Research | Open Access | Volume 9 (1): Article 41 | Published: 09 Mar 2026
Factors influencing vaccination coverage for the birth dose of hepatitis B vaccine in Kabadougou, Côte d’Ivoire, 2025: Analytical cross-sectional study
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Table 1: Description of infant vaccination service and resources, Kabadougou, 2025 | ||
|---|---|---|
| Variables | Frequency (n=15) | Percentage (%) |
| Type of health centre | ||
| Rural | 9 | 60 |
| Urban | 6 | 40 |
| Delivery service offered | ||
| Yes | 13 | 86.7 |
| No | 2 | 13.3 |
| Offer of the HepB-BD by the health centre (Yes) | 15 | 100 |
| Shortage of vaccine stocks in health centres over the last twelve months | ||
| HepB-BD (No) | 15 | 100 |
| BCG (No) | 15 | 100 |
| VPO0 (No) | 15 | 100 |
| Functional refrigerator for vaccines available in the health centre (Yes) | 15 | 100 |
| Refrigerator breakdown during the last 12 months (No) | 15 | 100 |
| Mean year of health worker experience in vaccination ± SD* (years) | 5.2 ± 3.8 | |
| Mean number of health workers trained in HepB-BD vaccination per facility ± SD (person) | 5 ± 4 | |
| SD*: Standard Deviation; HepB-BD: Birth dose of hepatitis B vaccine; BCG: Bacillus Calmette-Guerin Vaccine; VPO0: Zero dose of oral polio vaccine | ||
Table 1: Description of infant vaccination service and resources, Kabadougou, 2025
| Table 2: Bivariate analysis: sociodemographic characteristics of child guardians and non-vaccination against hepatitis at birth, Kabadougou, 2025 | ||||
|---|---|---|---|---|
| Variables | HepB-BD status | cOR (95% CI) | p-value | |
| Did not receive HepB n=142 n(%) | Received HepB-BD n=308 n(%) | |||
| Guardian’s Occupation | ||||
| Income-generating activity | 78 (54.9) | 112 (36.4) | 2.1 (1.4-3.2) | <0.001 |
| No-Income-generating activity | 64 (45.1) | 196 (63.6) | ||
| Mother’s Occupation | ||||
| Income-generating activity | 72 (50.7) | 100 (32.5) | 2.2 (1.4-3.2) | <0.001 |
| No-Income-generating activity | 70 (49.3) | 208 (67.5) | ||
| Guardian’s marital status | ||||
| Married/cohabiting | 108 (76.1) | 293 (95.1) | 0.2 (0.1-0.3) | <0.001 |
| Single | 34 (23.9) | 15 (4.9) | ||
| Mother’s marital status | ||||
| Married/cohabiting | 33 (23.2) | 15 (4.9) | 5.9 (3.1-11.3) | <0.001 |
| Single | 109 (76.8) | 293 (95.1) | ||
| Type of residence | ||||
| Urban | 84 (59.2) | 126 (40.9) | 2.1 (1.4-3.1) | <0.001 |
| Rural | 58 (40.8) | 182 (59.1) | ||
| Monthly household income according to the guaranteed minimum wage | ||||
| ≤75 000 Fcfa* | 116 (81.7) | 276 (89.6) | 0.5 (0.3-0.9) | 0.021 |
| >75 000 Fcfa | 26 (18.3) | 32 (10.4) | ||
| Reported history of an AEFI in a child from the guardian’s social network | ||||
| Yes | 13 (9.2) | 57 (18.5) | 0.4 (0.2-0.8) | 0.011 |
| No | 129 (90.8) | 251 (81.5) | ||
| Median guardian age in years (Interquartile range) | 30 (12) | 27 (10) | — | 0.007 |
| Median mother age in years (Interquartile range) | 28 (11) | 26 (9) | — | 0.246 |
| Median father age in years (Interquartile range) | 39 (13) | 38 (11) | — | 0.539 |
| Median child age in years (Interquartile range) | 2.2 (3.3) | 1.4 (2.6) | — | 0.013 |
| Fcfa*: African Financial Community Franc; (1): Chi-square test; (2): Mann-Whitney U test | ||||
Table 2: Bivariate analysis: sociodemographic characteristics of child guardians and non-vaccination against hepatitis at birth, Kabadougou, 2025
| Table 3: Bivariate analysis: Perinatal care and guardian awareness associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025 | ||||
|---|---|---|---|---|
| Variables | HepB-BD status | cOR (95% CI) | p-value | |
| Did not receive HepB n=142 n(%) | Received HepB-BD n=308 n(%) | |||
| Receipt of the mother-child health record book | ||||
| Yes | 112 (78.9) | 304 (98.7) | 0.05 (0.01-0.1) | <0.001 |
| No | 30 (21.1) | 4 (1.3) | ||
| Term birth (≥37 weeks gestation) | ||||
| Yes | 135 (95.1) | 305 (99) | 0.2 (0.05-0.7) | 0.021 |
| No | 7 (4.9) | 3 (1) | ||
| Place of delivery | ||||
| Home | 28 (19.7) | 10 (3.2) | 7.3 (3.4-15.6) | <0.001 |
| Health facility | 114 (80.3) | 298 (96.8) | ||
| Antenatal care visits attended by the mother | ||||
| Yes | 117 (82.4) | 304 (98.7) | 0.06 (0.02-0.2) | <0.001 |
| No | 25 (17.6) | 4 (1.3) | ||
| Mother received information on HepB-BD during antenatal care | ||||
| Yes | 50 (35.2) | 205 (66.6) | 0.4 (0.3-0.5) | <0.001 |
| No | 92 (64.8) | 103 (33.4) | ||
| Guardian’s knowledge of the existence of hepatitis B vaccine | ||||
| Yes | 47 (33.1) | 169 (54.9) | 0.4 (0.3-0.6) | <0.001 |
| No | 94 (66.9) | 139 (45.1) | ||
| Guardian’s knowledge of the HepB-BD | ||||
| Yes | 39 (27.5) | 164 (53.2) | 0.3 (0.2-0.5) | <0.001 |
| No | 103 (72.5) | 144 (46.8) | ||
| Guardian’s knowledge of the AEFI | ||||
| Yes | 44 (31) | 153 (49.7) | 0.5 (0.3-0.7) | <0.001 |
| No | 98 (69) | 155 (50.3) | ||
| Existence of traditional newborn care practices in your community | ||||
| Yes | 53 (37.3) | 79 (25.6) | 1.7 (1.1-2.6) | 0.011 |
| No | 89 (62.7) | 229 (74.4) | ||
| Existence of local beliefs concerning vaccination | ||||
| Yes | 35 (24.6) | 23 (7.5) | 4.1 (2.3-7.2) | <0.001 |
| No | 107 (75.4) | 285 (92.5) | ||
Table 3: Bivariate analysis: Perinatal care and guardian awareness associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025
| Table 4: Binary logistic regression of factors associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025 | |||
|---|---|---|---|
| Variables | Non-vaccination of HepB-BD | ||
| aOR | 95% CI | P value | |
| Child’s age at survey (years) | 1.4 | 1.2-1.7 | <0.001 |
| Guardian’s marital status | |||
| Married/Cohabiting | 0.3 | 0.1-0.7 | 0.005 |
| Single | 1 | ||
| Place of delivery | |||
| Home | 5.7 | 2.3-14.6 | <0.001 |
| Health facility | 1 | ||
| Guardian’s knowledge of the AEFI | |||
| Yes | 0.4 | 0.2-0.7 | 0.001 |
| No | 1 | ||
| Receipt of the mother-child health record book | |||
| Yes | 0.1 | 0.04-0.5 | 0.003 |
| No | 1 | ||
| Mother received information on HepB-BD during antenatal care | |||
| Yes | 0.3 | 0.2-0.5 | <0.001 |
| No | 1 | ||
| Existence of local beliefs concerning vaccination | |||
| Yes | 4.7 | 2.3-9.7 | <0.001 |
| No | 1 | ||
| aOR: Adjusted Odds Ratio | |||
Table 4: Binary logistic regression of factors associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025
Figures
Keywords
- Hepatitis B birth dose
- Vaccination coverage
- Mother-to-child transmission
- Kabadougou
- Côte d’Ivoire
Bi Tahié Aubain Hué1,2,&, Aimée Lulebo Mampasi3, Lepri Bernadin Nicaise Aka1,4, Jean Bosco Kasonga Ngindu3, Zolou Marie Tia1,2, Pierre Wilnique2,5, Pegnontaye Moussa Soro2,6, Innocent Kedja7, Isaac Tiembré2,4,6
1Épidémiological Surveillance Service, Directorate for the Coordination of the Expanded Vaccination Program, Abidjan, Côte d’Ivoire, 2Field Epidemiology Training Program, Abidjan, Côte d’Ivoire, 3Département Épidémiologie et Biostatistique, École de Santé Publique de Kinshasa, Kinshasa, République Démocratique du Congo, 4Public health, University Felix Houphouët Boigny, Abidjan, Côte d’Ivoire, 5African Field Epidemiology Network, Abidjan, Côte d’Ivoire, 6Epidemiological Surveillance Service, National Institute of Public Hygiene, Abidjan, Côte d’Ivoire, 7Regional Health Directorate of Kabadougou, Côte d’Ivoire
&Corresponding author: Bi Tahié Aubain Hué, Field Epidemiology Training Program, Abidjan, Côte d’Ivoire, Email: docteur.hbta@gmail.com, ORCID: https://orcid.org/0009-0008-8377-1172
Received: 04 Nov 2025, Accepted: 07 Mar 2026, Published: 09 Mar 2026
Domain: Vaccine Preventable Diseases
Keywords: Hepatitis B birth dose, Vaccination coverage, mother-to-child transmission, Kabadougou, Côte d’Ivoire
©Bi Tahié Aubain Hué et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Bi Tahié Aubain Hué et al., Factors influencing vaccination coverage for the birth dose of hepatitis B vaccine in Kabadougou, Côte d’Ivoire, 2025: Analytical cross-sectional study. Journal of Interventional Epidemiology and Public Health. 2026;9(1):41. https://doi.org/10.37432/jieph-d-25-00268
Abstract
Introduction: The hepatitis B birth dose (HepB-BD) vaccine prevents mother-to-child transmission of the virus. The World Health Organization recommends its administration within 24 hours after birth, targeting 90% vaccination coverage (VC). In the Kabadougou region, administrative VC ranged from 49% to 71% between 2020 and 2023, remaining below national targets. This study aimed to identify the factors associated with this low coverage.
Methods: A cross-sectional analytical study was conducted in August 2025 on 450 children born between 2020 and 2025 in Kabadougou, selected by two-stage cluster sampling. Sociodemographic and vaccination data were collected using a KoboCollect questionnaire through interviews, observations, and document review. The outcome variable was non-vaccination with the HepB-BD within three days after birth. Data were analyzed using binary logistic regression in SPSS-27 with α= 5% (p<0.05).
Results: HepB-BD vaccination coverage was 68.4% (95% CI: 64.1%-72.7%). No vaccine stockout was reported. On average, five trained health workers were available per health centre (standard deviation: four). Factors significantly associated with non-vaccination were: home birth (adjusted odds ratio [aOR]=5.7; 95% confidence interval [95% CI]:2.3-14.6), each one-year increase in the child’s age (aOR=1.4; 95% CI: 1.2-1.7), unfavourable local beliefs regarding the birth dose (aOR = 4.7; 95% CI: 2.3-9.7), receipt of a vaccination record (aOR=0.1; 95% CI: 0.04-0.5), and maternal counselling on the HepB-BD received during antenatal care (aOR =0.3; 95% CI: 0.2-0.5).
Conclusion: The HepB-BD coverage in Kabadougou remains low, driven by home births. Improvement requires promoting facility-based deliveries, community sensitization, and systematic provision of vaccination records.
Introduction
Vaccination is one of the most effective public health interventions and a cornerstone of primary health care. It reduces neonatal morbidity and mortality, particularly in sub-Saharan Africa [1–4]. To prevent mother-to-child transmission (MTCT) of the hepatitis B virus (HBV), the World Health Organization (WHO) recommends administration of the Hepatitis B Birth Dose (HepB-BD) within the first 24 hours of life [4–7].
Hepatitis B is a serious viral infection that can progress to cirrhosis or hepatocellular carcinoma [8]. In Africa, MTCT represents the predominant route of HBV transmission. To address this burden, the World Health Assembly set a target of achieving 90% coverage of HepB-BD by 2030 [8]. However, timely global coverage reached only 42% in 2021, below the 50% target set for 2020, while the WHO African Region reported coverage as low as 17% in the same year [9].
Côte d’Ivoire introduced HepB-BD into its Expanded Program on Immunisation in October 2019 [9-12]. Since its introduction, national coverage has remained low and variable, increasing from 9% in 2019 to 65% in 2020 and 66% in 2021 [8,11,13]. In the Kabadougou health region of the northwest of Côte d’Ivoire, reported administrative coverage for the HepB-BD remained below the national target of 95% between 2020 and 2023, fluctuating between 49% and 71% [14]. Previous studies have identified home birth, along with socio-economic and cultural constraints, as key barriers to timely HepB-BD uptake [8,12].
These suboptimal coverage levels sustain a high risk of neonatal HBV transmission. In this context, identifying the factors that influence uptake of the HepB-BD is essential. We therefore determined the prevalence of HepB-BD and factors associated with its uptake for children born between January 2020 and August 2025 in the Kabadougou health region.
Methods
Study setting and period
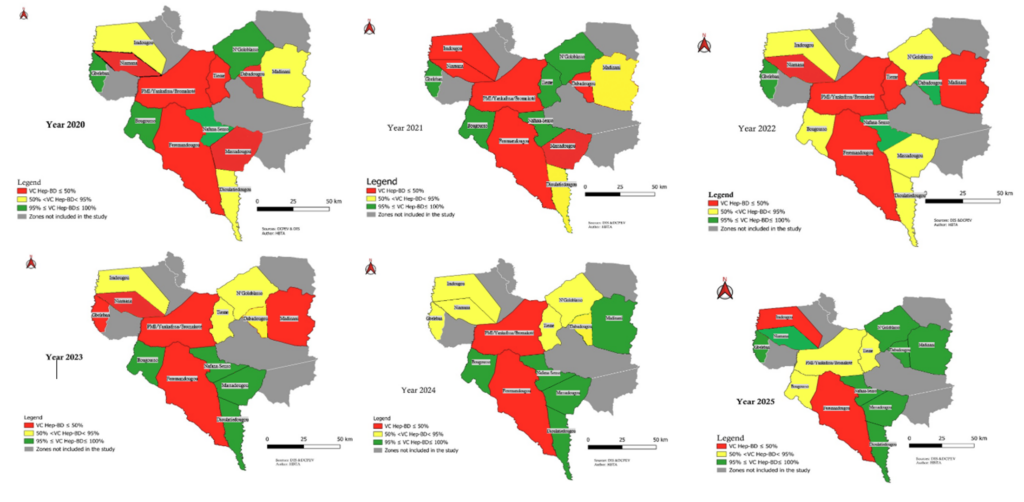
The study was conducted in the Kabadougou health region in Côte d’Ivoire in August 2025. This predominantly rural region comprises 73 health centres distributed across two health districts: Odienné and Madinani [14] (Figure 1). This was a cross-sectional analytical study using data on children born between January 2020 and August 2025.
Population and sampling
The minimum sample size was 388 children. This minimum size was obtained using the sample size estimation formula used by WHO in the cluster survey on immunisation coverage [15]. A design effect was applied to account for Intra-Cluster Correlation (ICC). Sample size was calculated as n = A x B x C. A = 1 stratum, B = Effective Sample Size = 53 (from WHO tables: 95% expected coverage, 10% precision, α = 5%, power = 80%), C = Design effect = 7.33 (calculated as 1 + (m-1) x ICC, with m = 20 minimum respondents per cluster and ICC = 0,333 (standard WHO value). A two-stage cluster sampling method was used. At the first stage, 15 clusters (health areas) were randomly selected from the Odienné and Madinani districts using probability proportional to population size. At the second stage, within each cluster, a census of households with children born between 2020 and 2025 was conducted, followed by a systematic random sampling of households to enrol 30 children per cluster, yielding a total sample of 450 children. Only one eligible child was included per household, selected by simple random sampling when more than one child met the eligibility criteria. If the child’s caregiver was absent, up to three revisits were made before classifying the household as a non-respondent and proceeding to the next household in the sample series without replacement.
Inclusion and exclusion criteria
The study included all live births during the study period residing in the Kabadougou region. The mother or caregiver was required to be available for the interview. Children who were absent at the time of data collection were excluded. Children whose guardians were unable to provide information were also excluded.
Variables of interest
The outcome variable was non-vaccination against hepatitis B at birth. The primary endpoint was not receiving the birth dose of the hepatitis B vaccine within three days (72 hours). This is a dichotomous variable coded as follows: “1” = (Event of interest): Non-vaccination with birth dose for hepatitis B (HepB-BD) and “0” = (Reference group): Vaccination with HepB-BD vaccine. This timeframe corresponds to the operational definition used by WHO for timely vaccination [5,16–18]. The independent variables included socio-demographic and economic characteristics. Perinatal factors, such as the place of delivery, were studied. Caregiver knowledge, beliefs, and perceptions were also explored. The coverage estimates were derived from the survey sample of 450 children and not from routine administrative data. For each health area and each birth year (2020-2025), the denominator was defined as the number of samples of children born alive in that year within the specific health area. The numerator was the number of those children who received the hepatitis B birth dose. Coverage was calculated as the proportion of vaccinated children among the sampled children in each health area and year.
Measurement of specific variables:
The variable “unfavourable local beliefs” was assessed using a set of five dichotomous (yes/no) items addressing community perceptions previously documented in the literature as barriers to newborn vaccination [19, 20]. Respondents were asked whether they agreed with the following statements: (1) “Vaccination at birth can weaken the newborn”; (2) “Vaccine-preventable diseases are determined by fate or divine will”; (3) “Traditional remedies are more effective or safer than vaccines for protecting newborns”; (4) “Vaccination may cause infertility in children”; (5) “Vaccines are a Western invention incompatible. with our cultural values”. Each “yes” response counted as one point. A cumulative score of ≥3 positive responses was used to classify the respondent as having unfavourable local beliefs. The variable “Knowledge of Adverse Event Following Immunisation (AEFI)” was assessed through an open-ended question (“Can you name any effects after vaccination?”) Followed by three verification questions on specific symptoms (fever, pain at the injection site, irritability). This approach, consistent with Knowledge, Attitudes and Practices (KAP) survey methodology, allowed the evaluation of both spontaneous recall and recognition of typical AEFI [19,21,22]. Respondents were classified as having adequate knowledge if they spontaneously mentioned at least one AEFI or correctly identified at least two of the three verification items; otherwise, knowledge was considered inadequate
Receipt of the mother-child health record book: dichotomous variable indicating whether the mother or guardian received the record book issued by health personnel either during the first antenatal care contact or childbirth. The variable was coded ‘Yes’ when the respondent presented the booklet during the survey or reported having it, and ‘No’ when no booklet had been received or was available.
Data collection
Three complementary techniques were used. First, structured face-to-face interviews were conducted using a standardized KoboCollect questionnaire with guardians of eligible children, as well as with 15 health workers (one each selected health centre). A guardian was defined as the person most knowledgeable about the child’s vaccination and responsible for the child’s daily care, including possession of the vaccination card or recall of received vaccines. This could include the mother, father, legal guardian, or another adult household member. Second, direct observation was carried out to assess material and organizational issues, including the functionality of the cold chain and the availability and quality of routine documentation. Third, a documentary review was performed to verify and validate the information reported by respondents. Vaccination-related records, including the mother-child health record book, birth registers, and vaccination registers, were consulted for triangulation. The mother-child health record book is a nationally standardized tool used for integrated maternal and child health monitoring. It records key information on antenatal care, childbirth details, postnatal follow-up, growth and nutritional status, and the child’s vaccination schedule [23].
For health centres, the questionnaire captured the number of staff currently posted at each facility who had received formal training on HepB-BD administration and counselling. A pre-test of the data collection tools was conducted with 30 participants in the Seydougou health area, who were not included in the study.
Data processing and analysis
Data processing included sorting, coding, and quality checks. Analyses were performed using the software Statistical Package for Social Sciences -27 (SPSS-27), accounting for the cluster sampling design. Descriptive statistics were generated as proportions with their 95% confidence intervals (95% CI) for categorical variables. For quantitative variables that were not normally distributed, the median and interquartile ranges were reported. The variable “Staff experience with immunisation” was normally distributed and was therefore summarised using the mean and standard deviation (SD).
Bivariate analysis used Pearson’s chi-square test, Yates’ corrected chi-square test, or Fisher’s exact test for categorical variables. The Mann-Whitney U test was applied for comparisons involving non-normally distributed quantitative variables (age of children and parents). Variables with a p-value less than 0.25 in bivariate analysis were included in multivariate analysis. Multivariate analysis was performed using binary logistic regression with a conditional bottom-up approach to estimate adjusted odds ratios (aOR) and identify factors independently associated with non-vaccination with HepB-BD. Statistical significance was set to α = 5% (p < 0.05).
Ethical considerations
The study protocol was submitted to and approved by the National Life Sciences and Health Ethics Committee (CNESVS). (approval number: N° 00133 25/MSHPCMU/CNESVS). Verbal informed consent was obtained from all participants. For illiterate participants, consent was obtained verbally after the study objectives and procedures were explained in the local language (Malinké).
Results
Estimated vaccination coverage
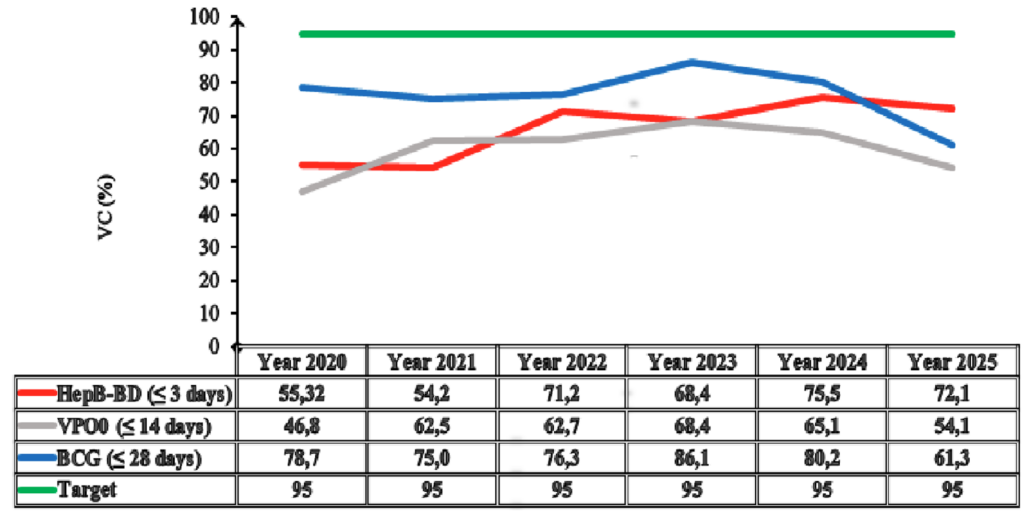
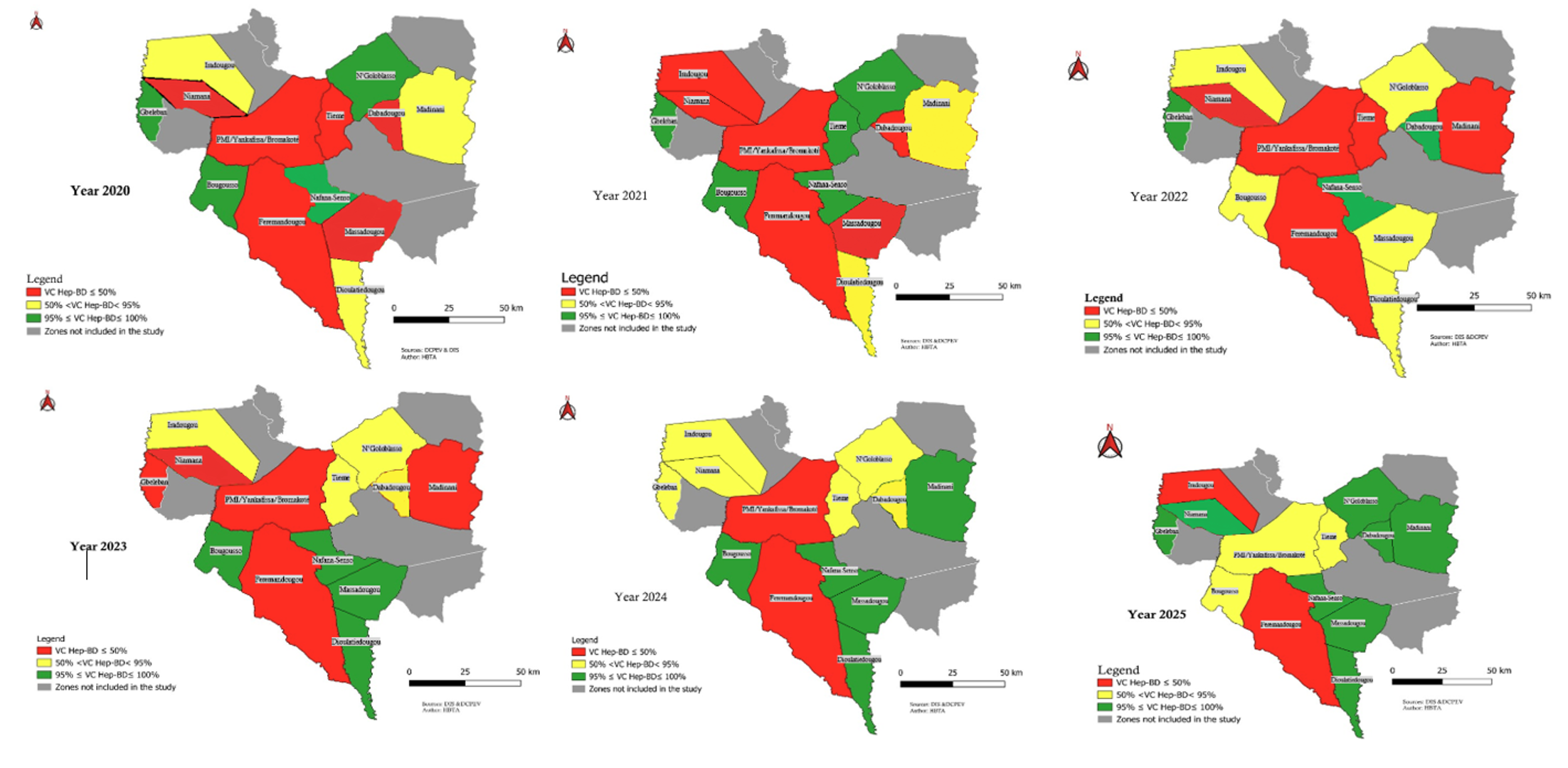
A total of 450 households were surveyed. Among the respondents, 92% were mothers, 5.6% were fathers, and 2.4% were other guardians. The estimated HepB-BD vaccination coverage (VC) in Kabadougou was 68.4% (95% CI: 64.1%-72.7%), which remained below the national target of 95% (Figure 2). Coverage varied widely between health areas: some facilities, such as the rural dispensary of Nafana Sienso, reached 100% coverage, whereas others, including the Niamana Rural Health Centre, reported 0% coverage (Figure 3).
All health facilities (100%) reported no vaccine stockouts during the study period, and each facility had a functional refrigerator. On average, five health workers per facility had received specific training in the administration of HepB-BD (SD: 4). The mean years of experience of vaccination staff was 5.2 years.” (SD: 3.8) (Table I).
Factors associated with vaccination coverage
The study identifies four groups of factors significantly associated with neonatal hepatitis B vaccination status. Socio-demographic and economic factors included the marital status of the mother or child’s guardian (p<0.001), the occupation of the mother or guardian (p< 0.001), and the household income relative to the minimum guaranteed wage (p=0.032) (Table 2). Geographical and accessibility factors were represented by the type of residence (p<0.001) (Table 2).
Knowledge and perceptions related factors included the occurrence of AEFI within the caregiver’s social circle (p=0.011) (Table 2), knowledge of hepatitis B and the HepB-BD vaccine (p<0.001), caregivers’ knowledge of AEFI (p<0.001), traditional practices (p=0.011), and local beliefs (p<0.001) (Table 3). Perinatal factors included the receipt of the mother-child health booklet (p<0.001), gestation age at delivery (p=0.021), place of delivery (p<0.001), number of antenatal care visits, and information received during antenatal consultations (p<0.001) (Table 3).
The multivariate analysis identified seven factors independently associated with non-vaccination with the HepB-BD. Home delivery was strongly associated with non-vaccination, with children born at home having nearly six times higher odds of not having received the birth dose (aOR=5.7; 95% CI:2.3-14.6; p<0.001).
Children who received the birth dose were significantly younger than those who were not vaccinated. Each one-year increase in age was associated with higher odds of non-vaccination (aOR=1.4; 95% CI: 1.2-1.7; p<0.001). Non-vaccination was significantly associated with the presence of unfavourable local beliefs (aOR =4.7; 95% CI: 2.3-9.7; p<0.001).
Receipt of the mother-child health book during antenatal care or at birth was significantly associated with lower odds of non-vaccination for the HepB-BD (aOR=0.1; 95% CI:0.04-0.5; p=0.003). Similarly, maternal awareness of the HepB-BD during antenatal care was associated with a 70% reduction in the odds of non-vaccination (aOR=0.3; 95% CI: 0.2-0.5; p<0.001).
In addition, children whose guardians were living with a partner had lower odds of non-vaccination compared to those with single guardians (aOR =0.3; 95% CI:0.1-0.7; p<0.005). Finally, the guardian’s knowledge of AEFI was associated with reduced odds of non-vaccination (aOR = 0.4; 95% CI: 0.2-0.7); p<0.001) (Table 4).
Discussion
The vaccine coverage for HepB-BD in Kabadougou showed a significant improvement and is well above the average reported in sub-Saharan Africa [9]. However, this effort is insufficient to achieve the global goal of 90% elimination of HBV [9].
Delivery in a health facility emerged as the primary determinant of HepB-BD coverage in this study. Children born in a health facility were significantly more likely to receive the hepatitis B vaccine birth dose compared with those delivered at home, consistent with findings from an earlier study [24]. Administering the HepB-BD outside health facilities remains challenging, reinforcing the need to strengthen service delivery strategies that promote institutional childbirth [8,9,12].
Once within the health system, however, the quality and distribution of human resources played a critical role. Despite an apparently adequate logistics and cold-chain system, characterised by the absence of stockouts and universally functional refrigerators, variations in staff training levels and availability were observed across health centres. Health care workers had a mean of 5.2 years of vaccination experience. In our setting, the HepB-BD vaccine is provided as a monovalent single-dose presentation, thereby eliminating concerns related to multidose vial opening and vaccine wastage. Moreover, HepB-BD is recommended immediately after every live birth in the delivery room, independent of scheduled immunisation sessions for other vaccines. However, operational challenges such as staff workload, limited availability of trained personnel during night shifts or weekends, and suboptimal coordination between maternity and immunisation services may contribute to delays beyond the recommended 24-hour period. These organisational factors may partially explain suboptimal timely HepB-BD coverage despite adequate vaccine supply and cold-chain capacity. This heterogeneity was strongly associated with coverage disparities. For example, the Niamana Rural Health Centre, which reported very few trained personnel, recorded 0% HepB-BD coverage, whereas the Nafana Sienso dispensary achieved 100%. These contrasting findings highlight critical gaps in staff deployment and retention, suggesting that the challenge extends beyond initial training alone.
These findings underscore the importance of strengthening training systems, improving staff management, and ensuring the equitable distribution of trained personnel. The shortage of qualified health workers is a well-documented barrier in the literature [25,26].
At the community level, knowledge and perception were also key determinants. The strong independent association between maternal receipt of information on hepatitis B birth dose during antenatal care and higher odds of vaccination (aOR=0.3; 95% CI: 0?2-0.5; p<0.001) highlights antenatal care as a crucial platform for delivering targeted information to improve timely HepB-BD uptake. Conversely, the persistence of unfavourable local beliefs was independently associated with a markedly higher odds of non-vaccination (aOR =4.7; 95% CI: 2.3-9.7; p<0.001), emphasizing the urgent need for culturally tailored community engagement and sensitization strategies.
Role of the mother-child health record book
Receipt of the mother-child health record book or vaccination record showed the strongest inverse association with HepB-BD non-vaccination (aOR=0.1; 95% CI:0.04-0.5; p=0.003). Receiving the health booklet indicates a successful initial contact with the health system. This contact is essential for ensuring the timely administration of the birth dose. The role of this document in ensuring vaccine traceability and continuity of immunisation service is well established in the literature, and these results are consistent with these observations. Conversely, its very low prevalence among non-vaccinated children may indicate missed opportunities for vaccination or gaps in service provision at the time of birth [9].
Implications for public health
The analysis showed that children from earlier birth cohorts, such as those born in 2020, had lower HepB-BD coverage compared with more recent cohorts (2024-2025). Age association suggests that gaps may accumulate over time in older cohorts, stressing the importance of assessing both the timely start of vaccination and missed opportunities in children who are no longer in regular contact with health services [27]. Achieving hepatitis B mother-to-child transmission MTCT elimination requires sustained HepB-BD coverage of at least 90%, in combination with systematic antenatal HBsAg screening and targeted antiviral prophylaxis for high-risk mothers [9,17,18].
Limitations
This cross-sectional study identifies associations, not causality. The reported vaccination status is subject to recall bias. Triangulation was used to mitigate this bias. The sample of healthcare professionals is small (n=15). This limits the generalizability of conclusions related to service provision. The results are specific to Kabadougou, a rural area of Côte d’Ivoire bordering Kankan in Guinea Conakry. Despite its limitations, the study relied on a robust methodology, such as data triangulation to ensure data validity and quality, and also a representative sampling method.
Conclusion
The VC of the hepatitis B vaccine at birth in Kabadougou is insufficient to achieve the goal of eliminating HBV. The study successfully identified independent factors associated with this suboptimal VC. Home births are the main logistical determinants of non-vaccination. Lack of knowledge and local beliefs are significant barriers.
To improve the VC, the provision of services around institutional delivery needs to be consolidated. It is imperative to strengthen staff training. Awareness strategies must target adverse beliefs. The systematic delivery of the vaccination booklet ensures the traceability of vaccines. Finally, the integration of prenatal HBV screening and prophylaxis is essential for the elimination of MTCT.
What is already known about the topic
- HepB-BD immunisation coverage continues to be low in highly endemic regions, particularly in sub-Saharan Africa;
- The lack of knowledge among guardians of children about hepatitis B virus is associated with non-vaccination for HepB-BD;
- The place of delivery is a factor associated with non-vaccination of HepB-BD.
What this study adds
- Traditional logistical problems were not the main obstacles in the rural area of Kabadougou (availability of vaccines, geographical accessibility of health centres, cold chain);
- The fact that the child’s guardians were single was associated with non-vaccination in HepB-BD;
- Receipt of vaccination record was a factor independently associated with non-vaccination in HepB-BD.
Acknowledgements
The authors gratefully acknowledge the institutional support provided by the African Field Epidemiology Network (AFENET), the Global Fund Project Coordination Unit (UCP-FM), the Kinshasa School of Public Health, the Africa Centres for Disease Control and Prevention (Africa CDC), and the U.S. Centres for Disease Control and Prevention (U.S. CDC). The authors also extend their sincere thanks to the Director of the National Institute of Public Hygiene (INHP), and the Director of the Coordination Directorate of the Expanded Vaccination Program (DC-PEV) for their essential guidance and support in facilitating the implementation of this study. Finally, they are deeply indebted to the health workers, community leaders, and participants of the health district of Kabadougou, without whom this research would not have been possible.
List of abbreviations
95% CI: Confidence interval at 95%
aOR: Adjusted odds ratio
cOR: Crude Odds ratioCNESVS: National Committee for Ethics in Life Sciences and Health
HBsAg: Hepatitis B surface Antigen
HBV: Hepatitis B virus
HepB-BD: birth dose of hepatitis B vaccine
KAP: Knowledge, Attitudes and Practices
AEFI: Adverse event following immunisation
MTCT:mother-to-child transmission
SPSS: Statistical Package for Social Sciences
SD: Standard Deviation
VC: vaccination coverage
WHO: World Health Organisation
Authors´ contributions
Bi Tahié Aubain HUE designed the study, supervised data collection and processing, analyzed the data, and drafted the manuscript. Aimée Lulebo MAMPASI revised the manuscript. Lepri Bernadin Nicaise AKA participated in the design of the study, supervised, critiqued, and revised the manuscript. Jean Bosco kasonga NGINDU participated in the writing of the manuscript. TIA Zolou Marie participated in the writing of the manuscript. Isaac TIEMBRE revised the manuscript. Pierre WILNIQUE participated in the writing of the manuscript. Pegnontaye Moussa SORO participated in writing the manuscript. KEDJA Innocent participated in supervising data collection and data entry for writing the manuscript. ALLO Juldas participated in supervising data collection. All authors read and approved the final manuscript.
| Table 1: Description of infant vaccination service and resources, Kabadougou, 2025 | ||
|---|---|---|
| Variables | Frequency (n=15) | Percentage (%) |
| Type of health centre | ||
| Rural | 9 | 60 |
| Urban | 6 | 40 |
| Delivery service offered | ||
| Yes | 13 | 86.7 |
| No | 2 | 13.3 |
| Offer of the HepB-BD by the health centre (Yes) | 15 | 100 |
| Shortage of vaccine stocks in health centres over the last twelve months | ||
| HepB-BD (No) | 15 | 100 |
| BCG (No) | 15 | 100 |
| VPO0 (No) | 15 | 100 |
| Functional refrigerator for vaccines available in the health centre (Yes) | 15 | 100 |
| Refrigerator breakdown during the last 12 months (No) | 15 | 100 |
| Mean year of health worker experience in vaccination ± SD* (years) | 5.2 ± 3.8 | |
| Mean number of health workers trained in HepB-BD vaccination per facility ± SD (person) | 5 ± 4 | |
| SD*: Standard Deviation; HepB-BD: Birth dose of hepatitis B vaccine; BCG: Bacillus Calmette-Guerin Vaccine; VPO0: Zero dose of oral polio vaccine | ||
| Table 2: Bivariate analysis: sociodemographic characteristics of child guardians and non-vaccination against hepatitis at birth, Kabadougou, 2025 | ||||
|---|---|---|---|---|
| Variables | HepB-BD status | cOR (95% CI) | p-value | |
| Did not receive HepB n=142 n(%) | Received HepB-BD n=308 n(%) | |||
| Guardian’s Occupation | ||||
| Income-generating activity | 78 (54.9) | 112 (36.4) | 2.1 (1.4-3.2) | <0.001 |
| No-Income-generating activity | 64 (45.1) | 196 (63.6) | ||
| Mother’s Occupation | ||||
| Income-generating activity | 72 (50.7) | 100 (32.5) | 2.2 (1.4-3.2) | <0.001 |
| No-Income-generating activity | 70 (49.3) | 208 (67.5) | ||
| Guardian’s marital status | ||||
| Married/cohabiting | 108 (76.1) | 293 (95.1) | 0.2 (0.1-0.3) | <0.001 |
| Single | 34 (23.9) | 15 (4.9) | ||
| Mother’s marital status | ||||
| Married/cohabiting | 33 (23.2) | 15 (4.9) | 5.9 (3.1-11.3) | <0.001 |
| Single | 109 (76.8) | 293 (95.1) | ||
| Type of residence | ||||
| Urban | 84 (59.2) | 126 (40.9) | 2.1 (1.4-3.1) | <0.001 |
| Rural | 58 (40.8) | 182 (59.1) | ||
| Monthly household income according to the guaranteed minimum wage | ||||
| ≤75 000 Fcfa* | 116 (81.7) | 276 (89.6) | 0.5 (0.3-0.9) | 0.021 |
| >75 000 Fcfa | 26 (18.3) | 32 (10.4) | ||
| Reported history of an AEFI in a child from the guardian’s social network | ||||
| Yes | 13 (9.2) | 57 (18.5) | 0.4 (0.2-0.8) | 0.011 |
| No | 129 (90.8) | 251 (81.5) | ||
| Median guardian age in years (Interquartile range) | 30 (12) | 27 (10) | — | 0.007 |
| Median mother age in years (Interquartile range) | 28 (11) | 26 (9) | — | 0.246 |
| Median father age in years (Interquartile range) | 39 (13) | 38 (11) | — | 0.539 |
| Median child age in years (Interquartile range) | 2.2 (3.3) | 1.4 (2.6) | — | 0.013 |
| Fcfa*: African Financial Community Franc; (1): Chi-square test; (2): Mann-Whitney U test | ||||
| Table 3: Bivariate analysis: Perinatal care and guardian awareness associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025 | ||||
|---|---|---|---|---|
| Variables | HepB-BD status | cOR (95% CI) | p-value | |
| Did not receive HepB n=142 n(%) | Received HepB-BD n=308 n(%) | |||
| Receipt of the mother-child health record book | ||||
| Yes | 112 (78.9) | 304 (98.7) | 0.05 (0.01-0.1) | <0.001 |
| No | 30 (21.1) | 4 (1.3) | ||
| Term birth (≥37 weeks gestation) | ||||
| Yes | 135 (95.1) | 305 (99) | 0.2 (0.05-0.7) | 0.021 |
| No | 7 (4.9) | 3 (1) | ||
| Place of delivery | ||||
| Home | 28 (19.7) | 10 (3.2) | 7.3 (3.4-15.6) | <0.001 |
| Health facility | 114 (80.3) | 298 (96.8) | ||
| Antenatal care visits attended by the mother | ||||
| Yes | 117 (82.4) | 304 (98.7) | 0.06 (0.02-0.2) | <0.001 |
| No | 25 (17.6) | 4 (1.3) | ||
| Mother received information on HepB-BD during antenatal care | ||||
| Yes | 50 (35.2) | 205 (66.6) | 0.4 (0.3-0.5) | <0.001 |
| No | 92 (64.8) | 103 (33.4) | ||
| Guardian’s knowledge of the existence of hepatitis B vaccine | ||||
| Yes | 47 (33.1) | 169 (54.9) | 0.4 (0.3-0.6) | <0.001 |
| No | 94 (66.9) | 139 (45.1) | ||
| Guardian’s knowledge of the HepB-BD | ||||
| Yes | 39 (27.5) | 164 (53.2) | 0.3 (0.2-0.5) | <0.001 |
| No | 103 (72.5) | 144 (46.8) | ||
| Guardian’s knowledge of the AEFI | ||||
| Yes | 44 (31) | 153 (49.7) | 0.5 (0.3-0.7) | <0.001 |
| No | 98 (69) | 155 (50.3) | ||
| Existence of traditional newborn care practices in your community | ||||
| Yes | 53 (37.3) | 79 (25.6) | 1.7 (1.1-2.6) | 0.011 |
| No | 89 (62.7) | 229 (74.4) | ||
| Existence of local beliefs concerning vaccination | ||||
| Yes | 35 (24.6) | 23 (7.5) | 4.1 (2.3-7.2) | <0.001 |
| No | 107 (75.4) | 285 (92.5) | ||
| Table 4: Binary logistic regression of factors associated with hepatitis B birth dose non-vaccination, Kabadougou, 2025 | |||
|---|---|---|---|
| Variables | Non-vaccination of HepB-BD | ||
| aOR | 95% CI | P value | |
| Child’s age at survey (years) | 1.4 | 1.2-1.7 | <0.001 |
| Guardian’s marital status | |||
| Married/Cohabiting | 0.3 | 0.1-0.7 | 0.005 |
| Single | 1 | ||
| Place of delivery | |||
| Home | 5.7 | 2.3-14.6 | <0.001 |
| Health facility | 1 | ||
| Guardian’s knowledge of the AEFI | |||
| Yes | 0.4 | 0.2-0.7 | 0.001 |
| No | 1 | ||
| Receipt of the mother-child health record book | |||
| Yes | 0.1 | 0.04-0.5 | 0.003 |
| No | 1 | ||
| Mother received information on HepB-BD during antenatal care | |||
| Yes | 0.3 | 0.2-0.5 | <0.001 |
| No | 1 | ||
| Existence of local beliefs concerning vaccination | |||
| Yes | 4.7 | 2.3-9.7 | <0.001 |
| No | 1 | ||
| aOR: Adjusted Odds Ratio | |||



References
- Bah I. Facteurs associés à la vaccination incomplète chez les enfants de 12 à 23 mois en République de Guinée : une étude des données de l’enquête démographique et de santé de 2018 [Factors associated with incomplete vaccination among children aged 12 to 23 months in the Republic of Guinea: a study of data from the 2018 Demographic and health survey] [master’s thesis on Internet]. Bruxelles (Belgique): Université Catholique de Louvain; 2022 [cited 2026 Mar 09]. 41 p. Available from: https://thesis.dial.uclouvain.be/server/api/core/bitstreams/d8207044-428f-43fe-91a3-39dae21e3d1f/content
- Pan American Health Organization World Health Organization. Vaccination of Newborns in the Context of the COVID-19 Pandemic, 19 May 2020 [Internet]. Washington (DC): Pan American Health Organization World Health Organization; 2020 [cited 2026 Mar 09]. 5 p. Available from: https://iris.paho.org/handle/10665.2/52226
- Fortmann MI, Dirks J, Goedicke-Fritz S, Liese J, Zemlin M, Morbach H, Härtel C. Immunization of preterm infants: current evidence and future strategies to individualized approaches. Semin Immunopathol [Internet]. 2022 Aug 3 [cited 2026 Mar 09];44(6):767–84. doi:10.1007/s00281-022-00957-1 Available from: https://link.springer.com/article/10.1007/s00281-022-00957-1
- Tee QW, Odisho R, Purcell E, Purcell R, Buttery J, Nold-Petry CA, Nold MF, Malhotra A. Safety of Hepatitis B Vaccines (Monovalent or as Part of Combination) in Preterm Infants: A Systematic Review. Vaccines [Internet]. 2024 Mar 1 [cited 2026 Mar 09];12(3):261. doi:10.3390/vaccines12030261 Available from: https://www.mdpi.com/2076-393X/12/3/261
- Veronese P, Dodi I, Esposito S, Indolfi G. Prevention of vertical transmission of hepatitis B virus infection. WJG [Internet]. 2021 Jul 14 [cited 2026 Mar 09];27(26):4182–93. doi:10.3748/wjg.v27.i26.4182 Available from: https://doi.org/10.3748/wjg.v27.i26.4182
- Kyuregyan KK, Kichatova VS, Isaeva OV, Potemkin IA, Malinnikova EYu, Lopatukhina MA, Karlsen AA, Asadi Mobarhan FA, Mullin EV, Slukinova OS, Ignateva ME, Sleptsova SS, Oglezneva EE, Shibrik EV, Isaguliants MG, Mikhailov MI. Coverage with Timely Administered Vaccination against Hepatitis B Virus and Its Influence on the Prevalence of HBV Infection in the Regions of Different Endemicity. Vaccines [Internet]. 2021 Jan 23 [cited 2026 Mar 09];9(2):82. doi:10.3390/vaccines9020082
- Tall H, Adam P, Tiendrebeogo ASE, Vincent JP, Schaeffer L, Von Platen C, Fernandes-Pellerin S, Sawadogo F, Bokoum A, Bouda G, Ouattara S, Ouédraogo I, Herrant M, Boucheron P, Sawadogo A, Betsem E, Essoh A, Kabore L, Ouattara A, Méda N, Hien H, Gosset A, Giles-Vernick T, Boyer S, Kania D, Vray M, Shimakawa Y. Impact of Introducing Hepatitis B Birth Dose Vaccines into the Infant Immunization Program in Burkina Faso: Study Protocol for a Stepped Wedge Cluster Randomized Trial (NéoVac Study). Vaccines [Internet]. 2021 Jun 1 [cited 2026 Mar 09];9(6):583. doi:10.3390/vaccines9060583
- Kabore HJ, Li X, Alleman MM, Manzengo CM, Mumba M, Biey J, Paluku G, Bwaka AM, Impouma B, Tohme RA. Progress Toward Hepatitis B Control and Elimination of Mother-to-Child Transmission of Hepatitis B Virus — World Health Organization African Region, 2016–2021. MMWR Morb Mortal Wkly Rep [Internet]. 2023 Jul 21 [cited 2026 Mar 09];72(29):782–7. doi:10.15585/mmwr.mm7229a2
- Njuguna HN, Ward JW, Kabore HJ, Hiebert L, Jacques-Carroll L, Khetsuriani N, Rania A, Tohme RA. Introduction of Hepatitis B Birth Dose Vaccination in Africa: A Toolkit for National Immunization Technical Advisory Groups 2022 [Internet]. Decatur (GA): Coalition for Global Hepatitis Elimination, Task Force for Global Health; 2022 [cited 2026 Mar 09]. 60 p. Available from: https://www.nitag-resource.org/sites/default/files/2022-12/HepB-BD%20NITAG%20toolkit%20final%20version_12-16-22_0.pdf
- Kouame MG. Consequences of HBV infection in HIV-positive individuals in Sub Saharan Africa during the era of early antiretroviral therapy [PhD thesis on Internet]. Bordeaux (France): University of Bordeaux; 2021 [cited 2026 Mar 09]. 124 p. Available from: https://theses.hal.science/tel-03288724/
- Anzouan-Kacou HYK, Dehinsala M, Bangoura AD, Kouamé DH, Doffou AS, Mahassadi AK, Yao FB, Attia AK. Socio-economic aspects of the management of chronic viral hepatitis in Côte-d’Ivoire. Ann Afr Med [Internet]. 2022 Sep 23 [cited 2026 Mar 09];15(4):e4770–8. doi:10.4314/aamed.v15i4.3
- Akani B, Ginsberg GM, Zengbé-Acray P, Douba A, Allah-Kouadio E. Cost-utility analysis of adding a fourth dose at birth to the hepatitis B vaccination schedule in Côte d’Ivoire. AJHE [Internet]. 2022 Jun 1 [cited 2026 Mar 09];11(1):1830. Available from: https://www.ajhe.org.in/article_html.php?did=14137&issueno=0
- Cheung KW, Lao TTH. Hepatitis B – Vertical transmission and the prevention of mother-to-child transmission. Best Practice & Research Clinical Obstetrics & Gynaecology [Internet]. 2020 Oct [cited 2026 Mar 09];68:78–88. doi:10.1016/j.bpobgyn.2020.02.014
- Ministère de la santé, de l’Hygiene Publique et de la Couverture Maladie Universelle (CIV). Monographie de la région sanitaire du Kabadougou en 2024 [Monograph DRS Kabadougou Year 2024] [Internet]. Abidjan (Cote d’Ivoire): Ministère de la santé, de l’Hygiene Publique et de la Couverture Maladie Universelle; 2024 [cited 2026 Mar 09]. 13 p.
- Organisation Mondiale de la Santé. Enquête de couverture vaccinale par sondage en grappes [Vaccination coverage survey using cluster sampling] [Internet]. Geneve (Suisse): Organisation Mondiale de la Santé; 2025 [cited 2026 Mar 09]. 312 p. Available from: https://cdn.who.int/media/docs/default-source/immunization/immunization-coverage/vaccination_coverage_cluster_survey_fr.pdf
- Riches N, Henrion MYR, MacPherson P, Hahn C, Kachala R, Mitchell T, Murray D, Mzumara W, Nkoka O, Price AJ, Riches J, Seery A, Thom N, Loarec A, Lemoine M, Ndow G, Shimakawa Y, Thompson P, Morgan C, Desai S, Easterbrook P, Stockdale AJ. Vertical transmission of hepatitis B virus in the WHO African region: a systematic review and meta-analysis. The Lancet Global Health [Internet]. 2025 Mar [cited 2026 Mar 09];13(3):e447–58. doi:10.1016/S2214-109X(24)00506-0
- Breakwell L, Tevi-Benissan C, Childs L, Mihigo R, Tohme R. The status of hepatitis B control in the African region. Pan Afr Med J [Internet]. 2017 Jun 22 [cited 2026 Mar 09];27. doi:10.11604/pamj.supp.2017.27.3.11981
- Shimakawa Y, Veillon P, Birguel J, Pivert A, Sauvage V, Guillou-Guillemette HL, Roger S, Njouom R, Ducancelle A, Amta P, Huraux JM, Adoukara JP, Lunel-Fabiani F. Residual risk of mother-to-child transmission of hepatitis B virus infection despite timely birth-dose vaccination in Cameroon (ANRS 12303): a single-centre, longitudinal observational study. The Lancet Global Health [Internet]. 2022 Apr 17 [cited 2026 Mar 09];10(4):e521–9. doi:10.1016/S2214-109X(22)00026-2
- Sanogo F. Connaissances, attitudes et pratiques des mères face aux signes de maladie chez l’enfant après la vaccination de routine à Tominian [Knowledge, attitudes and practical of mothers towards signs of illness in children after routine vaccination in Tominian]. Mali Sante Publique [Internet]. 2025 Jan 21 [cited 2026 Mar 09];79–83. doi:10.53318/msp.v13i2.2970
- Aslan A, Şaşmazer B, Ayar Y, Duran ZC, Akova M. Barriers Against Hepatitis B Vaccination in High-Risk Adults: A Cross-Sectional Study. Turkish Journal of Gastroenterology [Internet]. 2022 May 15 [cited 2026 Mar 09];33(5):427–33. doi:10.5152/tjg.2022.21257
- Vo LT, Phan DQ, Tran HG, Nguyen LTP, Gyan A, Nguyen HTN, Huynh G. Hepatitis B vaccine coverage in health care students: a cross-sectional study in Vietnam. Elshaarawy. PLoS ONE [Internet]. 2025 Mar 31 [cited 2026 Mar 09];20(3):e0320860. doi:10.1371/journal.pone.0320860
- Scherkoske M, Moran J, Barnett M, Lewin C, Campbell S, Novy P. Insights into hepatitis B vaccination decision-making: Exploring vaccine preferences, health disparities, and practice patterns among patients and healthcare providers. Hum Vaccin Immunother [Internet]. 2025 Dec 31 [cited 2026 Mar 09];21(1):2555698. doi:10.1080/21645515.2025.2555698
- Aka N, Georges BK. Le carnet de santé mère-enfant et le partogramme, outils d’information et de communication sanitaire sur l’issue de la grossesse et de l’accouchement [The mother-child health record and the partogram: health information and communication tools regarding pregnancy and childbirth outcomes]. RISLC [Internet]. 2019 Dec [cited 2026 Mar 09];3:107-126. Available from: https://www.revue-slc.org/wp-content/uploads/2023/11/6_NIAMKEY-Aka-BONI-Kouadio-Georges_RISLC-n%C2%B013-Decembre-2019.pdf
- Bassoum O, Kimura M, Tal Dia A, Lemoine M, Shimakawa Y. Coverage and Timeliness of Birth Dose Vaccination in Sub-Saharan Africa: A Systematic Review and Meta-Analysis. Vaccines [Internet]. 2020 Jun 11 [cited 2026 Mar 09];8(2):301. doi:10.3390/vaccines8020301
- Zhao H, Zhou X, Zhou YH. Hepatitis B vaccine development and implementation. Hum Vaccin Immunother [Internet]. 2020 Jul 2 [cited 2026 Mar 09];16(7):1533–44. doi:10.1080/21645515.2020.1732166
- Hussein NA, Ismail AM, Jama SS. Assessment of Hepatitis B Vaccination Status and Associated Factors among Healthcare Workers in Bosaso, Puntland, Somalia 2020. Biomed Res Int [Internet]. 2022 Jan 21 [cited 2026 Mar 09];2022(1):9074294. doi:10.1155/2022/9074294
- Deschamps C, Bardon T, Blaise T, Bonifay T, Boutrou M, Fremery A, Henry K, Lambert Y, Le Turnier P, Mutricy R, Oberlis M, Quintin B, Sauvage B, Thomas E, Epelboin L, Hureau-Mutricy L. 6e journées des travaux scientifiques des soignants de Guyane. Nos soignants ont du talent ! 25 & 26 mai 2023, Cayenne, Guyane [6th day dedicated to the scientific works of caregivers in French Guiana. Our caregivers have talent! May 25 & 26, 2023, Cayenne, French Guiana]. Med Trop Sante Int [Internet]. 2023 Oct 31 [cited 2026 Mar 09];3(4):mtsi.v3i4.2023.439. doi:10.48327/mtsi.v3i4.2023.439 Available from: https://revuemtsi.societe-mtsi.fr/index.php/bspe-articles/article/view/439
