Outbreak Investigation | Open Access | Volume 9 (1): Article 40 | Published: 06 Mar 2026
Anthrax outbreak linked to handling and consumption of infected meat, Sembabule District, Uganda, October–December 2024
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Exposure (n=19) | Frequency | % |
|---|---|---|
| Ate meat from animals that died suddenly | 15 | 79 |
| Participated in butchering dead animals | 11 | 58 |
| Carried meat home after butchering dead animals | 8 | 42 |
| Owned cattle | 5 | 26 |
| Moved out of Sembabule District since start of the outbreak | 3 | 16 |
Table 1: Possible risk factors associated with developing anthrax in Sembabule District, Uganda, December 2024
| Cases, n=45 | Controls, n=153 | ||
|---|---|---|---|
| Characteristic (n=198) | n(%) | n(%) | p-value |
| Age group (years) | |||
| <18 | 5 (11) | 18 (12) | 1 |
| ≥18 | 40 (89) | 135 (88) | |
| Sex | |||
| Female | 6 (13) | 26 (17) | |
| Male | 39 (87) | 127 (83) | 0.004 |
| Animal ownership | |||
| Yes | 17 (38) | 80 (52) | 0.002 |
| No | 28 (62) | 73 (48) | |
| Sub-county of residence | |||
| Kyera | 9 (20) | 39 (25) | |
| Lwemiyaga | 34 (76) | 105 (69) | 0.036 |
| Ntusi Town Council | 2 (4) | 9 (6) | |
| Livelihood | |||
| Livestock/meat-related occupation* | 32 (71) | 54 (35) | <0.001 |
| Other occupations | 13 (29) | 99 (65) | |
| *butchers, herdsmen, meat traders, meat transporters, slaughterers, meat roasters, and skinners | |||
| p-value calculated using χ² test | |||
| Exposure | Number of participants n (%) | cOR | aOR | p-Value | |
|---|---|---|---|---|---|
| Case (%) | Controls (%) | ||||
| Consumed meat from suddenly dead animal | |||||
| No | 11 (24) | 117 (74) | Ref | Ref | <0.001 |
| Yes | 34 (76) | 42 (26) | 8.6 (4.0-18.5) | 10.3 (3.5-29.7) | |
| Butchered/Handled a dead cow | |||||
| No | 12 (27) | 133 (84) | Ref | Ref | <0.001 |
| Yes | 33 (73) | 26 (16) | 14.1 (6.4-31.0) | 18.8 (3.3-107.3) | |
| Flaying/Skinning | |||||
| No | 30 (67) | 153 (96) | Ref | Ref | 0.05 |
| Yes | 15 (33) | 6 (4) | 12.8 (4.6-35.5) | 6.7 (1.0-45.2) | |
| Carried meat during butchery | |||||
| No | 23 (51) | 139 (87) | Ref | Ref | 0.00875 |
| Yes | 22 (49) | 20 (13) | 6.5 (3.2-14.1) | 0.25 (0.1-6.1.2) | |
| Carcass dressing | |||||
| No | 26 (58) | 153 (96) | Ref | Ref | 0.03 |
| Yes | 19 (42) | 6 (4) | 18.6 (6.8-51.1) | 5.9 (1.2-28.6) | |
| Cutting | |||||
| No | 26 (58) | 146 (92) | Ref | Ref | 0.583 |
| Yes | 19 (42) | 13 (9) | 8.2 (3.6-18.6) | 0.6 (0.1-3.1) | |
Figures
Keywords
- Anthrax
- Outbreak investigation
- Zoonotic diseases
- Uganda
Hannington Katumba1,&, Emmanuel Mfitundinda1, Gertrude Abbo1, Richard Migisha1, Jane Frances Zalwango1, Tom Ndaga2, Benon Kwesiga1, Lilian Bulage1, John Opolot3, Stella Atim4, Linda Atiku5, Alex Riolexus Ario1,3
1Uganda Public Health Fellowship Program-Field Epidemiology Track, Uganda National Institute of Public Health, Kampala, Uganda, 2Sembabule District Local Government, Sembabule, Uganda, 3Ministry of Health, Kampala, Uganda, 4Ministry of Agriculture, Animal Industry and Fisheries, Entebbe, Uganda, 5Uganda Virus Research Institute, Arua, Uganda
&Corresponding author: Hannington Katumba, Uganda Public Health Fellowship Program-Field Epidemiology Track, Uganda National Institute of Public Health, Kampala, Uganda. Email: hkatumba@uniph.go.ug ORCID: https://orcid.org/0009-0004-9552-097X
Received: 05 Nov 2025, Accepted: 27 Feb 2026, Published: 06 Mar 2026
Domain: Zoonosis, One Health
Keywords: Anthrax, Outbreak investigation, Zoonotic diseases, Uganda
©Hannington Katumba et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Hannington Katumba et al., Anthrax outbreak linked to handling and consumption of infected meat, Sembabule District, Uganda, October–December 2024. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):40. https://doi.org/10.37432/jieph-d-25-00271
Abstract
Introduction: Despite multiple outbreaks annually, Uganda still lacks a national anthrax control program. While the surrounding districts of Gomba, Kazo, and Kiruhura had experienced anthrax outbreaks previously, Sembabule had not documented any. On December 4, 2024, four meat dealers in Sembabule District developed skin lesions and were confirmed to have anthrax. We conducted an investigation to assess the magnitude of the outbreak, identify risk factors, and recommend control measures.
Methods: Suspected cutaneous anthrax was characterised by the onset of skin lesions (papule, vesicle, or eschar) in a resident of Sembabule District from October to December 2024. Suspected gastrointestinal anthrax was the acute-onset abdominal pain with diarrhea, vomiting, pharyngitis, or oropharyngeal lesions. Suspected animal anthrax was the sudden death of a domestic herbivore with bloody discharge from natural orifices. Laboratory samples were collected from a subset of suspected cases to confirm the outbreak etiology. A confirmed case was any suspected case with a clinical sample positive for Bacillus anthracis by polymerase chain reaction (PCR). We identified human cases by reviewing medical records and community case search. We explored risk factors through interviews with farmers and health workers. We compared exposures among 40 human cases and 158 neighbourhood controls to identify risk factors for anthrax.
Results: Among 45 human cases (36 suspected, 9 confirmed), two (4.4%) died. Thirty-four (76%) had cutaneous anthrax, five (11%) gastrointestinal anthrax, and six (13%) had both forms. Males were more affected than females (24 vs 4/100,000). Among 25 animal cases, two (8.0%) were confirmed. Infected cattle were introduced into Sembabule District from Kazo District in October 2024 and mixed with herds in Sembabule. When these animals died, meat dealers distributed the meat, selling it at one-third the normal price. Butchering (OR=13.3, 95%CI:5.8-30.7), carrying (OR=6.3, 95%CI:2.8-13.8), and eating meat from dead animals (OR=8.7, 95%CI:3.8-20) significantly increased infection risk.
Conclusion: The anthrax outbreak in Sembabule District was triggered by the introduction of sick cattle and spread by butchery and distribution of infected meat. Given the frequent outbreaks in this region of Uganda, a One Health approach, including livestock vaccination and sustained risk communication to change community practices, may be needed to facilitate anthrax control
Introduction
Anthrax, caused by the bacteria Bacillus anthracis, is a zoonotic disease that mainly affects herbivorous animals such as cattle, goats, and sheep. Human infection usually occurs through direct contact with infected animals or their products, ingestion of contaminated meat, or, less commonly, through inhalation of B. anthracis spores. The disease has three forms with different incubation periods: cutaneous anthrax (average: 5–7 days, range: 1–12 days), gastrointestinal anthrax (incubation: 1–6 days), and inhalation anthrax (average: 1–43 days, range: 1–60 days). While anthrax is a significant global public health concern, its impact is dire in sub-Saharan Africa, where the disease remains endemic, and outbreaks are frequent [1, 2].
Anthrax remains one of Uganda’s priority zoonotic diseases, but it lacks a dedicated, structured control program, leaving communities vulnerable to repeated outbreaks and ongoing transmission risks. Vaccines for anthrax are available, and livestock vaccination is the most effective way to prevent and control the disease in animals. In Uganda, anthrax outbreaks have been reported more frequently over the past decade (2014–2024), especially in the “cattle corridor,” which runs across the central and northeastern regions of the country. This area is characterized by a semi-arid climate and pastoralist communities whose livelihoods heavily depend on livestock farming. The districts within this corridor have historically experienced multiple outbreaks, resulting in economic losses, human health issues, and occasional deaths. However, there has been no reported human or animal anthrax outbreak in Sembabule District.
On November 26, 2024, the area veterinarian for Lwemiyaga Subcounty reported the sudden death of cattle to the Sembabule District Health Team (DHT) through the District Veterinary Officer (DVO). On December 4, 2024, meat vendors were admitted to Lwemiyaga Health Center III with skin lesions, fever, loss of appetite, cough, and swollen lymph nodes, which suggested anthrax. This prompted an alert to the Masaka Regional Emergency Operations Center (REOC). Laboratory samples were collected from the patients on the same day and sent to the Uganda Virus Research Institute (UVRI) for testing. Results returned on December 10, 2024, confirming an anthrax outbreak in the district. The MoH was notified on December 11, 2024, and the National Rapid Response Team (NRRT) was deployed on December 16, 2024, to assist Sembabule District with an epidemiological investigation. We conducted the investigation to identify the source of the outbreak, estimate its magnitude, determine risk factors for its spread, and recommend evidence-based control and prevention measures.
Methods
Outbreak setting
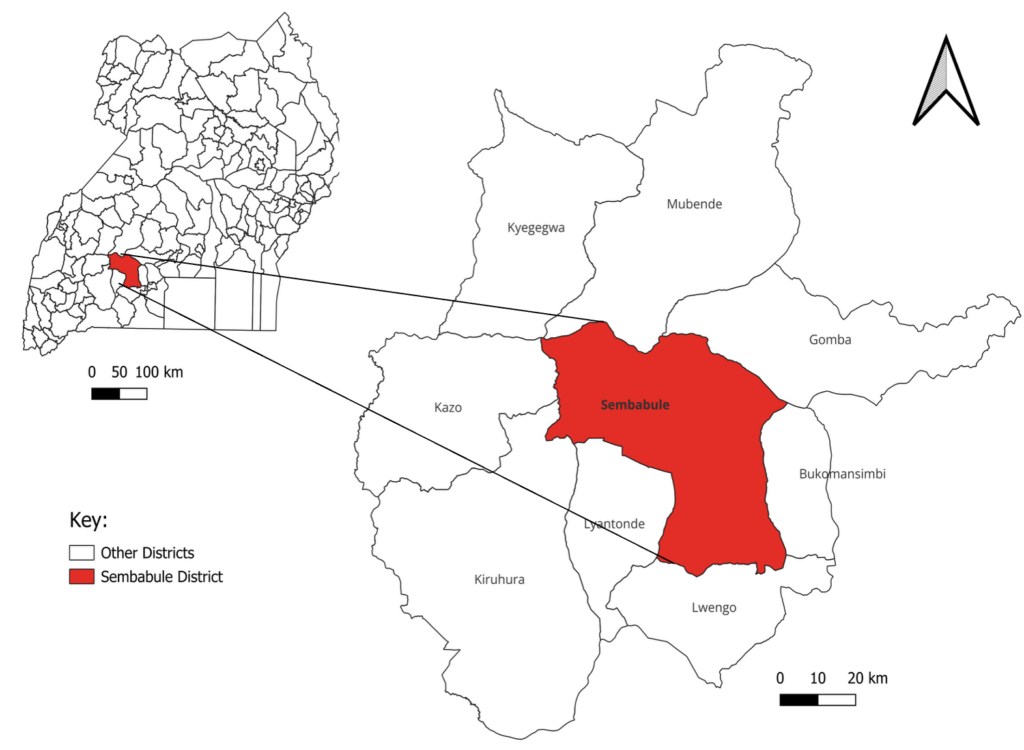
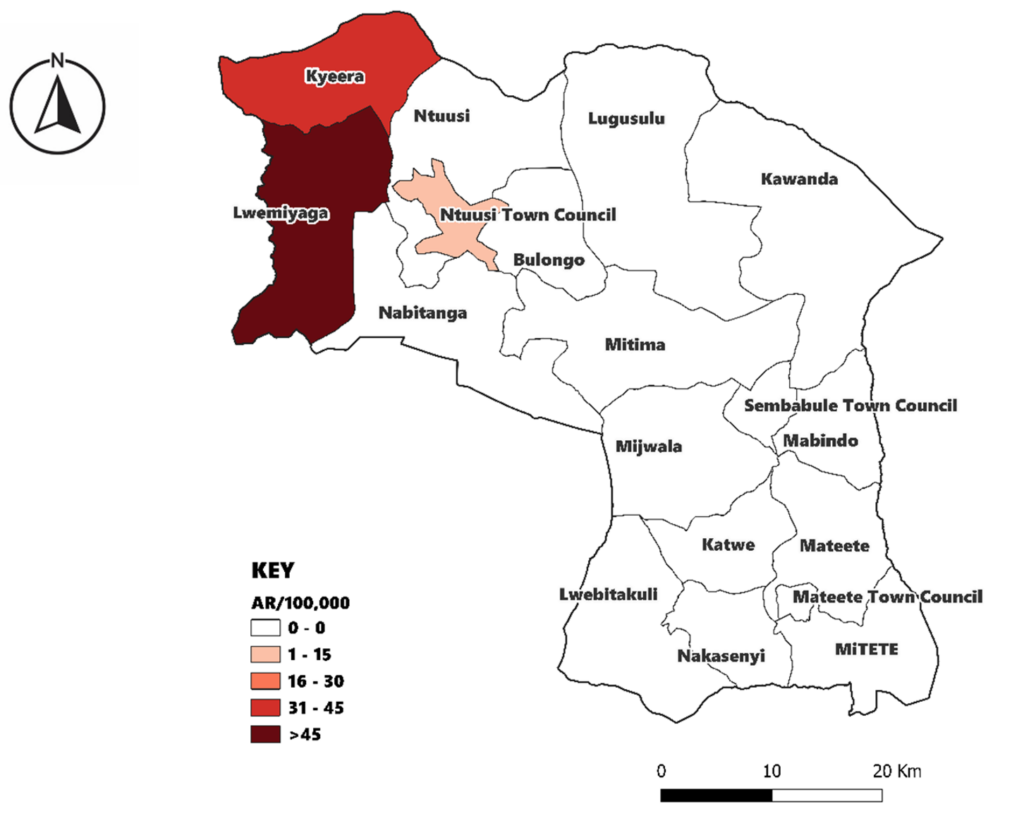
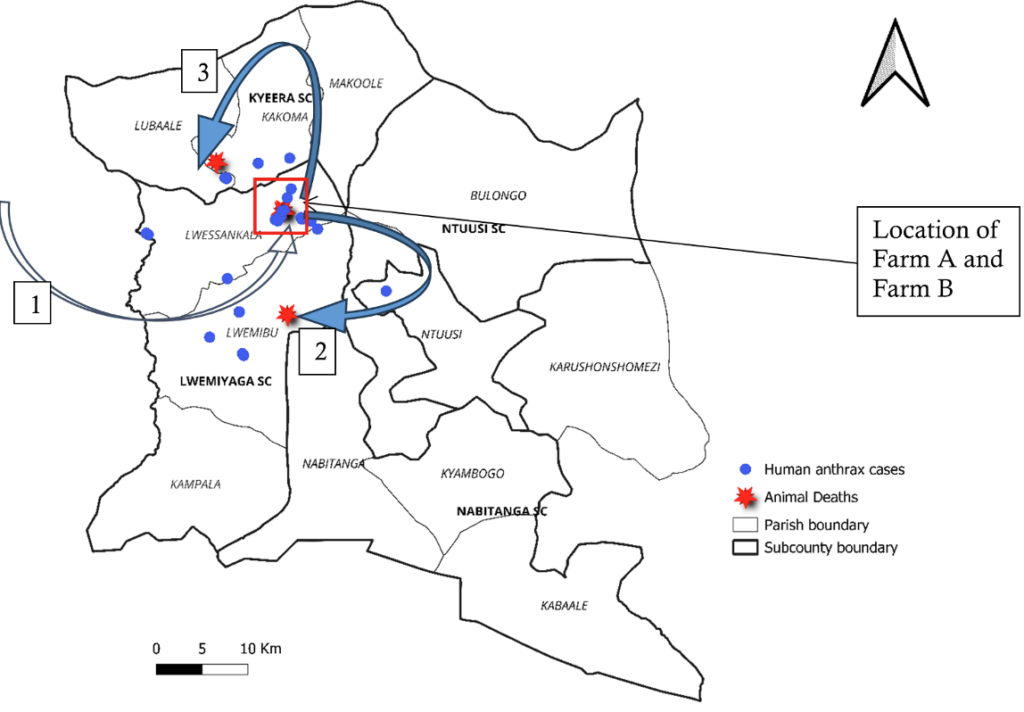
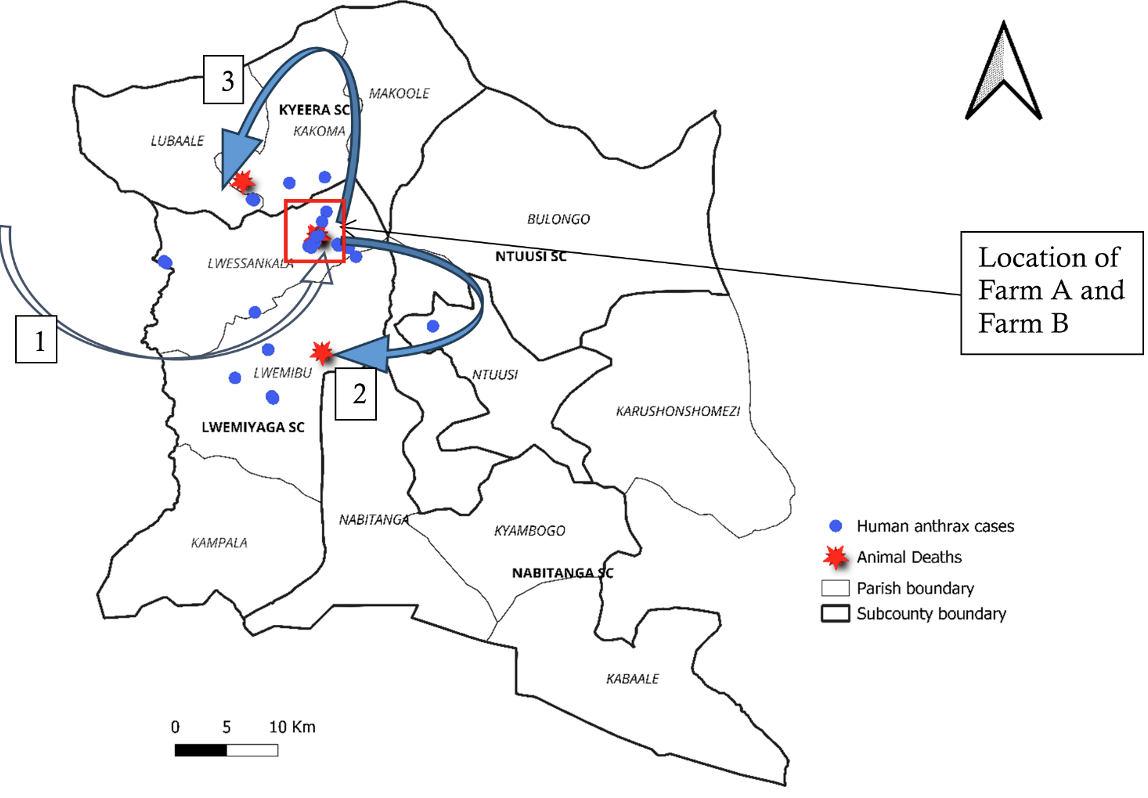
This outbreak occurred in Sembabule District, located in South-Central Uganda, encompassing 17 sub-counties and a population of 305,971. The main economic activity is agropastoralism, with a livestock population of 234,520 cattle, 128,261 goats, and 25,271 sheep, making it one of the highest milk-producing districts in Uganda. The district is bordered by eight districts: Bukomansimbi, Gomba, Lwengo, Lyantonde, Kazo, Kiruhura, Kyegegwa, and Mubende (Figure 1).
Case definitions
We defined a suspected cutaneous anthrax case as the onset of skin itching or swelling with any of the following: skin lesion (papule, vesicle, or eschar) or lymphadenopathy in a resident of Sembabule District from October to December 2024; a suspected gastrointestinal anthrax as the onset of abdominal pain and at least two of the following: diarrhea, vomiting, lymphadenopathy, pharyngitis, or oropharyngeal lesions in a person residing in Sembabule District from October to December 2024; and a confirmed human anthrax case as a suspected case with laboratory confirmation of Bacillus anthracis by PCR.
A suspected animal anthrax case was the sudden death of a domestic herbivore in Sembabule District from October to December 2024. A probable animal anthrax case was defined as a suspected case with unclotted blood emerging from body orifices and weak or no rigor mortis in Sembabule District during the same period. A confirmed animal anthrax case was any suspected or probable case with a positive PCR test for Bacillus anthracis.
Case finding and verification
This involved systematically identifying cases by reviewing medical records at Lwemiyaga HC III and noting all diagnoses consistent with anthrax from October to December 2024. Additional cases in the community were identified through active case searches and snowball sampling, in collaboration with the surveillance focal person (SFP), Community Health Workers (CHWs), Animal Health Workers (AHWs), and community leaders. Identified cases were verified through structured interviews to confirm symptoms and exposure histories, including consumption of meat from animals that died suddenly during the outbreak. Laboratory confirmation was not possible for recovered GI cases; specimens were only collected from individuals with cutaneous anthrax. A respondent was categorized as having both cutaneous and gastrointestinal anthrax if medical records, direct observations, and self-reported histories indicated consumption of meat from animals that died during the outbreak, followed by gastrointestinal symptoms. We created a line list capturing person, place, and time characteristics.
Environmental Assessment
We assessed the environment for potential factors that could facilitate the introduction and spread of anthrax in livestock and humans. We visited the affected farms and communities, assessing their farm production and biosecurity practices. These included grazing, watering, disease control, such as vaccination, and isolation of new or sick animals.
Laboratory investigations
Cases identified in the community were referred to Lwemiyaga Health Center III where laboratory samples were collected. We collected 15 human, two animal, and one environmental samples, and sent them to the Uganda Virus Research Institute (UVRI) and the National Animal Disease Diagnostics and Epidemiology Centre (NADDEC) for testing.
Descriptive epidemiology
We described the cases by animal/person, place, and time. Attack rates were calculated as the number of cases divided by the respective population at risk, multiplied by 100,000. For overall attack rates, the total district population was used as the denominator, while sex-specific and age-specific attack rates used the respective sex and age group populations obtained from district records. We described the cases by clinical manifestation and presented the clinical manifestations and constructed choropleth and spot maps to show the distribution of cases among the affected communities. We plotted an epidemic curve to describe the time component of the outbreak based on the dates of symptom onset for human cases and death for animal cases. We based our analysis on the incubation period of anthrax to determine the earliest epidemiologically linked human and animal cases, thus estimating when the outbreak likely started.
Hypothesis-generating interviews
We interviewed 19 community members to gather information about potential factors that contributed to the introduction of the disease and possible exposures. These included occupation, handling, and butchering of animals that died suddenly, consumption of meat from such animals, and how the meat was prepared before eating. We also conducted in-depth interviews with key informants, including farm owners, Village Health Teams (VHTs), health workers, veterinarians, meat dealers, and community leaders, to discuss key events, dates, and potential risk factors for the introduction and spread of anthrax in the community.
Case-control study
We conducted a case-control study in the affected subcounties of Lwemiyaga, Kyera, and Ntusi to test our hypothesis. To ensure geographic comparability and minimise differences in risk of exposure, controls were randomly selected from the same neighbourhood as the cases at a ratio of 1:4. The study included all 45 identified cases and 153 controls. To control for potential confounders such as environmental factors, occupation, and socioeconomic status, controls were chosen from the same subcounties where the cases resided. We defined a control as any resident of the subcounties of Lwemiyaga, Kyera, and Ntusi in Sembabule District, aged at least 15, who did not develop symptoms of cutaneous and/or gastrointestinal anthrax from October 2024 to December 2024. To further ensure that the controls resembled the cases, in each household, any member aged at least 15 who was present at home was eligible and was interviewed upon giving consent. We used a structured questionnaire administered to both cases and controls to gather demographic information such as age, sex, residence, and occupation. The tool was adapted from an investigation of a similar outbreak in Uganda [3] with modifications to reflect specific potential exposures and the context of this outbreak, including butchering and consumption of meat from animals that had died suddenly, as well as how such meat was prepared before eating. The exposure period was defined as any time from 1 to 12 days before symptom onset. Data were collected using Kobo Toolbox, cleaned in Microsoft Excel, and analyzed with Epi Info (CDC, Atlanta, USA).
Data analysis
We performed a descriptive analysis by computing frequencies and proportions of different exposures. Using logistic regression, we conducted a multivariable analysis to determine the level of association between individual exposures and contracting anthrax. Variables with a p-value < 0.2 at the bivariate level, as well as those deemed biologically relevant, were included in the final multivariable model from which we reported adjusted odds ratios (aORs) and their 95% confidence intervals.
Ethical considerations
This study was conducted in response to a public health emergency and as such it was determined to be non-research. As such, the MoH authorized this study and waved the requirement for IRB approvals and written consent from participants. The study was also approved by the Office of the Associate Director for Science, Center for Global Health, US Centers for Disease Control and Prevention (CDC) determined that this activity was not human subject research and with its primary intent being for public health practice or disease control. This activity was reviewed by CDC and was conducted consistent with applicable federal law and CDC policy. §§See e.g., 45 C.F.R. part 46, 21 C.F.R. part 56; 42 U.S.C. §241(d); 5 U.S.C. §552a; 44 U.S.C. §3501 et seq. 163 8 | Page 164. We obtained verbal consent from all the respondents before the interviews. All participants were duly informed that their participation was voluntary and assured that there would be no negative consequences if they declined to participate in the interview. All case-patients identified during case-finding were referred to the Lwemiyaga HC III for appropriate management. Data collected did not contain any individual personal identifiers, and information was stored in password-protected computers, which were inaccessible to anyone outside the investigation team.
Results
Descriptive epidemiology
We identified a total of 45 cases (36 suspects and nine confirmed cases) with an overall attack rate of 14/100,000 and a case fatality rate (CFR) of 4.4% (n=2). Males were more affected, with an attack rate of 25/100,000, compared to females, with an attack rate of 4/100,000. Individuals aged 40-49 years were most affected with an attack rate of 99/100,000, followed by those aged 50-59 years with an attack rate of 65/100,000.
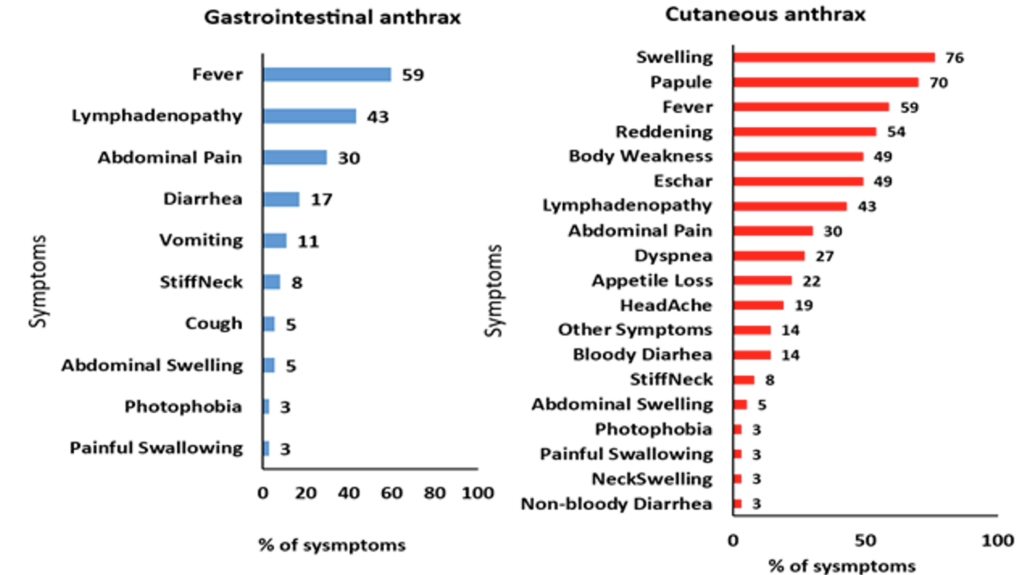
There were 34 (76%) cases of cutaneous, 5 (11%) gastrointestinal, and 6 (13%) cases with both forms of anthrax. Most cases of gastrointestinal anthrax presented with fever, lymphadenopathy, abdominal pain, diarrhea, and vomiting, while most cases of cutaneous anthrax presented with skin lesions, fever, body weakness, and lymphadenopathy (Figure 2).
The affected subcounties were Lwemiyaga, Kyera, and Ntuusi. Among these, Lwemiyaga was the most affected with 34 cases (AR=162/100,000). Kyera had 9 cases (AR=36/100,000), and Ntuusi was least affected with 2 cases (AR=14/100,000) (Figure 3).
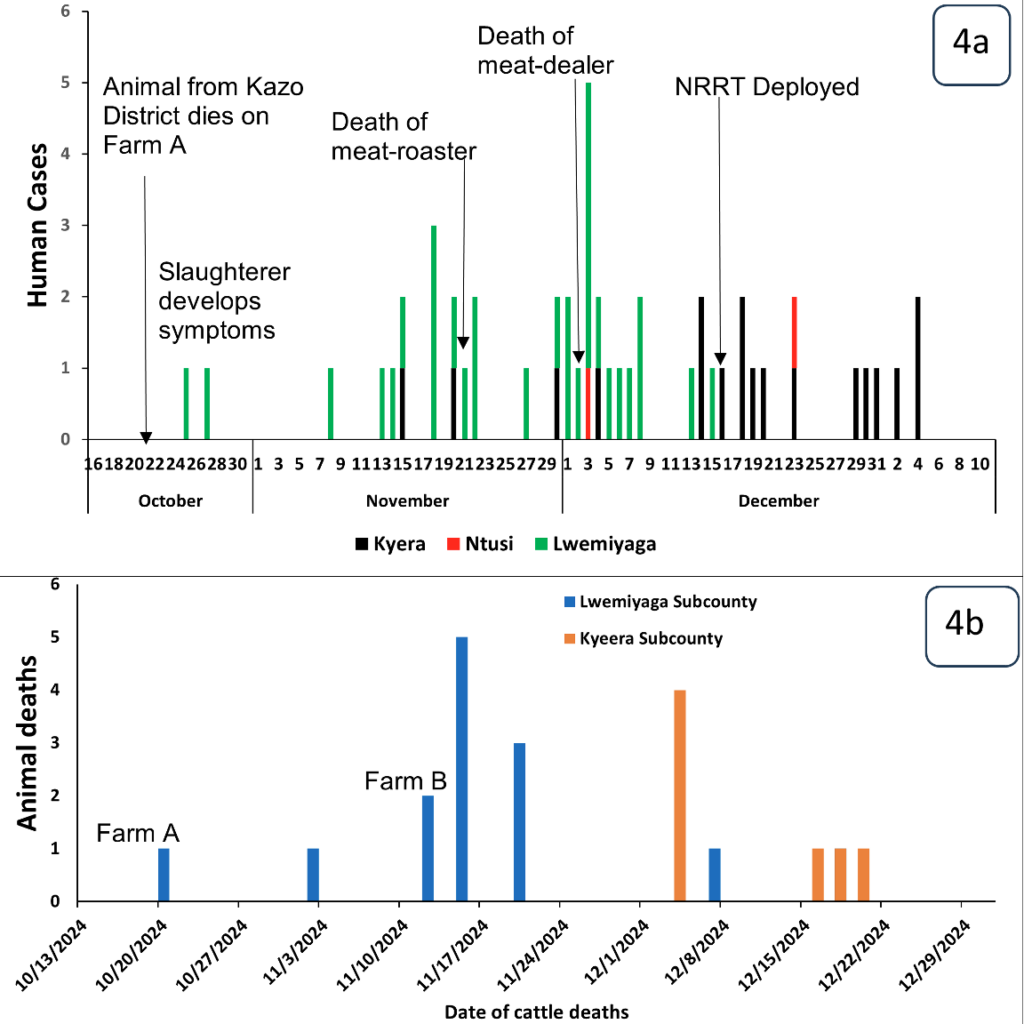
On October 18, 2024, Farm A in Lwensakala Parish, Lwemiyaga Subcounty received two cattle from another farm in Kazo District. These cows were immediately mixed with the rest of the herd on the farm, without following recommended farm biosecurity practices, including isolating the new livestock for at least 14-21 days. One of the new cows died 2 days later, on October 20, 2024, and was butchered for consumption. One community member who led the butchering of this cow developed symptoms of anthrax after a week on October 27, 2024. The meat from this butchering activity was bought by meat roasters, meat dealers, and community members.
On November 2, 2024, this farm lost a second cow, and later, due to continued mixing of herds from different villages in a communal grazing area and at a watering point at the periphery of this farm, other animal deaths were reported in farm A, and also in farm B in Nyendo, a neighboring village. All the dead cattle were butchered, and the meat was sold to meat roasters, meat dealers, restaurants, and the communities in Lwensankala and Lwemibu Parishes. Later, on November 18, 2024, a meat dealer who was also involved in meat roasting developed symptoms of anthrax and died, 2 days later, on November 22, 2024.
More cattle deaths were then reported in different villages in Lwemiyaga S/C, with more carcass buyers coming in from neighbouring subcounties such as Kyera and Ntuusi. This likely led to the death of another meat dealer in Lwensankala Parish on December 1, 2024, and a subsequent increase in human cases in Lwemiyaga and other subcounties in the first week of December 2024. Public health measures were initiated by Dec 17, 2024 (Figure 4).
Laboratory findings
Of the 15 human samples submitted, results were received for nine, all of which were positive for Bacillus anthracis by PCR. By the time of the report, the response team had not received results for the six human samples, two animal samples, and one environmental sample. These were classified as suspect based on clinical presentation and epidemiological information.
Environmental assessment findings
Introduction of new livestock
Cattle had been dying suddenly on farm X in Kakindo Parish, Engari Subcounty in Kazo District since August 2024. This is the subcounty from which farm A in Sembabule District received new livestock (Figure 5, Arrow 1).
By the time of our investigation, farm X in Kazo District had cumulatively lost 25 cattle.
We interviewed the Veterinary Officer and Veterinary Surveillance Focal Person (VSFP) for Kazo District. He was quoted thus:
“Cattle have been dying in Engari Subcounty for some time, and right now we are in outbreak mode in Kazo District.”
Farm biosecurity practices: grazing in farms where butchering had occurred
There was livestock mixing during grazing with livestock grazing on the farms where butchering of dead cows had taken place. It was after cattle deaths in farm A that cattle deaths were reported in farm B in the same village Makukulu, in Lwensakala parish,
and later in farm C in Nyendo village in a Lwemibu (Fig 5, Arrow 2), a neighboring parish. Both these parishes are in Lwemiyaga Subcounty. Cattle deaths were later reported in Kyera Subcounty (Fig. 5, Arrow 3).
Hypothesis generation findings
Livestock owners
The owner of Farm A had received cattle from his in-laws in Kazo District in October 2024. These were gifts which had been promised to him at a traditional marriage ceremony for his daughter, which he hosted earlier in the year (May 2024). He reported that these 2 cows didn’t look as healthy as the ones on his farm. He hoped they would eventually gain weight, so he immediately mixed them with the rest of his herd. Unfortunately, one of the cows died shortly after arrival.
Farmers had been losing cattle and selling the carcasses to meat dealers. Farmers viewed the sale of the carcass of their dead livestock as a means to salvage some farm running costs. One of the farmers had this to say: “Cattle always die, and it’s a regular practice to sell the carcass so that we don’t make a total loss. The little money from selling the carcass can help to ease some farm expenses, such as buying drugs.
Animal Health Worker
Cattle deaths were first reported in Makukulu village at farm A, which had received two cows from Engari subcounty in Kazo District. There were no proper herd health/biosecurity measures as the new cows were immediately mixed with the existing herd. With communal grazing, more cattle started dying in Nyendo – the neighboring village, and later in Kyera, the neighboring subcounty (Figure 5).
“Not all livestock deaths are reported to me. Sometimes I find out later during my farm visits about cattle that died weeks ago. The owners are usually concerned that I will condemn the meat, so most of the carcasses are sold to meat dealers” (Animal Health Worker)
Illegal meat network
From our interview with a local meat dealer, we learned that the district has a well-established network of illegal meat traders. He revealed that trading in meat from dead livestock is a lucrative business because many livestock owners frequently lose cattle and goats. According to him, the venture requires minimal capital investment, as dealers often purchase carcasses on credit and make payments only after selling the meat. He further explained that the ready market encourages farmers to contact these dealers promptly whenever their animals die. The only essential requirement to operate in this business, he noted, is having the means to transport the carcass or meat from the farm where the animal has died, and in his words:
“The business is lucrative and easy to operate as long as farmers have your phone number” (Meat Dealer 1).
We further probed Dealer 1 to understand whether he was aware that his business was illegal and posed a threat to public health. He acknowledged this, saying, “I know this is illegal business, but we have never had this disease; everything has been okay. Most of the meat we sell is okay. The people have no problem with it. Only that I cannot give it to my family”
Another Meat Dealer, who specializes in selling deboned meat, described his trade as more profitable than selling meat locally within Sembabule to roadside roasters or restaurant operators. He explained that his clients come from as far as Kampala City and pay in cash upon delivery. “I have clients who come for large orders, sometimes buying an entire carcass. All I need to do is debone the meat for them. I can buy a carcass at Shs 150,000/=* and sell it immediately to buyers from Kampala at Shs 800,000/=*. This is better than selling to meat roasters here” (Dealer 2).
(*1.0 USD = 3,650 Shs)
Community perceptions
We found that there was a low-risk perception concerning the consumption of meat from dead cattle, as it was the first time they had ever seen anyone with such severe lesions. The disease was associated with witchcraft, and they didn’t believe in getting medical care. We captured some perceptions in the words of one of the case-patients, who had been using herbs to treat the disease.
“I know this sickness, and these herbs will help me. If you get and go for injections, you can die” (Case-patient)
Case-patients’ perspective
Fifteen of the nineteen (79%) case-patients interviewed had eaten meat from animals that had died suddenly, 11/19(58%) had participated in the butchering of animals that had died suddenly, and 8/19 (42%) had carried meat after the butchering of cattle that had died suddenly (Table 1).
Analytical epidemiology
Hypothesis generation
Based on descriptive epidemiology, environmental assessment, and hypothesis-generating interviews, we hypothesized that the outbreak could have started with the introduction of infected cattle and propagated through cattle deaths during grazing. We further hypothesized that the spread of the disease among humans was likely associated with participation in butchering , carrying or consuming meat from these cows that had died suddenly. We designed a case-control study with a 1:4 case-to-control ratio. A total of 198 residents (cases and controls) of Sembabule District were included. Baseline demographic and contextual characteristics of the cases and controls are shown in Table 2.
Case-Control study findings
We found that individuals who consumed meat from animals that died suddenly had higher odds of getting anthrax (aOR=10.3, 95%CI:3.5-27.7) compared to those who did not. Additionally, those who participated in handling and butchering of animals that had died suddenly also had higher odds of getting cutaneous anthrax compared to those who did not (aOR=18.8, 95%CI:3.3-103.7). Our adjusted odds ratios had wide confidence intervals, indicating imprecision in effect-size estimates (Table 3).
Discussion
This investigation confirmed an anthrax outbreak in Sembabule District (October–December 2024) affecting humans and livestock, manifesting as cutaneous, gastrointestinal, and mixed forms. Infection was associated with butchering, handling, and consuming meat from animals that died suddenly, disproportionately affecting adult males. Delayed detection and care-seeking, influenced by community perceptions, coupled with illegal meat trade networks amplifying dissemination, highlighted critical gaps in surveillance, farm biosecurity, and risk communication.
Strong associations were observed between anthrax infection, primarily cutaneous, and butchering, handling, and consumption of meat from animals that died suddenly, consistent with established transmission drivers [4, 5]. Given the inherent overlap among these concurrent behavioural exposures, collinearity was a concern, and results were interpreted by the direction of association rather than by aOR magnitude. These findings emphasize the necessity for enhanced community education on safe carcass disposal and rigorous enforcement of public health and veterinary regulations concerning the trade and consumption of meat from deceased animals. Sustained risk communication and legal enforcement are crucial to mitigate recurrent human-animal anthrax transmission
Adult males were disproportionately affected, consistent with their occupational and culturally assigned roles in livestock handling and meat processing [6] This demographic pattern aligns with previous studies on anthrax risk determinants[7, 8], and necessitates targeted prevention strategies for high-risk occupational groups involved in slaughter, butchery, and meat trade.
Consumption of meat from animals that died suddenly strongly correlated with gastrointestinal anthrax risk. Unlike some prior research suggesting that cooking methods achieving higher temperatures reduced infection risk [9, 10], our analysis found no difference in anthrax risk based on the method of meat preparation, likely due to insufficient cooking that negated any potential protective effect. These findings suggests that proper preparation alone is inadequate for preventing anthrax infection during outbreaks. Consumption of such meat must be strictly discouraged. Sustainable control, especially routine livestock vaccination, is crucial for reducing exposure from contaminated meat[11].
This first reported anthrax outbreak in Sembabule District, surrounded by historically affected districts [4, 7, 10, 12, 13], and epidemiologically linked to infected livestock imported from Kazo District. The index case involved butchering and consuming meat from animals originating from this area, where a farm had lost over 25 cattle [12, 14]. This highlights inter-district anthrax transmission, consistent with regional livestock movement patterns [15]. The emergence in a previously unaffected district underscores concerns regarding unregulated livestock movement and geographic expansion. Stricter cross-district controls and joint surveillance are recommended to enhance zoonotic disease prevention.
Environmental assessment indicated poor farm biosecurity, including lack of quarantine and vaccination for imported livestock from an endemic area, as a key factor in initial outbreak spread across farms and sub-counties. This aligns with previous studies showing unvaccinated herds contribute to anthrax transmission [11, 16, 17]. Given unregulated livestock movement across porous administrative borders and recurrent outbreaks in new districts, long-term anthrax control requires sustainable, proactive solutions beyond reactive measures. Policy options for anthrax vaccination in this setting were previously described [18]. We recommend strengthening veterinary extension services, farmer biosecurity training, and expanding nationwide access to affordable anthrax vaccines
Illegal meat dealer networks amplified the outbreak by distributing meat from deceased animals, reaching urban centers like Kampala, but primarily impacting Sembabule communities where human cases clustered. This pattern mirrors observations in other anthrax outbreaks where informal trade facilitated transmission[7, 14, 17, 3]. Driven by socio-economic factors and weak regulatory enforcement, these networks undermine veterinary oversight and public trust, sustaining zoonotic transmission [19]. Strengthening enforcement of relevant Animal Diseases and Public Health Acts is crucial to disrupt this structural amplification of outbreaks.
The delayed outbreak detection (nearly two months after the initial livestock deaths) enabled extensive carcass distribution and consumption, facilitating the spread of infection. Community misconceptions, including attributing illness to witchcraft and distrust of formal healthcare, led to delayed care-seeking and reporting, a pattern seen in prior outbreaks [20]. Even Village Health Teams (VHTs), critical for community-level health reporting, initially lacked awareness of anthrax. Such delays escalate community exposure and complicate containment. Strengthening risk communication and VHT capacity through a One Health approach and Integrated Disease Surveillance and Response (IDSR) is essential for timely reporting of human and animal health events, thereby enhancing early response [21].
Study limitations
Limitations include potential underestimation of the magnitude of human cases due to low health worker suspicion during this district’s first reported outbreak. Recall bias in exposure histories, particularly for gastrointestinal anthrax, is possible given the investigation’s delayed start. GI cases relied on clinical records and self-reported exposure without microbiological confirmation, impacting classification precision. Unreported animal deaths to veterinary authorities, instead diverted to illegal meat dealers, suggest a potential underestimation of overall outbreak magnitude. We did not receive laboratory results for all samples collected, which limited the number of laboratory-confirmed cases and may have resulted in an underestimation of the magnitude of this outbreak. Finally, the closely related exposure variables (e.g., butchery, handling, consumption) – which are inherently overlapping and likely to occur in the same individual raise concerns of collinearity in this case-control analysis. Since formal statistical testing fir collinearity was not done, the inflated adjusted odds ratios and wide confidence intervals for these variables, necessitate cautious interpretation focused on association direction rather than magnitude of the estimates. Future studies could prioritize laboratory confirmation across all clinical forms where feasible.
Conclusion
The Sembabule District anthrax outbreak stemmed from infected cattle introduction, amplified by unsafe carcass handling and distribution of meat from animals that died suddenly. The outbreak, predominantly affecting adult males with cutaneous and gastrointestinal forms, was strongly linked to butchering, handling, and consuming infected meat. Poor farm biosecurity, illegal meat trade, delayed healthcare-seeking due to community misconceptions, and late detection collectively contributed to sustained transmission and widespread disease. These findings underscore anthrax as a persistent zoonotic threat in Uganda, perpetuated by regulatory gaps in livestock movement, inadequate biosecurity, and limited community awareness. Proactive, One Health-grounded control measures are crucial, including strengthened community-based surveillance for human and animal health, targeted risk communication, rigorous enforcement of meat safety and livestock movement laws, and expanded anthrax livestock vaccination. Prioritizing these interventions is vital to mitigating recurrent outbreaks and preventing geographic spread. Public health actions included reinforced risk communication and local authority operations to address illegal meat trade and promote safe practices.
What is already known about the topic
- Anthrax is a priority zoonotic disease in Uganda, with recurrent outbreaks predominantly in the cattle corridor but also in districts outside it, linked to handling and consumption of meat from dead animals.
- Butchering, handling, and consuming meat from dead animals are established risk factors, disproportionately affecting adult males in livestock-keeping communities.
- Despite the availability of anthrax vaccines, Uganda lacks a national anthrax control program, leaving communities vulnerable to repeated outbreaks and associated losses and disruptions.
What this study adds
- This is the first documented anthrax outbreak in Sembabule District in Uganda, demonstrating geographic expansion through weakly-regulated inter-district livestock movement.
- A well-established illegal meat dealer network extending to urban centersamplified transmission and complicated containment.
- Community misconceptions, including attribution of illness to witchcraft, delayed care-seeking and reporting, underscore critical gaps in risk communication and community-level surveillance.
Funding
This study was supported by the President’s Emergency Plan for AIDS Relief (PEPFAR) through the United States Centers for Disease Control and Prevention Cooperative Agreement number GH001353-01 through Makerere University School of Public Health to the Uganda Public Health Fellowship Program, Ministry of Health. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of the US Centers for Disease Control and Prevention and the Department of Health and Human Services, Makerere University School of Public Health, or the Uganda Ministry of Health.
Acknowledgements
We acknowledge and appreciate the Ministry of Health for granting authorization to conduct this investigation in Sembabule District. Additionally, we appreciate the Sembabule District Task Force (DTF) for the support in providing information and guiding the investigation team in the affected communities. Final appreciation goes to the US-CDC for the continuous support rendered to the Uganda Public Health Fellowship Program (UPHFP).
Authors´ contributions
HK led the investigation and drafted the manuscript. HK, EM, GA, TN and JFZ investigated the outbreak and participated in the design of data collection tools, data collection, entry and cleaning, analysis, and review of the manuscript draft. JO, RM, and BK supervised the outbreak investigation and reviewed the manuscript for intellectual content. SAA and LA supported sample collection and laboratory analysis. RM, BK, LB, JO and ARA reviewed manuscript draft for intellectual content and scientific integrity. All authors reviewed and approved the final draft of the manuscript.
| Exposure (n=19) | Frequency | % |
|---|---|---|
| Ate meat from animals that died suddenly | 15 | 79 |
| Participated in butchering dead animals | 11 | 58 |
| Carried meat home after butchering dead animals | 8 | 42 |
| Owned cattle | 5 | 26 |
| Moved out of Sembabule District since start of the outbreak | 3 | 16 |
| Cases, n=45 | Controls, n=153 | ||
|---|---|---|---|
| Characteristic (n=198) | n(%) | n(%) | p-value |
| Age group (years) | |||
| <18 | 5 (11) | 18 (12) | 1 |
| ≥18 | 40 (89) | 135 (88) | |
| Sex | |||
| Female | 6 (13) | 26 (17) | |
| Male | 39 (87) | 127 (83) | 0.004 |
| Animal ownership | |||
| Yes | 17 (38) | 80 (52) | 0.002 |
| No | 28 (62) | 73 (48) | |
| Sub-county of residence | |||
| Kyera | 9 (20) | 39 (25) | |
| Lwemiyaga | 34 (76) | 105 (69) | 0.036 |
| Ntusi Town Council | 2 (4) | 9 (6) | |
| Livelihood | |||
| Livestock/meat-related occupation* | 32 (71) | 54 (35) | <0.001 |
| Other occupations | 13 (29) | 99 (65) | |
| *butchers, herdsmen, meat traders, meat transporters, slaughterers, meat roasters, and skinners | |||
| p-value calculated using χ² test | |||
| Exposure | Number of participants n (%) | cOR | aOR | p-Value | |
|---|---|---|---|---|---|
| Case (%) | Controls (%) | ||||
| Consumed meat from suddenly dead animal | |||||
| No | 11 (24) | 117 (74) | Ref | Ref | <0.001 |
| Yes | 34 (76) | 42 (26) | 8.6 (4.0-18.5) | 10.3 (3.5-29.7) | |
| Butchered/Handled a dead cow | |||||
| No | 12 (27) | 133 (84) | Ref | Ref | <0.001 |
| Yes | 33 (73) | 26 (16) | 14.1 (6.4-31.0) | 18.8 (3.3-107.3) | |
| Flaying/Skinning | |||||
| No | 30 (67) | 153 (96) | Ref | Ref | 0.05 |
| Yes | 15 (33) | 6 (4) | 12.8 (4.6-35.5) | 6.7 (1.0-45.2) | |
| Carried meat during butchery | |||||
| No | 23 (51) | 139 (87) | Ref | Ref | 0.00875 |
| Yes | 22 (49) | 20 (13) | 6.5 (3.2-14.1) | 0.25 (0.1-6.1.2) | |
| Carcass dressing | |||||
| No | 26 (58) | 153 (96) | Ref | Ref | 0.03 |
| Yes | 19 (42) | 6 (4) | 18.6 (6.8-51.1) | 5.9 (1.2-28.6) | |
| Cutting | |||||
| No | 26 (58) | 146 (92) | Ref | Ref | 0.583 |
| Yes | 19 (42) | 13 (9) | 8.2 (3.6-18.6) | 0.6 (0.1-3.1) | |





References
- Sushma B, Shedole S, Suresh KP, Leena G, Patil SS, Srikantha G. An Estimate of Global Anthrax Prevalence in Livestock: A Meta-analysis. Vet World [Internet]. 2021 May 22 [cited 2026 Mar 06];1263–71. doi:10.14202/vetworld.2021.1263-1271 Available from: https://www.veterinaryworld.org/Vol.14/May-2021/26.html
- Madsen JM. Biological agents as weapons [Internet]. Kenilworth (NJ): Merck Sharp & Dohme LLC; 2024 Oct [cited 2026 Mar 06]. Available from: https://www.msdmanuals.com/professional/injuries-poisoning/mass-casualty-weapons/biological-agents-as-weapons
- Tumusiime L, Kizza D, Kiyimba A, Nabatta E, Waako S, Byaruhanga A, Kwesiga B, Migisha R, Bulage L, Ario AR. Anthrax outbreak linked to consumption and handling of meat from unexpectedly deceased cattle, Kyotera district, Uganda, June–December 2023. One Health Outlook [Internet]. 2025 May 7 [cited 2026 Mar 06];7(1):29. doi:10.1186/s42522-025-00151-x Available from: https://link.springer.com/article/10.1186/s42522-025-00151-x
- Ntono V, Eurien D, Bulage L, Kadobera D, Harris J, Ario AR. Cutaneous anthrax outbreak associated with handling dead animals, Rhino Camp sub-county: Arua District, Uganda, January–May 2018. One Health Outlook [Internet]. 2021 [cited 2026 Mar 06];3(1):8. doi:10.21203/rs.3.rs-38244/v2 Available from: https://www.researchsquare.com/article/rs-38244/v2
- Musewa A, Mirembe BB, Monje F, Birungi D, Nanziri C, Aceng FL, Kabwama SN, Kwesiga B, Ndumu DB, Nyakarahuka L, Buule J, Cossaboom CM, Lowe D, Kolton CB, Martson CK, Stoddard RA, Hoffmaster AR, Ario AR, Zhu BP. Outbreak of cutaneous anthrax associated with handling meat of dead cows in southwestern Uganda, May 2018. Trop Med Health [Internet]. 2022 Aug 6 [cited 2026 Mar 06];50(1):52. doi:10.1186/s41182-022-00448-y Available from: https://link.springer.com/article/10.1186/s41182-022-00445-0
- Oketch DCO, Njoroge R, Ngage TO, Omar AA, Magarre A, Pasha R, Gachohi J, Muriuki SW, Khamadi SA, Boru AD, Bodha B, Kilowua L, Nyaga NW, Njaanake HK, Kamaara E, Jaoko W, Njenga MK, Osoro E, Omia D. Cultural and behavioral drivers of zoonotic disease transmission and persistence among diverse pastoralist communities in East Africa. One Health Outlook [Internet]. 2025 Jul 14 [cited 2026 Mar 06];7(1):36. doi:10.1186/s42522-025-00153-9 Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12257696/
- Migisha R, Mbatidde I, Agaba DC, Turyakira E, Tumwine G, Byaruhanga A, Siya A, Ruzaaza GN, Kirunda H. Risk factors for human anthrax outbreak in Kiruhura District, Southwestern Uganda: a population-based case control study. PAMJ-OH [Internet]. 2021 Jun 30 [cited 2026 Mar 06];5:13. doi:10.11604/pamj-oh.2021.5.13.29385 Available from: https://www.one-health.panafrican-med-journal.com/content/article/5/13/full/
- Chowdhury S, Islam MdS, Haider N, Hossain MB, Alam MdA, Sharif MdAR, Uzzaman MS, Rahman M, Rahman M, Haque F. Risk factors associated with cutaneous anthrax outbreaks in humans in Bangladesh. Front Public Health [Internet]. 2024 Oct 15 [cited 2026 Mar 06];12:1442937. doi:10.3389/fpubh.2024.1442937 Available from: https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2024.1442937/full
- Nakanwagi M, Ario AR, Kwagonza L, Aceng FL, Mwesigye J, Bulage L, Buule J, Sendagala JN, Downing R, Zhu BP. Outbreak of gastrointestinal anthrax following eating beef of suspicious origin: Isingiro District, Uganda, 2017. PLoS Negl Trop Dis [Internet]. 2020 Feb 27 [cited 2026 Mar 06];14(2):e0008026. doi:10.1371/journal.pntd.0008026 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0008026
- Kibwika B, Namulondo E, Aanyu D, Nuwamanya Y, Rek J, Kiza D, Kamukama A, Ssemanda I, Waako S, Komugisha M, Namara B, Orit D, Tumusiime L, Mwebaza S, Baliruno LN, Atim P, Kiyimba A, Nankya MA, Arinaitwe E, Bahizi G, Gidudu S, Migisha R, Gonahasa DN, Kwesiga B, Kadobera D, Ario RA. Anthrax outbreak associated with handling and consuming meat from animals that died suddenly—Ibanda District, Uganda, May 2023. UNIPH [Internet]. 2023 Oct 13 [cited 2026 Mar 06];8(3):3. Available from: https://uniph.go.ug/wp-content/uploads/2023/09/Anthrax-outbreak-associated-with-handling-and-consuming-meat-from-animals-that-died-suddenly-Ibanda-District-Uganda-May-2023.pdf
- Shadomy S, ElIdrissi A, Raizman E, Pittiglio C, Bruni M, Lubroth J. [Anthrax report or factsheet] [Internet]. Rome (Italy): FAO; 2016 [cited 2026 Mar 06]. 8 p. Available from: https://openknowledge.fao.org/server/api/core/bitstreams/59269d58-654e-4790-ac60-a9ea7edd3a99/content
- Senyange S, Nsubuga EJ, Kwesiga B, Bulage L, Ario AR. Knowledge, attitudes, and practices regarding anthrax among affected communities, Kazo district, South-Western Uganda, May 2022. BMC Public Health [Internet]. 2025 Jul 2 [cited 2026 Mar 06];25(1):2249. doi:10.1186/s12889-025-23436-2 Available from: https://link.springer.com/article/10.1186/s12889-025-23436-2
- Monje F, Kisakye E, Eurien D, Kwesiga B, Kadobera D, Ario AR. Animal Anthrax outbreak caused by slaughtering infected carcasses on and or near the pastureland, Kiruhura District, Uganda, May-October 2018. UNIPH [Internet]. 2021 Oct 2 [cited 2026 Mar 06];3(4). Available from: https://uniph.go.ug/animal-anthrax-outbreak-caused-by-slaughtering-infected-carcasses-on-and-or-near-the-pastureland-kiruhura-district-uganda-may-october-2018/
- Nuwamanya Y, Kizza D, Kiyimba A, Ssemanda I, Waako S, Aanyu D, Monje F, Kwesiga B, Migisha R, Ario AR. Anthrax outbreaks in western Uganda: The role of illegal meat dealers in spreading the infection, August 2022–April 2023. UNIPH [Internet]. 2024 Jan-Mar [cited 2026 Mar 06];9(1):1. Available from: https://uniph.go.ug/wp-content/uploads/2024/04/Anthrax-outbreaks-in-western-Uganda-The-role-of-illegal-meat-dealers-in-spreading-the-infection-August-2022%E2%80%93April-2023.pdf
- Hasahya E, Thakur K, Dione MM, Kerfua SD, Mugezi I, Lee HS. Analysis of patterns of livestock movements in the Cattle Corridor of Uganda for risk-based surveillance of infectious diseases. Front Vet Sci [Internet]. 2023 Jan 23 [cited 2026 Mar 06];10:1095293. doi:10.3389/fvets.2023.1095293 Available from: https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2023.1095293/full
- Youssef DM, Wieland B, Knight GM, Lines J, Naylor NR. The effectiveness of biosecurity interventions in reducing the transmission of bacteria from livestock to humans at the farm level: A systematic literature review. Zoonoses Public Health [Internet]. 2021 Feb 4 [cited 2026 Mar 06];68(6):549–62. doi:10.1111/zph.12807 Available from: https://onlinelibrary.wiley.com/doi/10.1111/zph.12807
- Vieira AR, Salzer JS, Traxler RM, Hendricks KA, Kadzik ME, Marston CK, Kolton CB, Stoddard RA, Hoffmaster AR, Bower WA, Walke HT. Enhancing Surveillance and Diagnostics in Anthrax-Endemic Countries. Emerg Infect Dis [Internet]. 2017 Dec [cited 2026 Mar 06];23(Suppl 1):S147–S153. doi:10.3201/eid2313.170431 Available from: https://wwwnc.cdc.gov/eid/article/23/13/17-0431_article
- Amanya G, Migisha R, Kisakye E. Re-classifying anthrax as a public good disease: policy brief. UNIPH Quarterly Epidemiological Bulletin [Internet]. 2021 Oct-Dec [cited 2026 Mar 06];6(40):2. Available from: https://uniph.go.ug/wp-content/uploads/2021/12/Re-classifying-Anthrax-as-a-Public-Good-Disease-Policy-Brief.pdf
- Masudi SP, Hassell J, Cook EA, Van Hooft P, Van Langevelde F, Buij R, Otiende MY, Ochieng JW, Santangeli A, Happi A, Akpan SN, Thomas LF. Limited knowledge of health risks along the illegal wild meat value chain in the Nairobi Metropolitan Area (NMA). PLoS ONE [Internet]. 2025 Mar 26 [cited 2026 Mar 06];20(3):e0316596. doi:10.1371/journal.pone.0316596 Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0316596
- Martínez-Campreciós J, Moreno M, Salvador F, Barrio-Tofiño ED, Nindia A, Aznar ML, Molina I. Impact of traditional cutaneous scarification on anthrax lesions: A series of cases from Cubal, Angola. Int J Infect Dis [Internet]. 2024 Mar [cited 2026 Mar 06];140:104–9. doi:10.1016/j.ijid.2024.01.004 Available from: https://www.sciencedirect.com/science/article/pii/S1201971224000043
- Stärk KDC, Arroyo Kuribreña M, Dauphin G, Vokaty S, Ward MP, Wieland B, Lindberg A. One Health surveillance – More than a buzzword? Prev Vet Med [Internet]. 2015 Jun [cited 2026 Mar 06];120(1):124–30. doi:10.1016/j.prevetmed.2015.01.019 Available from: https://www.sciencedirect.com/science/article/abs/pii/S0167587715000355
