Volume 9 (1): Article 33 | Published: 23 Feb 2026
Clinical presentation and risk factors for severe outcomes in suspected kush poisoning, Sierra Leone, June–November 2025
Menu, Tables and Figures
Navigate this article
Tables
| Table 1: Demographic, geographic, and substance use characteristics (N=312) | |
|---|---|
| Characteristic | n (%) or median [IQR] |
| Treatment centre (District) | |
| Kissy Mental Health Unit (Western Area Urban) | 142 (46) |
| Bo Government Hospital (Bo) | 78 (25) |
| Port Loko Government Hospital (Port Loko) | 52 (17) |
| Kenema Government Hospital (Kenema) | 40 (13) |
| Demographics | |
| Age, years | 24 [19–31] |
| Male sex | 294 (94) |
| Education level | |
| None | 78 (25) |
| Primary | 131 (42) |
| Secondary | 89 (29) |
| Tertiary | 14 (4) |
| Occupation | |
| Unemployed | 163 (52) |
| Informal work | 87 (28) |
| Formal employment | 37 (12) |
| Student | 25 (8) |
| Residence | |
| Urban | 241 (77) |
| Rural | 71 (23) |
| Substance use history | |
| Duration of kush use >1 year | 89 (29) |
| Previous overdose episode | 89 (29) |
| Polysubstance use* | 156 (50) |
| Alcohol | 98 (31) |
| Cannabis | 67 (21) |
| Tramadol | 34 (11) |
| Presentation | |
| Time from use to presentation >6 hours† | 112/290 (39) |
| *Substance categories not mutually exclusive. †22 records missing time data; percentage based on valid responses (n=290). IQR = interquartile range. | |
Table 1: Demographic, geographic, and substance use characteristics (N=312)
| Table 2: Clinical presentation of cases among suspected kush poisoning, Sierra Leone, June-November 2025 (N=312) | |
|---|---|
| Clinical finding | n (%) or median [IQR] |
| Neurological findings | |
| Altered consciousness (GCS ≤14) | 234 (75) |
| Glasgow Coma Scale score | 11 [8–14] |
| GCS <8 (severe obtundation) | 67 (21) |
| Miosis | 198 (63) |
| Respiratory findings | |
| Respiratory compromise (composite)* | 128 (41) |
| Respiratory rate <12/min | 94 (30) |
| SpO₂ <92% | 76 (24) |
| Skin findings (comorbid condition) | |
| Skin ulcerations present | 112 (36) |
| Lower limb† | 78 (70) |
| Upper limb† | 45 (40) |
| Wound infection | 98 (31) |
| qSOFA ≥2 | 23 (7) |
| Treatment | |
| Naloxone administered | 186 (60) |
| Response to naloxone‡ | 143 (77) |
| Repeat dosing required‡ | 39 (27) |
| Supplemental oxygen required | 156 (50) |
| *RR <12/min OR SpO₂ <92%; 42 had both. †Among 112 with ulcerations; not mutually exclusive. ‡Among naloxone recipients (n=186 for response; n=143 for repeat dosing). GCS = Glasgow Coma Scale; qSOFA = quick Sequential Organ Failure Assessment. | |
Table 2: Clinical presentation of cases among suspected kush poisoning, Sierra Leone, June-November 2025 (N=312)
| Table 3: Risk factors for severe outcome—univariable and multivariable analysis (N=312) | |||||
|---|---|---|---|---|---|
| Characteristic | Severe n=45 (%) | Non-severe n=267 (%) | p-value | aOR (95% CI) | Adj. p |
| Age, median [IQR] | 26 [20–33] | 24 [19–30] | 0.12* | 1.2 (0.8–1.8)† | 0.34 |
| Male sex | 43 (96) | 251 (94) | 0.67‡ | 1.4 (0.3–6.2) | 0.67 |
| Education ≤primary | 32 (71) | 177 (66) | 0.52 | 1.2 (0.6–2.4) | 0.62 |
| Polysubstance use | 31 (69) | 125 (47) | 0.006 | 2.4 (1.2–4.6) | 0.010 |
| Previous overdose | 20 (44) | 69 (26) | 0.014 | 2.3 (1.2–4.5) | 0.016 |
| Duration >1 year | 18 (40) | 71 (27) | 0.07 | 1.8 (0.9–3.6) | 0.09 |
| Presentation >6 h | 23 (51) | 89 (33) | 0.015 | 2.0 (1.0–3.9) | 0.043 |
| Wound infection | 21 (47) | 77 (29) | 0.019 | 2.1 (1.1–4.2) | 0.032 |
| Bold indicates statistically significant independent risk factors (adjusted p<0.05). aOR = adjusted odds ratio from mixed-effects logistic regression with random intercept for treatment centre. *Mann-Whitney U test. †Per 10-year increase. ‡Fisher’s exact test. Model: C-statistic 0.72 (95% CI 0.64–0.80); Hosmer-Lemeshow χ²=6.2, p=0.62; ICC 0.05; all VIF <2.0. Sensitivity analysis (complete-case, n=278): polysubstance use aOR 2.3 (95% CI 1.1–4.7), previous overdose aOR 2.2 (95% CI 1.1–4.6), delayed presentation aOR 1.9 (95% CI 0.9–3.8), wound infection aOR 2.0 (95% CI 1.0–4.3). | |||||
Table 3: Risk factors for severe outcome—univariable and multivariable analysis (N=312)
Figures
Keywords
- Nitazenes
- Synthetic opioids
- Kush
- Risk factors
- Sierra Leone
Eric Nzirakaindi Ikoona1,&, Lucy Namulemo2, Mary Magdelene Sinnah1, Mohammad Alex Vandi1, Foday Sahr1
1National Public Health Agency, Freetown, Sierra Leone, 2Lindsey Wilson University, School of Professional Counseling, Columbia, Kentucky, USA
&Corresponding author: Eric Nzirakaindi Ikoona; National Public Health Agency, Freetown, Sierra Leone; Email: ikoonae@yahoo.com, ORCID: https://orcid.org/0000-0003-3402-1961
Received: 21 Dec 2025, Accepted: 20 Feb 2026, Published: 23 Feb 2026
Domain: Mental Health
Keywords: Nitazenes, synthetic opioids, kush, risk factors, Sierra Leone
©Eric Nzirakaindi Ikoona et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Eric Nzirakaindi Ikoona et al., Clinical presentation and risk factors for severe outcomes in suspected Kush poisoning, Sierra Leone, June–November 2025. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):33. https://doi.org/10.37432/jieph-d-25-00329
Abstract
Introduction: Sierra Leone declared a national public health emergency in April 2024 in response to widespread use of kush, an illicit substance containing nitazene-class synthetic opioids and synthetic cannabinoids. We describe clinical presentation and identify risk factors for severe outcomes among patients presenting with suspected acute kush poisoning.
Methods: Guided by the Risk Environment Framework, we conducted a multicentre retrospective cohort study at four treatment centres in Sierra Leone (June–November 2025). Cases included patients with acute opioid toxidrome and reported kush use within 24 hours. Severe outcomes were defined hierarchically as death, mechanical ventilation, or clinical instability (GCS <8 or qSOFA ≥2). Mixed-effects logistic regression identified independent risk factors for severe outcomes.
Results: Among 312 patients, the median age was 24 years (IQR 19–31); 94% were male, and 45 (14%) had experienced severe outcomes, including 9 deaths (3%). Clinical findings reflected opioid toxidrome: altered consciousness (75%), miosis (63%), and respiratory depression (41%). Independent risk factors for severe outcomes included polysubstance use (adjusted odds ratio [aOR] 2.4; 95% CI 1.2–4.6), previous overdose (aOR 2.3; 95% CI 1.2–4.5), delayed presentation >6 hours (aOR 2.0; 95% CI 1.0–3.9), and wound infection (aOR 2.1; 95% CI 1.1–4.2). Among naloxone recipients, 77% responded; 27% required repeat dosing.
Conclusion: Kush poisoning presents with opioid toxidrome consistent with nitazene pharmacology. Polysubstance use, prior overdose, delayed presentation, and wound infection are identifiable and modifiable risk factors for severity. Findings support targeted naloxone distribution, community-based harm reduction interventions, and integrated treatment services.
Introduction
Sierra Leone is confronting a growing substance use crisis driven by the widespread consumption of kush, an illicit drug locally manufactured from imported synthetic compounds and dried plant material [1,2]. In contrast to cannabis strains colloquially called kush in other contexts, the Sierra Leonean variant contains no natural cannabis but instead consists of plant matter adulterated with potent synthetic psychoactive agents [3,4].
Chemical analysis of seized drug samples by law enforcement partners has identified the principal toxic constituents. Using Fourier-transform infrared spectroscopy (FTIR) for initial screening, with confirmation by liquid chromatography–tandem mass spectrometry (LC–MS/MS), testing detected nitazene-class synthetic opioids (including protonitazene, metonitazene, and isotonitazene) and synthetic cannabinoids (including MDMB-4en-PINACA). Notably, composition varies considerably across samples and over time [3,4].
Nitazenes, originally synthesized in the 1950s but never approved for clinical use, are benzimidazole-derived synthetic opioids with potency that rivals or exceeds fentanyl [5]. Their steep dose-response curves and variable potency across analogues create significant overdose risk [6,7]. Fatalities involving nitazenes have risen in Europe and North America since 2020, typically in the context of polysubstance use [8,9].
On 5 April 2024, the President of Sierra Leone declared kush a national public health emergency [10]. The declaration, formalised through Constitutional Instrument No. 5 of 2024, authorised the establishment of specialised treatment centres and emergency response coordination [11]. The World Health Organization subsequently deployed technical support for clinical management and surveillance strengthening [12]. Despite these efforts, peer-reviewed literature characterising the clinical manifestations and severity of kush intoxication in Sierra Leone remains limited to phenomenological studies exploring user experiences and recovery challenges [1,2].
Understanding the clinical presentation and identifying risk factors for severe outcomes is essential for three reasons. First, healthcare workers need evidence-based guidance to recognise kush poisoning and initiate appropriate treatment in resource-limited settings where toxicological confirmation is unavailable. Second, identification of modifiable risk factors can inform targeted harm reduction interventions. Third, characterisation of clinical patterns can support the development of clinical protocols and resource allocation decisions for the expanding treatment centre network.
This study was guided by the Risk Environment Framework [13], which explains how drug-related harms emerge from interactions between individual factors (substance use patterns, prior overdose, comorbidities) and environmental factors (healthcare accessibility, care-seeking behaviour). This framework links the risk factors examined to actionable harm reduction strategies.
This study addresses the evidence gap by describing the clinical presentation and demographic characteristics of patients with suspected acute kush poisoning, and identifying pre-existing, behavioural, and comorbid risk factors associated with severe outcomes. These findings aim to inform risk stratification, harm reduction strategies, and clinical management protocols.
Methods
Study design and setting
We conducted a multicentre retrospective cohort study at four government-designated kush treatment centres across four districts in Sierra Leone: Western Area Urban District (Kissy Mental Health Unit, 50 beds), Bo District (Bo Government Hospital Kush Unit, 40 beds), Port Loko District (Port Loko Government Hospital Kush Unit, 30 beds), and Kenema District (Kenema Government Hospital Kush Unit, 25 beds). These facilities received patients through self-presentation, family referral, law enforcement transport, and ambulance transfer. The study period extended from 1 June to 30 November 2025.
Case definition
We defined suspected acute kush poisoning as presentation with acute opioid toxidrome (two or more of: altered consciousness with Glasgow Coma Scale [GCS] ≤14, miosis, respiratory compromise defined as respiratory rate [RR] <12/min or oxygen saturation [SpO₂] <92% on room air) plus self-reported or witness-reported kush use within the preceding 24 hours. We excluded patients with alternative primary diagnoses explaining the presentation: severe malaria with positive rapid diagnostic test, clinical meningitis with cerebrospinal fluid abnormalities, diabetic emergency with confirmatory glucose, or primary sepsis without temporal relationship to kush use.
Data collection and procedures
Data abstractors were community health officers (diploma-level clinical officers) or registered nurses with at least two years of experience in emergency or mental health care. Two abstractors were assigned to each site and received standardised training on the abstraction protocol during a two-day workshop that included review of case definitions, hands-on practice with sample records, and calibration exercises to ensure consistent interpretation of clinical documentation. Data were extracted from three source documents at each site: emergency department registers, ward admission logs, and individual case files. Abstractors followed a standardised workflow: first screening registers for potentially eligible patients by date and presenting complaint, then retrieving and reviewing individual case files, and finally entering data into the electronic abstraction form. This workflow was documented in a standard operating procedure manual distributed to all sites.
To ensure standardisation across sites, we conducted weekly teleconferences among all site teams to review abstraction challenges, discuss ambiguous cases, and clarify variable definitions. Abstraction was supervised by a site physician who reviewed completed forms weekly for completeness and clinical plausibility. When abstractors disagreed on coding, they first attempted consensus through joint re-review of source documents; unresolved discrepancies were escalated to the supervising physician for final adjudication.
We collected demographic characteristics, including age, sex, education level, occupation, and district of residence; substance use history (self-reported or proxy-reported); clinical findings at presentation; treatment administered; and outcomes. Inter-rater reliability, assessed on 30 randomly selected records abstracted independently by both abstractors at each site, showed substantial agreement (Cohen’s kappa 0.84, 95% CI 0.78–0.90). For patients with multiple presentations during the study period, only the first episode meeting case definition was included.
Instruments
The standardised electronic data abstraction form was developed using KoboToolbox (Harvard Humanitarian Initiative, Cambridge, MA) and included built-in skip logic, range checks, and mandatory fields to minimise missing data and entry errors. The instrument comprised five domains: (1) patient demographics (age, sex, education, occupation, residence); (2) substance use history (kush use duration and frequency, polysubstance use, previous overdose); (3) clinical presentation (vital signs, Glasgow Coma Scale, respiratory status, pupil examination, skin findings); (4) treatment received (naloxone administration and response, oxygen therapy, level of care); and (5) outcomes (discharge status, length of stay, complications). The form was pilot tested on 20 records at each site prior to study initiation; feedback from pilot testing led to refinements in variable definitions and response options to improve clarity and capture site-specific documentation practices. The data abstraction form is available from the corresponding author upon request. Data were uploaded to a secure central server at the National Public Health Agency with automated quality checks for completeness and logical consistency.
Variable definitions
Potential risk factors were limited to characteristics identifiable before or independent of the acute presentation: (1) demographic factors (age, sex, education level categorised as none/primary vs secondary/tertiary, occupation categorised as unemployed, informal work, formal employment, or student, and urban/rural residence); (2) substance use history (duration of kush use, frequency of use, polysubstance use, previous overdose); and (3) comorbid conditions (wound infection, defined as purulent discharge with surrounding erythema). Time from last use to presentation was included as a behavioral factor reflecting delay in care-seeking. Polysubstance use was defined as concurrent use of alcohol, cannabis, tramadol, or other substances within 24 hours of kush use.
Outcomes
Severe outcome was defined hierarchically as: (1) death during the treatment episode, (2) respiratory failure requiring mechanical ventilation among survivors, or (3) clinical instability, defined as GCS <8 or quick Sequential Organ Failure Assessment (qSOFA) ≥2, among survivors not requiring mechanical ventilation. This hierarchical definition ensures mutually exclusive categories based on objective clinical criteria while capturing the full spectrum of severe presentations. Non-severe outcome was defined as survival to discharge without mechanical ventilation or clinical instability.
Naloxone dosing
Naloxone was administered per national clinical guidelines, with an initial dose of 0.4 mg intravenously or intramuscularly, repeated at 2–3-minute intervals, up to a maximum of 10 mg if the initial response was inadequate [14].
Statistical analysis
We summarised continuous variables as medians with interquartile ranges (IQR) and categorical variables as frequencies with percentages. We compared characteristics between patients with and without severe outcomes using chi-square tests (or Fisher’s exact test when expected cell counts <5) for categorical variables.
We used mixed-effects logistic regression with a random intercept for treatment centre to identify independent risk factors for severe outcomes. Candidate predictors were pre-specified based on clinical rationale and limited to factors identifiable before or independent of acute presentation: age, sex, education level, polysubstance use, previous overdose history, duration of kush use >1 year, time from use to presentation >6 hours, and wound infection. We assessed multicollinearity using variance inflation factors (VIF <2.5 considered acceptable). Model discrimination was assessed using the c-statistic; calibration was assessed using the Hosmer-Lemeshow goodness-of-fit test.
We conducted sensitivity analyses using complete-case analysis, excluding records with any missing data. Missingness varied by variable: time from use to presentation was missing in 22 (7%) records and blood glucose was missing in 47 (15%); blood glucose missingness did not affect model variables. Statistical significance was set at α=0.05. Analyses were performed using Stata 17 (StataCorp, College Station, TX). Reporting follows STROBE guidelines [15].
Ethical considerations
The Sierra Leone Ethics and Scientific Review Committee approved this study (SLESRC-2025-043) as a retrospective records review with waiver of individual informed consent. Data were anonymised prior to analysis by removing names and assigning study identifiers. The retrospective design and consent waiver reflect pragmatic considerations for research in resource-limited settings where prospective consent would be infeasible.
Results
Study population
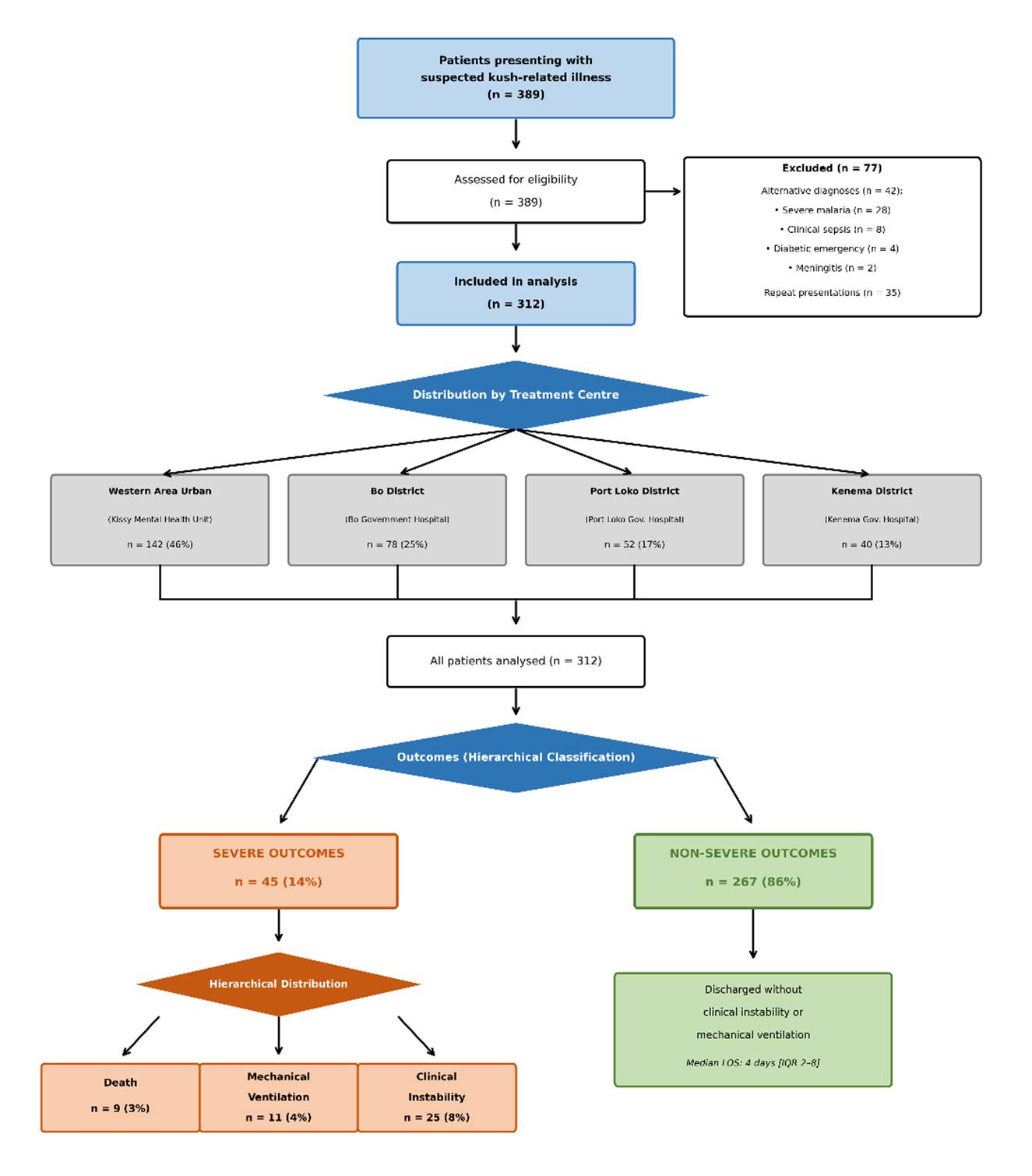
During the study period, 389 patients presented with suspected kush-related illness. After excluding 42 with alternative diagnoses (28 severe malaria, 8 clinical sepsis without kush use, 4 diabetic emergencies, 2 meningitis) and 35 repeat presentations, 312 unique patients meeting the case definition were included (Figure 1). The largest proportion presented to Western Area Urban District (142, 46%), followed by Bo District (78, 25%), Port Loko District (52, 17%), and Kenema District (40, 13%).
Demographic characteristics
Patients were predominantly young males with limited education and unstable employment (Table 1). Median age was 24 years (IQR 19–31), with 294 (94%) males. Educational attainment was low: 78 (25%) had no formal education, 131 (42%) had primary education only, 89 (29%) had secondary education, and 14 (4%) had tertiary education. The majority were unemployed (163, 52%) or engaged in informal work (87, 28%); 25 (8%) were students, and 37 (12%) had formal employment. Urban residence was reported by 241 (77%) patients.
Substance use characteristics
Polysubstance use was reported in 156 (50%) patients, most commonly involving alcohol (98, 31%), cannabis (67, 21%), and tramadol (34, 11%); categories were not mutually exclusive. Duration of kush use exceeding one year was reported by 89 (29%). Previous overdose episodes were reported by 89 (29%).
Clinical presentation
Clinical findings at presentation are summarised in Table 2. Presentation was consistent with opioid toxidrome: altered consciousness (GCS ≤14) in 234 (75%), miosis in 198 (63%), and respiratory compromise (RR <12/min or SpO₂ <92%) in 128 (41%). Median GCS was 11 (IQR 8–14); 67 (21%) presented with GCS <8. Respiratory rate <12/min was documented in 94 (30%) and SpO₂ <92% in 76 (24%), with 42 patients having both. Skin ulcerations, analysed as a comorbid condition, were present in 112 (36%) patients, predominantly affecting lower limbs. Wound infection complicated 98 (31%) of presentations; 23 (7%) met criteria for qSOFA ≥2.
Treatment and outcomes
Naloxone was administered to 186 (60%) patients, with response observed in 143/186 (77%). Among responders, 39/143 (27%) required repeat dosing within 2 hours. Supplemental oxygen was required in 156 (50%) patients. Median length of stay was 4 days (IQR 2–8).
Severe outcomes occurred in 45 (14%) patients, distributed hierarchically as: 9 (3%) deaths, 11 (4%) mechanical ventilation among survivors, and 25 (8%) clinical instability (GCS <8 or qSOFA ≥2) among those not ventilated. All deaths occurred within 72 hours of presentation; causes included respiratory failure (n=6), aspiration pneumonia (n=2), and sepsis (n=1). Severe outcome rates varied modestly by district: 22/142 (15%) in Western Area Urban, 12/78 (15%) in Bo, 7/52 (13%) in Port Loko, and 4/40 (10%) in Kenema.
Risk factors for severe outcome
Univariable and multivariable associations between pre-existing characteristics and severe outcome are presented in Table 3. Patients with severe outcomes were more likely to report polysubstance use (69% vs 47%), previous overdose (44% vs 26%), presentation >6 hours after use (51% vs 33%), and wound infection (47% vs 29%). In the mixed-effects logistic regression model, independent risk factors for severe outcome were: polysubstance use (adjusted odds ratio [aOR] 2.4, 95% CI 1.2–4.6, p=0.010), previous overdose (aOR 2.3, 95% CI 1.2–4.5, p=0.016), delayed presentation >6 hours (aOR 2.0, 95% CI 1.0–3.9, p=0.043), and wound infection (aOR 2.1, 95% CI 1.1–4.2, p=0.032). Age, sex, and education level were not significantly associated with outcome. Model discrimination was moderate (c-statistic 0.72, 95% CI 0.64–0.80), indicating the model reasonably distinguishes patients with and without severe outcomes. Calibration was adequate (Hosmer-Lemeshow χ²=6.2, p=0.62), confirming predicted probabilities match observed outcomes. The random intercept variance was 0.19 (SE 0.15), corresponding to an intraclass correlation coefficient (ICC) of 0.05, indicating modest clustering by centre. Complete-case analysis (n=278) yielded similar effect estimates (polysubstance use aOR 2.3 [95% CI 1.1–4.7], previous overdose aOR 2.2 [95% CI 1.1–4.6], delayed presentation aOR 1.9 [95% CI 0.9–3.8], wound infection aOR 2.0 [95% CI 1.0–4.3]).
Discussion
This multicentre study provides systematic clinical characterisation of suspected acute kush poisoning across four districts in Sierra Leone and identifies pre-existing and behavioural risk factors for severe outcomes. The 14% rate of severe outcomes and 3% mortality underscore the clinical significance of this emerging crisis.
The demographic profile—predominantly young males (median age 24 years, 94% male) with limited education (67% primary or less) and unstable employment (80% unemployed or informal work)—reflects the socioeconomic vulnerability of the affected population. The 8% who were students indicates that kush use extends into educational settings. These characteristics align with the phenomenological literature describing kush use concentrated among economically marginalised urban youth [1,2]. While demographic factors were not independently associated with severe outcomes in our analysis, they inform the targeting and design of prevention and treatment services.
Clinical presentation was consistent with opioid toxidrome, reflecting the nitazene content identified in chemical testing of Sierra Leonean kush samples [3,4]. Altered consciousness (75%), miosis (63%), and respiratory depression (41%) formed the classic triad. The 77% naloxone response rate supports the predominant opioid mechanism, while the 27% requiring repeat dosing aligns with the prolonged receptor binding and slow dissociation kinetics characteristic of nitazenes [6,7,16]. These clinical patterns can guide healthcare workers to recognise kush poisoning and initiate appropriate treatment, including naloxone with anticipation of repeat dosing.
Our finding that polysubstance use significantly increased the risk of severe outcomes has direct implications for harm reduction. Half of patients reported concurrent substance use, predominantly alcohol and cannabis, meaning individuals using multiple substances had more than double the likelihood of severe outcomes, including death or ventilation. Few clinical cohort studies of nitazene-associated poisoning have been reported [18], and this pattern is consistent with high-income settings where polysubstance involvement characterises most nitazene-related fatalities [8,9]. The pharmacological synergy of multiple CNS depressants increases the risk of respiratory failure. Harm reduction messaging should explicitly address risks of combining kush with alcohol or other substances.
Previous overdose as a risk factor identifies a population that might benefit from targeted naloxone distribution and overdose prevention education. The 29% prevalence of prior overdose indicates substantial recurrent risk in this population. Take-home naloxone programmes, successfully implemented in high-income settings for opioid use disorder, could be adapted for kush users and their social networks in Sierra Leone [18,19].
Delayed presentation was associated with severe outcome, likely because prolonged unconsciousness leads to respiratory compromise and allows time for complications such as aspiration. This finding supports community-based early recognition programmes and accessible treatment pathways to reduce time to care.
Wound infection as a risk factor highlights the burden of comorbidity in this population. The 36% prevalence of skin ulcerations—whose aetiology remains uncertain and may involve thermal injury, direct chemical effects, secondary infection, or poverty-related dermatoses—creates vulnerability to sepsis that compounds overdose risk. Integrated wound care within substance use treatment services addresses this compounding vulnerability.
Strengths and limitations
Strengths include multicentre design capturing presentations across four districts, standardised data collection with demonstrated inter-rater reliability, comprehensive demographic characterisation including education and occupation, and application of mixed-effects modelling to account for centre-level clustering.
Several limitations warrant consideration. First, absence of patient-level toxicological confirmation means we characterised suspected rather than confirmed kush poisoning; however, requiring both clinical toxidrome and reported kush use strengthens case ascertainment. Second, polysubstance use, previous overdose, and duration of use relied on self-report or proxy report, introducing potential recall bias; standardised abstractor training and inter-rater reliability assessment helped minimise measurement error. Third, treatment centre patients represent a selected population that reached care; inclusion of four geographically dispersed centres partially addresses this, but findings may not generalise to community overdoses. Fourth, the modest model discrimination (c-statistic 0.72) indicates that identified risk factors explain only part of outcome variability—unmeasured factors, including dose, specific drug composition, and clinical management, contribute substantially. Fifth, the relatively small number of severe outcomes (n=45) limits the precision of effect estimates. Finally, the observational design precludes causal inference.
Public health implications
These findings support several actionable public health priorities, though implementation will require addressing infrastructure limitations, supply chain constraints, and workforce capacity in Sierra Leone’s health system. First, naloxone distribution should target individuals with previous overdose history and their social networks, with repeat dosing protocols. Second, harm reduction messaging should address polysubstance risks. Third, accessible treatment pathways should minimise delays to care. Fourth, integrated services addressing substance use and wound care are needed. Fifth, services should target the young, male, economically marginalised population, including students. Sixth, surveillance should monitor evolving drug composition.
Community-based interventions are essential to extend the reach of facility-level findings. Community health workers should be trained in overdose recognition and basic supportive care. Community naloxone distribution should be established in high-burden areas with training for family and peers. Community-based wound care should be integrated into outreach programmes. Peer education networks can extend harm reduction messaging into underserved communities.
Conclusion
Acute kush poisoning in Sierra Leone presents with opioid toxidrome consistent with nitazene exposure and predominantly affects young males with limited education and unstable employment. Polysubstance use, previous overdose, delayed presentation, and wound infection are identifiable and potentially modifiable risk factors for severe outcomes. These findings support targeted naloxone distribution, harm reduction messaging addressing polysubstance risks, accessible treatment pathways, and integrated wound care within substance use services. Community-based interventions are essential to extend the reach of facility-level services. Continued clinical surveillance and toxicological monitoring are essential to track evolving drug composition and guide adaptive response strategies.
What is already known about the topic
- Kush is an illicit synthetic drug in Sierra Leone containing nitazene-class opioids with potency rivalling or exceeding fentanyl
- Sierra Leone declared kush a national public health emergency in April 2024 due to escalating deaths and morbidity
- Clinical characterisation of kush poisoning and risk factors for severe outcomes have not been systematically described in West Africa
What this study adds
- Provides the first multicentre clinical profile of suspected kush poisoning in Sierra Leone, documenting opioid toxidrome and 14% severe outcome rate
- Identifies modifiable risk factors (polysubstance use, previous overdose, delayed presentation, wound infection) that can guide clinical triage and prevention
- Supports evidence-based policies for targeted naloxone distribution, harm reduction messaging, and integrated wound care services
Acknowledgements
We thank the staff at Kissy Mental Health Unit, Bo Government Hospital, Port Loko Government Hospital, and Kenema Government Hospital for their support in data collection. We acknowledge the patients whose clinical records contributed to this study.
Data availability
Anonymised data are available upon reasonable request to the corresponding author with appropriate ethics approval.
Abbreviations
aOR: adjusted odds ratio
CI: confidence interval
FTIR: Fourier-transform infrared spectroscopy
GCS: Glasgow Coma Scale
ICC: intraclass correlation coefficient
IQR: interquartile range
LC–MS/MS: liquid chromatography–tandem mass spectrometry
qSOFA: quick Sequential Organ Failure Assessment
RR: respiratory rate
SpO₂: peripheral oxygen saturation
VIF: variance inflation factor
Authors´ contributions
Eric Nzirakaindi Ikoona: conceptualisation, methodology, formal analysis, writing—original draft. Lucy Namulemo: conceptualisation, methodology, data collection, data curation. Mary Magdelene Sinnah: methodology, data collection, data curation. Mohammad Alex Vandi: methodology, supervision, writing—review and editing. Foday Sahr: conceptualisation, supervision, writing—review and editing. All authors read and approved the final manuscript.
| Table 1: Demographic, geographic, and substance use characteristics (N=312) | |
|---|---|
| Characteristic | n (%) or median [IQR] |
| Treatment centre (District) | |
| Kissy Mental Health Unit (Western Area Urban) | 142 (46) |
| Bo Government Hospital (Bo) | 78 (25) |
| Port Loko Government Hospital (Port Loko) | 52 (17) |
| Kenema Government Hospital (Kenema) | 40 (13) |
| Demographics | |
| Age, years | 24 [19–31] |
| Male sex | 294 (94) |
| Education level | |
| None | 78 (25) |
| Primary | 131 (42) |
| Secondary | 89 (29) |
| Tertiary | 14 (4) |
| Occupation | |
| Unemployed | 163 (52) |
| Informal work | 87 (28) |
| Formal employment | 37 (12) |
| Student | 25 (8) |
| Residence | |
| Urban | 241 (77) |
| Rural | 71 (23) |
| Substance use history | |
| Duration of kush use >1 year | 89 (29) |
| Previous overdose episode | 89 (29) |
| Polysubstance use* | 156 (50) |
| Alcohol | 98 (31) |
| Cannabis | 67 (21) |
| Tramadol | 34 (11) |
| Presentation | |
| Time from use to presentation >6 hours† | 112/290 (39) |
| *Substance categories not mutually exclusive. †22 records missing time data; percentage based on valid responses (n=290). IQR = interquartile range. | |
| Table 2: Clinical presentation of cases among suspected kush poisoning, Sierra Leone, June-November 2025 (N=312) | |
|---|---|
| Clinical finding | n (%) or median [IQR] |
| Neurological findings | |
| Altered consciousness (GCS ≤14) | 234 (75) |
| Glasgow Coma Scale score | 11 [8–14] |
| GCS <8 (severe obtundation) | 67 (21) |
| Miosis | 198 (63) |
| Respiratory findings | |
| Respiratory compromise (composite)* | 128 (41) |
| Respiratory rate <12/min | 94 (30) |
| SpO₂ <92% | 76 (24) |
| Skin findings (comorbid condition) | |
| Skin ulcerations present | 112 (36) |
| Lower limb† | 78 (70) |
| Upper limb† | 45 (40) |
| Wound infection | 98 (31) |
| qSOFA ≥2 | 23 (7) |
| Treatment | |
| Naloxone administered | 186 (60) |
| Response to naloxone‡ | 143 (77) |
| Repeat dosing required‡ | 39 (27) |
| Supplemental oxygen required | 156 (50) |
| *RR <12/min OR SpO₂ <92%; 42 had both. †Among 112 with ulcerations; not mutually exclusive. ‡Among naloxone recipients (n=186 for response; n=143 for repeat dosing). GCS = Glasgow Coma Scale; qSOFA = quick Sequential Organ Failure Assessment. | |
| Table 3: Risk factors for severe outcome—univariable and multivariable analysis (N=312) | |||||
|---|---|---|---|---|---|
| Characteristic | Severe n=45 (%) | Non-severe n=267 (%) | p-value | aOR (95% CI) | Adj. p |
| Age, median [IQR] | 26 [20–33] | 24 [19–30] | 0.12* | 1.2 (0.8–1.8)† | 0.34 |
| Male sex | 43 (96) | 251 (94) | 0.67‡ | 1.4 (0.3–6.2) | 0.67 |
| Education ≤primary | 32 (71) | 177 (66) | 0.52 | 1.2 (0.6–2.4) | 0.62 |
| Polysubstance use | 31 (69) | 125 (47) | 0.006 | 2.4 (1.2–4.6) | 0.010 |
| Previous overdose | 20 (44) | 69 (26) | 0.014 | 2.3 (1.2–4.5) | 0.016 |
| Duration >1 year | 18 (40) | 71 (27) | 0.07 | 1.8 (0.9–3.6) | 0.09 |
| Presentation >6 h | 23 (51) | 89 (33) | 0.015 | 2.0 (1.0–3.9) | 0.043 |
| Wound infection | 21 (47) | 77 (29) | 0.019 | 2.1 (1.1–4.2) | 0.032 |
| Bold indicates statistically significant independent risk factors (adjusted p<0.05). aOR = adjusted odds ratio from mixed-effects logistic regression with random intercept for treatment centre. *Mann-Whitney U test. †Per 10-year increase. ‡Fisher’s exact test. Model: C-statistic 0.72 (95% CI 0.64–0.80); Hosmer-Lemeshow χ²=6.2, p=0.62; ICC 0.05; all VIF <2.0. Sensitivity analysis (complete-case, n=278): polysubstance use aOR 2.3 (95% CI 1.1–4.7), previous overdose aOR 2.2 (95% CI 1.1–4.6), delayed presentation aOR 1.9 (95% CI 0.9–3.8), wound infection aOR 2.0 (95% CI 1.0–4.3). | |||||

Abbreviations: GCS = Glasgow Coma Scale; qSOFA = quick Sequential Organ Failure Assessment; LOS = length of stay; IQR = interquartile range.
Hierarchical outcome: Patients classified by the most severe outcome achieved
(death > ventilation > clinical instability [GCS < 8 or qSOFA ≥ 2]).
Flow diagram showing patient enrolment from initial presentations (n=389) through exclusions
(alternative diagnoses n=42; repeat presentations n=35) to final cohort (n=312), distributed across
four treatment centres (Western Area Urban n=142, Bo n=78, Port Loko n=52, Kenema n=40), with
hierarchical severe outcomes (death n=9; mechanical ventilation n=11; clinical instability n=25)
and non-severe outcomes (n=267).
References
- Lahai M, Vandy A, Turay A, Kolipha-Kamara M, Conteh E. Synthetic Cannabinoids in Sierra Leone: Understanding the Use of ‘Kush’ Among Youths and Its Socioeconomic Impact in Sierra Leone and Sub-Region. Public Health Challenges [Internet]. 2025 Feb 6 [cited 2026 Feb 23];4(1):e70031. doi:10.1002/puh2.70031.
- Ikoona E, Namulemo L, Kaluya R, Vandi MA, Sahr F, Kitara D. Lived Experiences and Recovery Challenges of Youth Facing the KUSH Crisis in Sierra Leone: A Qualitative Study [Internet]. 2025 Sep 30 [cited 2026 Feb 23]. Available from: https://www.ssrn.com/abstract=5543304 doi:10.2139/ssrn.5543304.
- Bird L, de Bruijne K. Kush in Sierra Leone: West Africa’s growing synthetic drugs challenge [Internet]. Geneva (Switzerland): Global Initiative; 2025 Feb 25 [cited 2026 Feb 23]. Available from: https://globalinitiative.net/analysis/kush-in-sierra-leone/.
- Global Initiative Against Transnational Organized Crime. Kush: FTIR spectrometer testing indicates presence of synthetic cannabinoids and nitazenes in Freetown and Bissau [Internet]. Geneva (Switzerland): Global Initiative; 2024 Jun 12 [cited 2026 Feb 23]. Available from: https://globalinitiative.net/analysis/kush-testing-drug-markets-sierra-leone-guinea-bissau/.
- Bendjilali-Sabiani JJ, Eiden C, Lestienne M, Cherki S, Gautre D, Van den Broek T, Mathieu O, Peyrière H.Isotonitazene, a synthetic opioid from an emerging family: The nitazenes. Therapies [Internet]. 2024 [cited 2026 Feb 23];79(6):655–658. doi: 10.1016/j.therap.2024.05.004
- Vandeputte MM, Stove CP. Navigating nitazenes: A pharmacological and toxicological overview of new synthetic opioids with a 2-benzylbenzimidazole core. Neuropharmacology [Internet]. 2025 Apr 17 [cited 2026 Feb 23];275:110470. doi:10.1016/j.neuropharm.2025.110470.
- Glatfelter GC, Vandeputte MM, Chen L, Walther D, Tsai MHM, Shi L, Stove CP, Baumann MH. Alkoxy chain length governs the potency of 2-benzylbenzimidazole ‘nitazene’ opioids associated with human overdose. Psychopharmacology [Internet]. 2023 Dec 1 [cited 2026 Feb 23];240(12):2573–84. doi:10.1007/s00213-023-06451-2.
- Vandeputte MM, Krotulski AJ, Walther D, Glatfelter GC, Papsun D, Walton SE, Logan BK, Baumann MH, Stove CP. Pharmacological evaluation and forensic case series of N-pyrrolidino etonitazene (etonitazepyne), a newly emerging 2-benzylbenzimidazole ‘nitazene’ synthetic opioid. Archives of Toxicology [Internet]. 2022 Jun[cited 2026 Feb 23];96(6):1845–1863. doi:10.1007/s00204-022-03276-4.
- Montanari E, Madeo G, Pichini S, Busardò FP, Carlier J. Acute Intoxications and Fatalities Associated With Benzimidazole Opioid (Nitazene Analog) Use: A Systematic Review. Ther Drug Monit [Internet]. 2022 Aug [cited 2026 Feb 23];44(4):494–510. doi:10.1097/FTD.0000000000000970.
- Fofana U. Sierra Leone declares emergency over drug kush – made from human bones. BBC News [Internet]. 2024 Apr 5 [cited 2026 Feb 23]. Available from: https://www.bbc.com/news/world-africa-68742694.
- Government of Sierra Leone. The Constitution of Sierra Leone (Emergency Powers) Regulations, 2024: Constitutional Instrument No. 5 of 2024 [Internet]. Freetown (Sierra Leone): Government of Sierra Leone; 2024 Apr 9 [cited 2026 Feb 23]. Available from: https://www.parliament.gov.sl/uploads/statutory_instruments/Constitutional%20Instrument%20No.%205%20of%202024%20Gazette%20VO1.%20CLXV%2C%20NO.%2031.pdf.
- World Health Organization Regional Office for Africa. Tackling substance abuse: WHO supports efforts to combat the Kush epidemic in Sierra Leone [Internet]. Geneva (Switzerland): World Health Organization; 2024 Apr 19 [cited 2026 Feb 23]. Available from: https://www.afro.who.int/countries/sierra-leone/news/tackling-substance-abuse-who-supports-efforts-combat-kush-epidemic-sierra-leone.
- Rhodes T. The ‘risk environment’: a framework for understanding and reducing drug-related harm. International Journal of Drug Policy[Internet]. 2002[cited 2026 Feb 23];13(2):85–94. doi:10.1016/S0955-3959(02)00007-5.
- Ministry of Health (Sierra Leone). Clinical guidelines for the management of acute substance intoxication: With Special Focus on Kush (Nitazene-Class Synthetic Opioids) [Internet]. Freetown (Sierra Leone): Ministry of Health; 2024 [cited 2026 Feb 23]. Available from: https://example.org/clinical-guidelines-intoxication-kush.pdf.
- Elm EV, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. Strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ [Internet]. 2007 Oct 20 [cited 2026 Feb 23];335(7624):806–8. doi:10.1136/bmj.39335.541782.AD.
- Dahan A, Aarts L, Smith TW. Incidence, Reversal, and Prevention of Opioid-induced Respiratory Depression. Anesthesiology [Internet]. 2010 Jan 1 [cited 2026 Feb 23];112(1):226–38. doi:10.1097/ALN.0b013e3181c38c25.
- Roberts DM, Tisdell B, Sajeev MF, Jiranantakan T, Harvey C, Brown JA. Clinical Experiences with the Nitazene Class of Synthetic Opioids: A Cohort Study. Ann Emerg Med [Internet]. 2025 Nov [cited 2026 Feb 23];86(5):475–83. doi:10.1016/j.annemergmed.2025.06.619.
- World Health Organization. Opioid overdose [Internet]. Geneva (Switzerland): World Health Organization; 2025 Aug 29 [cited 2026 Feb 23]. Available from: https://www.who.int/news-room/fact-sheets/detail/opioid-overdose.
- World Health Organization. Community management of opioid overdose [Internet]. Geneva (Switzerland): World Health Organization; 2014 Nov 2 [cited 2026 Feb 23]. Available from: https://www.who.int/publications/i/item/9789241548816.
