Research | Open Access | Volume 9 (1): Article 39 | Published: 06 Mar 2026
Typhoid fever in the West Gonja Municipality, Ghana: Analysis of 2022 laboratory surveillance data
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Table 1: Demographic characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022 | |||
|---|---|---|---|
| Variable | Suspected Cases N=2998 N (%) | Tested N=377 N (%) | Positivity % [95% CI] |
| Age (years), Mean (SD) | 30.8 (±18.2) | ||
| Age group | |||
| <11 | 546 (18.2) | 54 (14.3) | 55.6 [41.4-69.1] |
| 11-20 | 258 (8.6) | 65 (17.2) | 52.3 [39.5-64.9] |
| 21-30 | 415 (13.8) | 85 (22.6) | 44.7 [33.9-55.9] |
| 31-40 | 478 (15.9) | 80 (21.2) | 55.0 [43.5-66.2] |
| 41-50 | 566 (18.9) | 32 (8.5) | 40.6 [23.7-59.4] |
| 51-60 | 317 (10.6) | 31 (8.2) | 61.3 [42.2-78.2] |
| 61+ | 418 (13.9) | 30 (8.0) | 63.3 [43.9-80.1] |
| Sex | |||
| Female | 1,953 (65.1) | 206 (54.6) | 49.0 [42.0-56.1] |
| Male | 1,045 (34.9) | 171 (45.4) | 56.1 [48.4-63.7] |
| Sub-District | |||
| Busunu | 25 (0.8) | 5 (1.3) | 20.0 [0.5-71.6] |
| Canteen | 2085 (69.5) | 293 (77.7) | 55.3 [49.4-61.1] |
| DASS | 717 (23.9) | 6 (1.6) | 83.3 [35.9-99.6] |
| Damongo | 7 (0.2) | 71 (18.8) | 40.8 [29.3-53.2] |
| Larabanga | 164 (5.5) | 2 (0.5) | 0.0 [0.0-1.8] |
Table 1: Demographic characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022
| Table 2: Seroprevalence and antibody characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022, N=377 | |||
|---|---|---|---|
| Variable | Frequency | Percentage | 95% CI |
| Overall seroprevalence | 197 | 52.3 | 47.1-57.4 |
| Serotype | |||
| Both IgM & IgG Negative | 180 | 47.7 | 42.6-52.9 |
| Both IgM & IgG Positive | 62 | 16.5 | 12.8-20.6 |
| Only IgM Positive | 111 | 29.4 | 24.9-34.3 |
| Only IgG Positive | 24 | 6.4 | 4.1-9.3 |
Table 2: Seroprevalence and antibody characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022, N=377
| Table 3: Age, sex and Sub-municipal association with typhoid fever antibody status in the West Gonja Municipality, Ghana, 2022 | ||||
|---|---|---|---|---|
| Variable | Typhoid Positive N=197 n (%) | Typhoid Negative N=180 n (%) | Crude OR (95% CI) | p-value |
| Age (years) | ||||
| <11 | 30 (15.2) | 24 (13.3) | 1 | |
| 11-20 | 34 (17.3) | 31 (17.2) | 1.14 (0.55-2.35) | 0.724 |
| 21-30 | 38 (19.3) | 47 (26.1) | 1.54 (0.78-3.07) | 0.213 |
| 31-40 | 44 (22.3) | 36 (20.0) | 1.02 (0.51-2.05) | 0.949 |
| 41-50 | 13 (6.6) | 19 (10.6) | 1.83 (0.75-4.43) | 0.183 |
| 51-60 | 19 (9.6) | 12 (6.7) | 0.79 (0.32-1.94) | 0.607 |
| 61+ | 19 (9.6) | 11 (6.1) | 0.72 (0.29-1.81) | 0.489 |
| Sex | ||||
| Female | 101 (51.3) | 105 (58.3) | 1 | |
| Male | 96 (48.7) | 75 (41.7) | 0.75 (0.50-1.13) | 0.169 |
| Sub-District | ||||
| Busunu | 1 (0.5) | 4 (2.2) | 1 | |
| Canteen | 162 (82.2) | 131 (72.8) | 0.20 (0.02-1.83) | 0.155 |
| DASS | 5 (2.5) | 1 (0.6) | 0.05 (0.00-1.07) | 0.056 |
| Damongo | 29 (14.7) | 42 (23.3) | 0.36 (0.04-3.47) | 0.374 |
| Larabanga* | 0 (0.0) | 2 (1.1) | – | |
| * Larabanga was omitted from the analysis due to no data for typhoid positive cases, hence its COR could not be estimated by the software. | ||||
Table 3: Age, sex and Sub-municipal association with typhoid fever antibody status in the West Gonja Municipality, Ghana, 2022
Figure
Keywords
- Typhoid fever
- Seroprevalence
- Laboratory surveillance data
- West Gonja Municipality
- Ghana
Farouk Umar Saeed1,&, Samuel Jerry Apetorgbor2, Gyesi Razak Issahaku3, George Akowuah3, Magdalene Odikro3, Ernest Kenu3, Joseph Alphonsus Chantiwuni Nindow4, Chrysantus Kubio1
1Savannah Regional Health Directorate, Ghana Health Service, Damongo, Ghana, 2Laboratory Department, St. Anne’s Hospital, Damongo, Ghana, 3Ghana Field Epidemiology and Laboratory Training Program, School of Public Health, University of Ghana, Accra, Ghana, 4Ghana Health Service, Bole District Hospital, Bole, Ghana
&Corresponding author: Farouk Umar Saeed, Savannah Regional Health Directorate, Ghana Health Service, Damongo, Ghana, Email: fkumar470@gmail.com, ORCID: https://orcid.org/0009-0003-5768-3017
Received: 16 Dec 2024, Accepted: 27 Feb 2026, Published: 06 Mar 2026
Domain: Infectious Disease Epidemiology
Keywords: Typhoid fever, seroprevalence, laboratory surveillance data, West Gonja Municipality, Ghana
©Farouk Umar Saeed et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Farouk Umar Saeed et al., Typhoid fever in the West Gonja Municipality, Ghana: Analysis of 2022 laboratory surveillance data. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):39. https://doi.org/10.37432/jieph-d-24-02048
Abstract
Introduction: Typhoid fever (TF) is a major public health problem in developing countries. Surveillance data from the West Gonja Municipality (WGM) of Ghana have not been analysed to provide an understanding of the disease burden. This study aimed to determine the seroprevalence, trend and distribution of TF in the WGM of Ghana.
Method: We conducted a secondary data analysis by collecting data on laboratory-tested cases of suspected TF between January and December 2022. We determined the seroprevalence based on either IgM or IgG or both. We tested associations between demographics and TF antibody status using logistic regression. A p-value less than 0.05 was considered statistically significant at 95% confidence interval (CI).
Results: A total of 2,998 suspected and 377 (12.6%) tested TF records were analysed. The mean age of the tested cases was 30.8 years (SD ± 18.2). The overall seroprevalence of TF was 52.3% (197/377), [95% CI (47.1-57.4)]. There was no significant association between typhoid serum antibody status with age groups, sex and sub-municipality. Seroprevalence was highest among individuals aged 61 years and above (63.3%), 51-60 years (61.3%), and children <11 years (55.6%). Males had a higher rate, 56.1% (96/171). The Dass and Canteen Sub-municipalities had the highest positivity rates, 83.3% (5/6) and 55.3% (162/293) respectively. Over the period, cases fluctuated with peaks in March and April.
Conclusion: The seroprevalence of TF was high in the municipality with males, the oldest aged groups and children having the highest burden of infection. Although testing was low, there was a high positivity in March and April, with cases positively skewed towards Dass and Canteen areas. The health authorities should improve testing and institute laboratory-based culture testing and surveillance.
Introduction
The bacteria Salmonella enterica subspecies enterica serovars typhi is the cause of the acute, potentially fatal febrile disease known as typhoid fever. In Africa, South-East Asia, the Americas, and the Eastern Mediterranean, it is a serious public health issue. People who live in places without access to safe drinking water and sanitation facilities are the most at risk of typhoid fever [1]. Estimates from the global burden of diseases indicate that there are about 11 million cases of typhoid fever around the world, resulting in 117,000 deaths [2]. The highest estimated incidences occurred in the WHO South-East Asian (306 cases per 100,000 persons), Eastern Mediterranean (187/100,000 persons) and African regions (111 cases per 100,000 persons) [3].
In low- and middle-income countries like Ghana, the disease burden is substantial. The latest report by the Ghana Health Service ranked typhoid fever amongst the leading causes of outpatient diseases [4]. A total of 365,148 cases were recorded in 2017 and ranked among the top twenty causes of morbidity in Ghana [5]. Incidence rates obtained in a study among children under 15 years of age were over 100 cases per 100,000 persons and two to almost three times higher in rural areas of Ghana [6]. Ghana was enrolled on the programmes for surveillance of enteric fever in Africa in a bid to provide data for typhoid fever with emphasis on children. The data generated from other studies in Asia led to the development of the Typhoid Conjugate Vaccine (TCV) to prevent the disease [7]. The WHO suggested TCV as a standard immunisation for infants and children older than six months, with the possibility for catch-up campaigns for children up to 15 years old in high-endemic areas [8]. However, Ghana is yet to introduce the vaccine, partly due to insufficient data on the disease burden [3].
Although previous studies have been conducted in other parts of Ghana, there is a paucity of data on disease burden. This is partly due to limited local laboratory capacity to conduct culture tests. This notwithstanding, a study by Labi et al. reported a 6.5% prevalence of Salmonella bacteraemia in the Korle-Bu Teaching Hospital [9]. In the Asante Akyim North Municipality, Cruz Espinoza et al. reported incidence >100/100,000 persons among children below 15years old [6]. Another study by Fusheini et al. in the Hohoe municipality described the situation as a public health challenge and reported an incidence as high as 943/100,000 persons in the year 2015 [10]. Despite this available data, there is a need for context-specific understanding of the disease and hence the need for the current study.
To the best of our knowledge, there is no study on typhoid fever in the West Gonja Municipality. Surveillance for typhoid fever in the West Gonja Municipality of the Savannah Region is limited to the reporting of health facility-based data on clinically suspected cases and serologic testing either by the Widal reaction or Typhoid IgM and IgG (Typhidot). The Widal test performs poorly compared to the gold standard test (culture) [11]. The typhidot (anti-Salmonella antibody test) has proven reliability compared to the Widal test, which has lower sensitivity and specificity [12]–[15]. An analysis of the surveillance data has not been done to provide an understanding of the disease burden in the West Gonja Municipality. This analysis will provide empirical baseline information for researchers. It will also provide valuable insight into the introduction of vaccines to control typhoid fever in Ghana. We, therefore, sought to analyse the laboratory surveillance data of typhoid fever in the West Gonja Municipality of Ghana to determine the seroprevalence, trend and distribution of the disease.
Methods
Study design and setting
We conducted a secondary data analysis on health facility-based laboratory records in the West Gonja Municipality using data reported between January and December 2022. The municipality shares boundaries with the Central Gonja District to the east and south, the North Gonja District to the north, and the Bole and Sawla-Tuna-Kalba Districts to the west. For efficient service delivery, the municipality is split up into five administrative sub-municipalities. The Busunu, Canteen, Damongo, DASS, and Larabanga Sub-municipals. The municipality has twenty-six (26) healthcare facilities, including one district hospital, four health centers and twenty-one Community-based Health Planning and Services (CHPS). In addition, there are two private laboratory diagnostic facilities and one private maternity home. The estimated population of the municipality was 64,781 according to projections from the 2021 population census [16]. The municipality’s population is centered in a small number of accessible areas.
Operation of the typhoid fever surveillance in the West Gonja Municipality
The surveillance for typhoid fever in the West Gonja Municipality is a passive system that involves the detection of suspected typhoid fever cases by trained clinicians in health facilities. Cases are suspected based on disease symptoms by applying a standard case definition and reporting to the subsequent tier of the health system. The standard case definition in Ghana for a suspected typhoid fever case is any individual exhibiting fever, chills, malaise, headache, sore throat, cough, and occasionally abdominal pain and constipation or diarrhea, together with a gradually increasing and then persistently high temperature.
Blood samples from suspected cases are collected for laboratory testing. However, due to a lack of local capacity to conduct culture on blood and stool, typhoid fever testing is limited to the serologic confirmation, either by the Widal reaction or the Typhidot (IgM and IgG) methods. Surveillance officers at the health facilities collect aggregated data as part of the integrated disease surveillance on suspected cases from patient registers and report the cases through an electronic platform, the District Health Information Management Systems 2 (DHIMS 2), to the municipal and regional health authority in the Savannah Region. The timeline for reporting at the health facility level is usually by the end of 5th day of the ensuing month. The data is validated by health information officers at the sub-municipal, municipal and regional levels for quality checks before the 15th of the next month. The data is accessible at the national level for national officers to provide feedback and updates to the health facilities. The purpose of surveillance is to quickly identify high-risk locations and report occasional cases and outbreaks of typhoid fever to improve disease prevention through health promotion.
Inclusion and exclusion criteria
All suspected cases of typhoid fever tested using the Typhidot IgM and IgG methods were included in the study. We excluded suspected typhoid fever cases without laboratory tests, as well as cases tested by the Widal reaction method, due to the relatively high nonspecificity associated with the test [17].
Sampling size
We utilised all the available data in the municipality’s health facilities over the study’s duration.
Data collection
A form was designed to guide the collection of data. Data were collected from laboratory registers of all the laboratories in the municipality. Patient information collected included age, sex, sub-municipality of residence, date patient samples were tested and the results of the typhoid test (IgM and IgG). The data were entered into Microsoft Excel 2019. We assigned unique identity numbers to the cases for easy tracking and analysis.
Data analysis
We cleaned the Excel data by removing duplicates and missing information. The data were de-identified and exported to the statistical software package by StataCorp (STATA version 15) for analysis. We determined positivity to typhoid bacteria based on the presence of either anti-typhoid IgM or IgG or both in blood test results. We summarized continuous variables using mean and standard deviation and categorical variables using frequency and percentages. We examined relationships between the dependent variable, typhoid fever antibody status, and the independent variables- age groups, gender and sub-municipals. A bivariate logistic regression analysis was done to determine significant independent variables that will be eligible for a multivariable logistic model to adjust for the effect of confounders. A chi-square goodness-of-fit test was conducted for each independent variable before the bivariate analysis. The odds ratios were reported for each independent variable. We considered results statistically significant if the p-value was less than 0.05 at the 95% confidence level. The results were presented using tables and a graph.
Ethical considerations
We obtained permission to conduct the study from the office of the Municipal Health Directorate through the Municipal Director of Health Service for West Gonja. Ethical clearance was not sought because the data were generated and used for routine public health service provision. The identities and contact information of the cases were kept strictly confidential and were not included in the data collection and analysis. All data were secured on a computer with a security-protected password.
Results
Demographic and clinical characteristics of tested typhoid fever cases
Between 1st January, 2022 and 31st December, 2022, a total of 2,998 suspected and 377 (12.6%) tested typhoid fever cases were reported. Out of the 377 individuals tested, 206 were females (54.6%) and 171 were males (45.4%). Overall, 197 individuals tested positive, giving a positivity rate of 52.3% [95% CI (47.1-57.4)]. The positivity rate among females was 49.0% (101/206) while the positivity rate among males was higher at 56.1% (96/171). The mean age of the tested cases was 30.8 years (SD ± 18.2). Positivity rates varied across age groups, with the highest positivity rates observed among individuals aged 61 years and above (63.3%) and 51-60 years (61.3%). Children under 11 years and adults aged 31-40 also recorded relatively high positivity rates of 55.6% and 55.0%, respectively.
Relatively lower positivity rates were observed among individuals in the categories 41-50 years (40.6%) and 21-30 years (44.7%). Out of the five sub-municipalities, majority of cases suspected and tested were reported from Canteen, 69.5% and 77.7% respectively. Positivity rates also differed across sub-municipalities. The highest positivity rate was recorded in the DASS sub-municipality (83.3%), followed by Canteen (55.3%). Damongo recorded a positivity rate of 40.8% while Busunu had a lower rate of 20%. Larabanga did not record any positive cases, giving a positivity rate of 0.0% (Table 1). Of the 197 positives recorded, 111 were positives for only IgM antibody type and 24 for only IgG antibody type. There were sixty-two (62) positives for both IgG and IgM (Table 2).
Association between demographic variables and typhoid serum antibody status
In the bivariate logistic regression model, there was no significant association between the independent variables, age groups, gender and sub-municipality with the dependent variable, typhoid antibody status (p > 0.05) (Table 3).
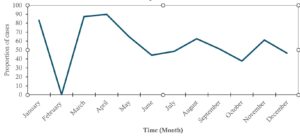
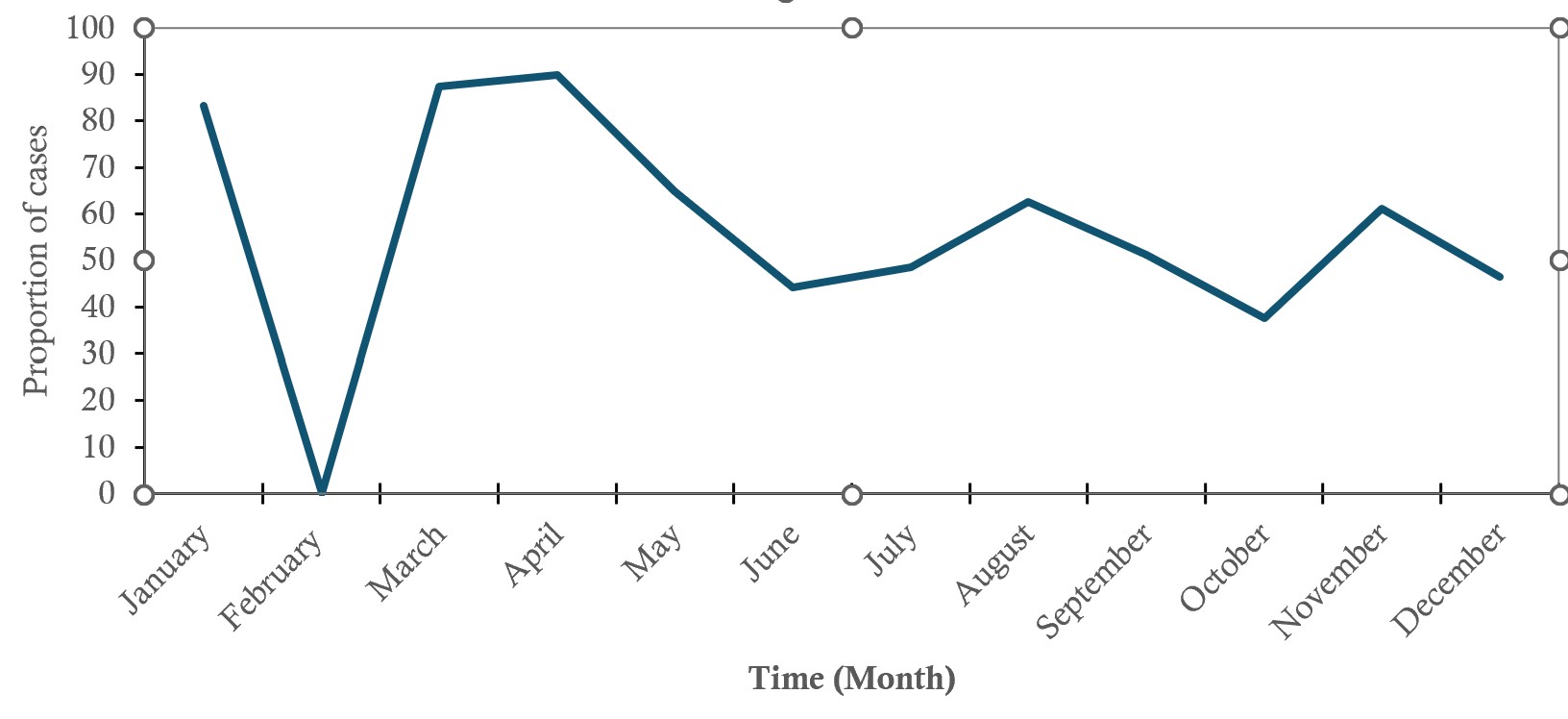
Trend in typhoid fever seropositivity
As shown in Figure 1, typhoid cases were recorded in each month except for February, 2022, which recorded zero positivity. Case positivity fluctuated over the period, with a sharp increase in positivity after February. The highest proportion of cases was recorded in the months of March 2022 and April 2022 (Figure 1).
Discussion
We analysed typhoid fever laboratory surveillance data in the West Gonja Municipality reported between 1st January, 2022 and 31st December, 2022. We found that the overall seroprevalence of typhoid fever was 52.3%, indicating a relatively high burden of typhoid infection. Variations were observed across age categories, sex and geographical locations. Among the age groups, individuals 61 years and above and those between 51 and 60 years had the highest positivity rates. In the sex analysis, we observed a higher positivity rate among the males compared to the females. There was no statistically significant association between demographic characteristics and typhoid infection status. Over the period there were fluctuations in positivity with the highest proportions in the months of March and April.
The overall seroprevalence of typhoid fever in the study population is high suggestive of high transmission relative to tested cases. This finding is much higher compared to the prevalence obtained in studies conducted in Eastern Ethiopia (11%) [18], Northwest Ethiopia (25.7%) [19], Ghana (6.5%) [9], Nigeria (14.1%) [20], Cameroon (30.1%) [21] and India (9.7%) [22]. Data available from the Ghana demographic and health survey suggest the lack of portable water and soap for handwashing among households in some parts of the study setting [23]. This situation might contribute to the high prevalence observed in the current study. Conversely, the prevalence in the current study is comparable to the prevalences obtained in a district-level study and among hospital patients in Ethiopia, 54.9% and 56.2%, respectively [24], [25]. Despite the varied epidemiology of typhoid fever, the variations in prevalence observed in the earlier studies could be attributed to the environmental differences, participants’ knowledge, attitude and perceptions about typhoid fever and diagnostic methods employed. For instance, a study among tertiary students in Nigeria using the Widal test and stool culture reported prevalences as high as 69.6% and 75.2%, respectively [26].
Concerning the age-specific distribution in this study, higher positivity rates were observed among the oldest age groups, 61 years and above and 51-60 years. Older age groups may have increased vulnerability due to weaker immune responses and a higher likelihood of comorbidities [27]. Children under 11 years and adults aged 31-40 years also recorded relatively high positivity rates. This finding is comparable with studies conducted in Cameroon [17], but in contrast with studies in Northwest Ethiopia [19] where persons below 30 years were the most infected. Our study also reported lower rates among individuals in the age group 41-50. These variations among the age groups are common in epidemiological studies and are influenced by immunity, exposure patterns, and behavioural risk factors [28]. Increased exposure in school settings among children and occupational or social mobility among the working-class adults may explain these findings. Moreover, the disparity observed with other studies could be linked with the environment, sample size and laboratory method differences. Overall, the age-related pattern in this study aligns with established epidemiological principles that disease occurrence is strongly influenced by age-related biological and social determinants.
Typhoid fever positivity rate was higher among men than women in this study. This finding agrees with a study conducted among febrile patients in Cameroon [21] but contrary to a study in Ethiopia [25].The observed sex differences in the current study may be attributed to behavioral, occupational and biological factors. Globally, sex differences in infectious diseases have been documented, with males often experiencing higher exposure risks due to occupational activities and health-seeking behaviours [29]. Studies have shown that men are less likely to seek early healthcare, which may lead to delayed diagnosis and higher positivity rates [30], [31]. In addition, biological differences in immune responses between males and females may influence susceptibility and disease outcomes. The observed difference in this study is therefore consistent with existing epidemiological literature indicating sex-based disparities.
Significant geographical variations were observed in the distribution of the case burden. Positivity rate was highest in the Dass sub-municipality followed by Canteen, whereas in Larabanga, no positive case was recorded. Such differences may reflect variations in population density, sanitation, environmental conditions, healthcare accessibility and local transmission dynamics. While areas with higher population concentration or limited public health infrastructure may experience increased transmissions rates, the availability of diagnostic services and data reporting systems could influence the interpretation of the pattern observed in this study. Moreover, the higher rate observed in Dass could be attributed to the small sample size tested as this could inflate percentage estimates. Nonetheless, the positivity rates observed in this study should be a source of concern particularly when drinking water quality in these settings has been reported to be suboptimal [32].
Though typhoid fever can be recorded throughout the year, the trend from this study indicates that the months of March and April had the highest positivity. While these periods should alert the health facilities to increase suspicion, typhoid fever remains a problem throughout the year. Moreover, testing was low over the period, and this could have affected the positivity observed in this study, particularly for February. The seasonal pattern of typhoid fever varies with geographical factors [33]. Studies conducted in Southwest Ethiopia reported similar findings, with the highest cases before the wet season [34]. In the context of our study setting, the period between the second and third quarters of the year experiences the highest rainfall with pockets of floods and overflows of riverbanks into nearby communities. People living in the communities utilize streams, ponds and rivers, possibly shared with animals, for household consumption. The situation increases the risk of transmission from person to person.
Limitations
Firstly, the results from this study cannot be generalised for the entire municipality since some individuals seek healthcare in health facilities where laboratory testing may not be available. Secondly, due to the overlapping symptoms between typhoid and other febrile illnesses, some cases may have been misdiagnosed. Lastly, the study did not include or consider the influence of other public health interventions, vaccinations, seasonality, mobility of people, and serotypes of typhoid bacteria. Therefore, the interpretation of this study should be done considering the above-mentioned limitations. Despite these limitations, our results offer useful information in settings where there is a lack of local laboratory capacity to confirm suspected typhoid fever using blood or stool culture and where there are inadequate resources to conduct population-based surveillance studies to establish true infection rates.
Conclusion
Typhoid fever seroprevalence is high in the West Gonja Municipality, with the most affected persons being males and the oldest aged groups 61 years and above, 51-60 years, and children under 11 years. Although testing was low throughout the period, the months of March and April received the highest positivity, with cases highly concentrated in the Dass and Canteen areas. We recommend the health authorities in the West Gonja Municipality to improve testing and reporting of typhoid fever. The Health Promotion Department should incorporate typhoid fever health education topics in health facilities and communities. The regional and national health divisions should institute laboratory culture-based surveillance to provide a detailed understanding of typhoid fever burden in the population. Further research is recommended to explore the socioeconomic and environmental impact on the typhoid fever seroprevalence.
What is already known about the topic
- Typhoid fever is a major public health problem in developing countries
- Typhoid fever prevalence is high in areas with poor access to portable water and improved sanitation facilities
What this study adds
- Typhoid fever seroprevalence is high in the West Gonja Municipality of Ghana
- Most affected persons with typhoid fever in the West Gonja Municipality are males, the aged groups 61 years and above, 51-60 years, and children under 11 years
Acknowledgements
The authors are grateful to the West Gonja Municipal Health Directorate for granting permission to conduct the research in the municipality. Special acknowledgement goes to the Ghana Field Epidemiology and Laboratory Training Programme for their guidance towards the writing of this manuscript.
Authors´ contributions
FUS, CK, JACN were responsible for the conceptualization and design of the study. FUS and SJA participated in data collection and analysis. FUS, CK and JACN drafted the manuscript. GRI, CK, GA, EK critically reviewed and equally contributed to the content of the manuscript. All authors read and approved the final manuscript.
| Table 1: Demographic characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022 | |||
|---|---|---|---|
| Variable | Suspected Cases N=2998 N (%) | Tested N=377 N (%) | Positivity % [95% CI] |
| Age (years), Mean (SD) | 30.8 (±18.2) | ||
| Age group | |||
| <11 | 546 (18.2) | 54 (14.3) | 55.6 [41.4-69.1] |
| 11-20 | 258 (8.6) | 65 (17.2) | 52.3 [39.5-64.9] |
| 21-30 | 415 (13.8) | 85 (22.6) | 44.7 [33.9-55.9] |
| 31-40 | 478 (15.9) | 80 (21.2) | 55.0 [43.5-66.2] |
| 41-50 | 566 (18.9) | 32 (8.5) | 40.6 [23.7-59.4] |
| 51-60 | 317 (10.6) | 31 (8.2) | 61.3 [42.2-78.2] |
| 61+ | 418 (13.9) | 30 (8.0) | 63.3 [43.9-80.1] |
| Sex | |||
| Female | 1,953 (65.1) | 206 (54.6) | 49.0 [42.0-56.1] |
| Male | 1,045 (34.9) | 171 (45.4) | 56.1 [48.4-63.7] |
| Sub-District | |||
| Busunu | 25 (0.8) | 5 (1.3) | 20.0 [0.5-71.6] |
| Canteen | 2085 (69.5) | 293 (77.7) | 55.3 [49.4-61.1] |
| DASS | 717 (23.9) | 6 (1.6) | 83.3 [35.9-99.6] |
| Damongo | 7 (0.2) | 71 (18.8) | 40.8 [29.3-53.2] |
| Larabanga | 164 (5.5) | 2 (0.5) | 0.0 [0.0-1.8] |
| Table 2: Seroprevalence and antibody characteristics of laboratory-tested cases of typhoid fever, West Gonja, Ghana, 2022, N=377 | |||
|---|---|---|---|
| Variable | Frequency | Percentage | 95% CI |
| Overall seroprevalence | 197 | 52.3 | 47.1-57.4 |
| Serotype | |||
| Both IgM & IgG Negative | 180 | 47.7 | 42.6-52.9 |
| Both IgM & IgG Positive | 62 | 16.5 | 12.8-20.6 |
| Only IgM Positive | 111 | 29.4 | 24.9-34.3 |
| Only IgG Positive | 24 | 6.4 | 4.1-9.3 |
| Table 3: Age, sex and Sub-municipal association with typhoid fever antibody status in the West Gonja Municipality, Ghana, 2022 | ||||
|---|---|---|---|---|
| Variable | Typhoid Positive N=197 n (%) | Typhoid Negative N=180 n (%) | Crude OR (95% CI) | p-value |
| Age (years) | ||||
| <11 | 30 (15.2) | 24 (13.3) | 1 | |
| 11-20 | 34 (17.3) | 31 (17.2) | 1.14 (0.55-2.35) | 0.724 |
| 21-30 | 38 (19.3) | 47 (26.1) | 1.54 (0.78-3.07) | 0.213 |
| 31-40 | 44 (22.3) | 36 (20.0) | 1.02 (0.51-2.05) | 0.949 |
| 41-50 | 13 (6.6) | 19 (10.6) | 1.83 (0.75-4.43) | 0.183 |
| 51-60 | 19 (9.6) | 12 (6.7) | 0.79 (0.32-1.94) | 0.607 |
| 61+ | 19 (9.6) | 11 (6.1) | 0.72 (0.29-1.81) | 0.489 |
| Sex | ||||
| Female | 101 (51.3) | 105 (58.3) | 1 | |
| Male | 96 (48.7) | 75 (41.7) | 0.75 (0.50-1.13) | 0.169 |
| Sub-District | ||||
| Busunu | 1 (0.5) | 4 (2.2) | 1 | |
| Canteen | 162 (82.2) | 131 (72.8) | 0.20 (0.02-1.83) | 0.155 |
| DASS | 5 (2.5) | 1 (0.6) | 0.05 (0.00-1.07) | 0.056 |
| Damongo | 29 (14.7) | 42 (23.3) | 0.36 (0.04-3.47) | 0.374 |
| Larabanga* | 0 (0.0) | 2 (1.1) | – | |
| * Larabanga was omitted from the analysis due to no data for typhoid positive cases, hence its COR could not be estimated by the software. | ||||
References
- World Health Organization. Typhoid [Internet]. Geneva (Switzerland): World Health Organization; 2023 Mar 20 [cited 2026 Mar 06]. Available from: https://www.who.int/news-room/fact-sheets/detail/typhoid
- Stanaway JD, Reiner RC, Blacker BF, Goldberg EM, Khalil IA, Troeger CE, Andrews JR, Bhutta ZA, Crump JA, Im J, Marks F, Mintz E, Park SE, Zaidi AKM, Abebe Z, Abejie AN, Adedeji IA, Ali BA, Amare AT, Atalay HT, Avokpaho EFGA, Bacha U, Barac A, Bedi N, Berhane A, Browne AJ, Chirinos JL, Chitheer A, Dolecek C, El Sayed Zaki M, Eshrati B, Foreman KJ, Gemechu A, Gupta R, Hailu GB, Henok A, Hibstu DT, Hoang CL, Ilesanmi OS, Iyer VJ, Kahsay A, Kasaeian A, Kassa TD, Khan EA, Khang YH, Magdy Abd El Razek H, Melku M, Mengistu DT, Mohammad KA, Mohammed S, Mokdad AH, Nachega JB, Naheed A, Nguyen CT, Nguyen HLT, Nguyen LH, Nguyen NB, Nguyen TH, Nirayo YL, Pangestu T, Patton GC, Qorbani M, Rai RK, Rana SM, Ranabhat CL, Roba KT, Roberts NLS, Rubino S, Safiri S, Sartorius B, Sawhney M, Shiferaw MS, Smith DL, Sykes BL, Tran BX, Tran TT, Ukwaja KN, Vu GT, Vu LG, Weldegebreal F, Yenit MK, Murray CJL, Hay SI. The global burden of typhoid and paratyphoid fevers: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect Dis [Internet]. 2019 Apr [cited 2026 Mar 06];19(4):369–81. doi:10.1016/S1473-3099(18)30685-6
- Hancuh M, Walldorf J, Minta AA, Tevi-Benissan C, Christian KA, Nedelec Y, Heitzinger K, Mikoleit M, Tiffany A, Bentsi-Enchill AD, Breakwell L. Typhoid Fever Surveillance, Incidence Estimates, and Progress Toward Typhoid Conjugate Vaccine Introduction — Worldwide, 2018–2022. MMWR Morb Mortal Wkly Rep [Internet]. 2023 Feb 17 [cited 2026 Mar 06];72(7):171–6. doi:10.15585/mmwr.mm7207a2
- Ministry of Health (Ghana). Health sector annual programme of work: 2021 holistic assessment report [Internet]. Accra (Ghana): Ministry of Health (Ghana); 2022 Apr [cited 2026 Mar 06]. 77 p. Available from: https://www.moh.gov.gh/wp-content/uploads/2022/09/2021-Holistic-Assessment-Report_v1.7.3.pdf
- Ghana Health Service. The health sector in Ghana facts and figures [Internet]. Accra (Ghana): Ghana Health Service; 2015 [cited 2026 Mar 06]. 47 p. Available from: https://www.moh.gov.gh/wp-content/uploads/2017/07/Facts-and-figures-2015.pdf
- Cruz Espinoza LM, Nichols C, Adu-Sarkodie Y, Al-Emran HM, Baker S, Clemens JD, Dekker DM, Eibach D, Krumkamp R, Boahen K, Im J, Jaeger A, Von Kalckreuth V, Pak GD, Panzner U, Park SE, Park JK, Sarpong N, Schütt-Gerowitt H, Toy T, Wierzba TF, Marks F, May J. Variations of Invasive Salmonella Infections by Population Size in Asante Akim North Municipal, Ghana. Clin Infect Dis [Internet]. 2016 Mar 15 [cited 2026 Mar 06];62(suppl 1):S17–22. doi:10.1093/cid/civ787
- Carey ME, Steele AD. The Severe Typhoid Fever in Africa Program Highlights the Need for Broad Deployment of Typhoid Conjugate Vaccines. Clin Infect Dis [Internet]. 2019 Oct 30 [cited 2026 Mar 06];69(Supplement_6):S413–6. doi:10.1093/cid/ciz637
- World Health Organization. Background paper to sage on typhoid vaccine policy recommendations [Internet]. Geneva (Switzerland): World Health Organization; 2017 Sep 27 [cited 2026 Mar 06]. 87 p. Available from: https://cdn.who.int/media/docs/default-source/immunization/position_paper_documents/typhoid/1-typhoid-sage-background-paper-final-v3b.pdf?sfvrsn=ddf418c3_2
- Labi AK, Obeng-Nkrumah N, Addison NO, Donkor ES. Salmonella blood stream infections in a tertiary care setting in Ghana. BMC Infect Dis [Internet]. 2014 Dec [cited 2026 Mar 06];14(1):3857. doi:10.1186/s12879-014-0697-7
- Fusheini A, Gyawu SK. Prevalence of Typhoid and Paratyphoid Fever in the Hohoe Municipality of the Volta Region, Ghana: A Five-Year Retrospective Trend Analysis. Ann Glob Health [Internet]. 2020 Sep 3 [cited 2026 Mar 06];86(1):111. doi:10.5334/aogh.2833
- Mawazo A, Bwire GM, Matee MIN. Performance of Widal test and stool culture in the diagnosis of typhoid fever among suspected patients in Dar es Salaam, Tanzania. BMC Res Notes [Internet]. 2019 Jun 5 [cited 2026 Mar 06];12(1):316. doi:10.1186/s13104-019-4340-y
- Keddy K, Sooka A, Letsoalo M, Hoyland G, Chaignat CL, Morrissey A, Crump J. Sensitivity and specificity of typhoid fever rapid antibody tests for laboratory diagnosis at two sub-Saharan African sites. Bull World Health Organ [Internet]. 2011 Sep 1 [cited 2026 Mar 06];89(9):640–7. doi:10.2471/BLT.11.087627
- Mengist HM, Tilahun K. Diagnostic Value of Widal Test in the Diagnosis of Typhoid Fever: A Systematic Review. J Med Microb Diagn [Internet]. 2017 Jun [cited 2026 Mar 06];06(01). doi:10.4172/2161-0703.1000248
- Salama RI, Said NM. A Comparative Study of the Typhidot (Dot-EIA) versus Widal Test in Diagnosis of Typhoid Fever among Egyptian Patients. OJGas [Internet]. 2019 Jun [cited 2026 Mar 06];09(06):91–8. doi:10.4236/ojgas.2019.96011
- Norsiah W, Oktiyani N. Evaluation of the Diagnosis of Typhoid Fever Using the Widal Test and the Anti Salmonella typhi IgM Test. MedLabTechJ [Internet]. 2020 Nov 29 [cited 2026 Mar 06];6(2):128. doi:10.31964/mltj.v0i0.327
- Ghana Statistical Service. Population and Housing Census (PHC) General Report Population Regions and Districts [Internet]. Accra (Ghana): Ghana Statistical Service; 2021 [cited 2026 Mar 06].
- Ousenu K, Ali IM, Sama LF, Ndam MN, Tchouangueu TF, Tume CB. A Cross-Sectional Comparative Study of the Performance of the Widal Test and the Typhidot Immunoassay for Typhoid Fever Diagnosis in the West Region of Cameroon. Can J Infect Dis Med Microbiol [Internet]. 2021 Aug 7 [cited 2026 Mar 06];2021:1–9. doi:10.1155/2021/8279122
- Admassu D, Egata G, Teklemariam Z. Prevalence and antimicrobial susceptibility pattern of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi among febrile patients at Karamara Hospital, Jigjiga, eastern Ethiopia. SAGE Open Med [Internet]. 2019 Jan [cited 2026 Mar 06];7:2050312119837854. doi:10.1177/2050312119837854
- Mulu W, Akal CG, Ababu K, Getachew S, Tesfaye F, Wube A, Chekol D. Seroconfirmed Typhoid Fever and Knowledge, Attitude, and Practices among Febrile Patients Attending at Injibara General Hospital, Northwest Ethiopia. BioMed Res Int [Internet]. 2021 Jan [cited 2026 Mar 06];2021(1):8887266. doi:10.1155/2021/8887266
- Ohanu ME. Typhoid fever among febrile Nigerian patients: Prevalence, diagnostic performance of the Widal test and antibiotic multi-drug resistance. Mal Med J [Internet]. 2019 Sep 3 [cited 2026 Mar 06];31(3):184–92. doi:10.4314/mmj.v31i3.4
- Njoya HF, Awolu MM, Christopher TB, Duclerc JF, Ateudjieu J, Wirsiy FS, Atuhaire C, Cumber SN. Prevalence and awareness of mode of transmission of typhoid fever in patients diagnosed with Salmonella typhi and paratyphi infections at the Saint Elisabeth General Hospital Shisong, Bui Division, Cameroon. Pan Afr Med J [Internet]. 2021 Oct 7 [cited 2026 Mar 06];40:83. doi:10.11604/pamj.2021.40.83.16893
- John J, Van Aart CJC, Grassly NC. The Burden of Typhoid and Paratyphoid in India: Systematic Review and Meta-analysis. PLoS Negl Trop Dis [Internet]. 2016 Apr 15 [cited 2026 Mar 06];10(4):e0004616. doi:10.1371/journal.pntd.0004616
- Ghana Statistical Service, Ghana Health Service, ICF International. Ghana Demographic and Health Survey 2014 [Internet]. Rockville (Maryland): ICF International; 2015 [cited 2026 Mar 06]. 530 p. Available from: https://dhsprogram.com/pubs/pdf/fr307/fr307.pdf
- Eba K, Bekele D. Prevalence of Typhoid Fever and its Risk Factors in Lalo Assabi District, West Wollega, Oromiya, Ethiopia. J Bacteriol Parasitol [Internet]. [cited 2026 Mar 06];10:365. doi:10.35248/2155-9597.19.10.365
- Deksissa T, Gebremedhin EZ. A cross-sectional study of enteric fever among febrile patients at Ambo hospital: prevalence, risk factors, comparison of Widal test and stool culture and antimicrobials susceptibility pattern of isolates. BMC Infect Dis [Internet]. 2019 Dec [cited 2026 Mar 06];19(1):288. doi:10.1186/s12879-019-3917-3
- Abioye J, Adiuku B, Adogo L. The prevalence of typhoid fever in Bingham University. GSC Biol Pharm Sci [Internet]. 2017 Dec 30 [cited 2026 Mar 06];1(3):037–43. doi:10.30574/gscbps.2017.1.3.0052
- Centers for Disease Control and Prevention (GA), Office of Workforce and Career Development (GA). Principles of Epidemiology in Public Health Practice: An Introduction to Applied Epidemiology and Biostatistics: Third Edition: Self-Study Course SS1978 [Internet]. Atlanta (Georgia): Centers for Disease Control and Prevention; 2012 May 1 [cited 2026 Mar 06]. 511 p. Available from: https://stacks.cdc.gov/view/cdc/13178
- Quiros-Roldan E, Sottini A, Natali PG, Imberti L. The Impact of Immune System Aging on Infectious Diseases. Microorganisms [Internet]. 2024 Apr 11 [cited 2026 Mar 06];12(4):775. doi:10.3390/microorganisms12040775
- Biswas A, Tiong M, Irvin E, Zhai G, Sinkins M, Johnston H, Yassi A, Smith PM, Koehoorn M. Gender and sex differences in occupation-specific infectious diseases: a systematic review. Occup Environ Med [Internet]. 2024 Aug [cited 2026 Mar 06];81(8):425–32. doi:10.1136/oemed-2024-109451
- Teo CH, Ng CJ, Booth A, White A. Barriers and facilitators to health screening in men: A systematic review. Soc Sci Med [Internet]. 2016 Aug 1 [cited 2026 Mar 06];165:168–76. doi:10.1016/j.socscimed.2016.07.023
- Christie-de Jong F, Oyeniyi OS, Nnyanzi LA, Ling J, Murphy MK, Eberhardt J, Jarrar R, Kabuye J, Kalemba M, Robb KA. Barriers and facilitators to accessing healthcare for early diagnosis of prostate cancer for black men—a qualitative exploration in North-East England and Scotland. BMC Public Health [Internet]. 2025 Jul 14 [cited 2026 Mar 06];25(1):2454. doi:10.1186/s12889-025-23650-y
- Amuah EEY, Bekoe EMO, Kazapoe RW, Dankwa P, Nandomah S, Douti NB, Abanyie SK, Okyere IK. Sachet water quality and Vendors’ practices in Damongo, northern Ghana during the emergence of SARS-CoV-2 using multivariate statistics, water quality and pollution indices, and panel assessment. Environ Challenges [Internet]. 2021 Aug [cited 2026 Mar 06];4:100164. doi:10.1016/j.envc.2021.100164
- Saad NJ, Lynch VD, Antillón M, Yang C, Crump JA, Pitzer VE. Seasonal dynamics of typhoid and paratyphoid fever. Sci Rep [Internet]. 2018 May 2 [cited 2026 Mar 06];8(1):6870. doi:10.1038/s41598-018-25234-w
- Atikilt Yemata G, Yenew C, Mamuye M, Tiruneh M, Assfaw T, Mulatu S, Sisay E, Tadele F. Descriptive Analysis of Typhoid Fever Surveillance Data in the Jimma Zone, Southwest Ethiopia (2015–2019). Interdiscip Perspect Infect Dis [Internet]. 2021 Dec 13 [cited 2026 Mar 06];2021:1–9. doi:10.1155/2021/1255187
