Research | Open Access | Volume 9 (1): Article 44 | Published: 11 Mar 2026
Prevalence of urinary schistosomiasis in school-aged children, knowledge, attitude and practice of household heads regarding hematuria, Haho Health District, Togo
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Variable | Proportion (%) | 95% CI |
|---|---|---|
| Children age (years) | ||
| 5–9 | 30.0 (162/540) | 26.3 – 34.0 |
| 10–14 | 34.0 (146/429) | 29.8 – 38.6 |
| Children sex | ||
| Male | 32.8 (210/641) | 29.2 – 36.5 |
| Female | 29.9 (98/328) | 25.2 – 35.0 |
| Place of residence of heads of households | ||
| Rural | 30.9 (231/747) | 27.7 – 34.3 |
| Urban | 34.7 (77/222) | 28.4 – 41.3 |
| Education level of the heads of households | ||
| Never been in school | 37.1 (163/440) | 32.7 – 41.7 |
| Primary school | 31.1 (97/312) | 26.2 – 36.4 |
| Secondary school | 22.4 (47/210) | 16.9 – 28.6 |
| University | 14.3 (1/6) | 0.4 – 57.9 |
Table 1: Prevalence of urinary schistosomiasis by school aged children age groups, sex and by heads of household place of residence and education level, Wahala health area, Togo, March 2020.
| Names of hematuria in local languages | Number | Percentage | 95% CI |
|---|---|---|---|
| Community of Kabyè-Lamba (Respondents: 165/221) | |||
| Eyawou | 128 | 77.6 | 70.4 – 83.7 |
| Himah | 18 | 10.9 | 6.6 – 16.7 |
| Him Ni Tchalim | 17 | 10.3 | 6.1 – 16.0 |
| Kpètrè | 1 | 0.6 | 0.02 – 3.3 |
| Ouma | 1 | 0.6 | 0.02 – 3.3 |
| Community of Naouda (Respondents: 75/86) | |||
| Hawem djim | 39 | 52.0 | 40.2 – 63.7 |
| Ahoran djim | 36 | 48.0 | 36.3 – 59.9 |
| Community of Adja-Ewé-Fon (Respondents: 51/84) | |||
| Ehoudoudo | 39 | 76.5 | 62.5 – 87.2 |
| Adoudove | 12 | 23.5 | 12.8 – 37.5 |
| Community of Kotocoli (Respondents: 8/18) | |||
| Kpédéyou | 4 | 50.0 | 15.7 – 84.3 |
| Tchesso | 3 | 37.5 | 8.5 – 75.5 |
| Fim nazima | 1 | 12.5 | 0.3 – 52.7 |
| Community of Moba (Respondents: 6/7) | |||
| Fida | 1 | 16.7 | 0.4 – 64.1 |
| Hogna ninesoum | 1 | 16.7 | 0.4 – 64.1 |
| Ngnani Nassou | 1 | 16.7 | 0.4 – 64.1 |
| Oninyiam nissan | 1 | 16.7 | 0.4 – 64.1 |
| Ogmoriouso | 1 | 16.7 | 0.4 – 64.1 |
| Song | 1 | 16.7 | 0.4 – 64.1 |
| Community of Ifè (Respondents: 3/3) | |||
| Nto Owoun Onbarz | 2 | 66.7 | 9.43 – 99.16 |
| Toembra | 1 | 33.3 | 0.84 – 90.57 |
| Community of Ana (Respondents: 2/2) | |||
| Etraba | 2 | 100 | 15.8 – 100.0 |
Table 2: Distribution of the names of hematuria in community language groups, Wahala Health area, Haho Health District, Togo, March 2020.
| Variables | Proportion | 95% CI |
|---|---|---|
| Knowledge on hematuria in school aged children by gender of the head of household | ||
| Hematuria is sign of a disease | ||
| Men | 99.6 (274/275) | 97.99 – 99.99 |
| Women | 99.3 (140/141) | 96.1 – 99.98 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Knew the name of hematuria in the local language | ||
| Men | 80.2 (227/283) | 75.1 – 84.7 |
| Women | 57.3 (82/143) | 48.8 – 65.6 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Men | 48.7 (130/267) | 42.6 – 54.9 |
| Women | 33.3 (46/138) | 25.5 – 41.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Men | 73.7 (207/281) | 68.1 – 78.7 |
| Women | 73.2 (104/142) | 65.2 – 80.8 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in them exposes one to risk of hematuria | ||
| Men | 98.9 (278/281) | 96.9 – 99.8 |
| Women | 95.7 (134/140) | 90.9 – 98.4 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any means of control of hematuria | ||
| Men | 4.2 (12/283) | 2.2 – 7.3 |
| Women | 1.4 (2/143) | 0.2 – 5.0 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Men | 38.9 (110/283) | 33.2 – 44.8 |
| Women | 42.7 (82/143) | 34.4 – 51.2 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Men | 44.2 (125/283) | 38.3 – 50.2 |
| Women | 32.2 (46/143) | 26.6 – 40.5 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children by age groups of the heads of household | ||
| Hematuria is a sign of a disease | ||
| Age (years) | ||
| 20- <30 | 97.6 (41/42) | 87.4 – 99.9 |
| 30-<45 | 100.0 (193/193) | 98.1 – 100.0 |
| 45-<60 | 100.0 (121/121) | 97.0 – 100.0 |
| 60-90 | 98.4 (60/61) | 91.2 – 99.9 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of hematuria in the local language | ||
| Age (years) | ||
| 20- <30 | 66.7 (28/42) | 50.5 – 80.4 |
| 30-<45 | 74.8 (148/198) | 68.1 – 80.6 |
| 45-<60 | 72.8 (91/125) | 64.1 – 80.9 |
| 60-90 | 69.4 (43/62) | 56.4 – 80.4 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Age (years) | ||
| 20- <30 | 40.50 (19/40) | 31.5 – 63.9 |
| 30-<45 | 44.68 (84/188) | 37.4 – 52.1 |
| 45-<60 | 43.33 (52/120) | 34.3 – 52.7 |
| 60-90 | 37.93 (22/58) | 25.5 – 51.6 |
| Overall | 43.60 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Age (years) | ||
| 20- <30 | 74.4 (30/42) | 55.4 – 84.3 |
| 30-<45 | 72.5 (142/196) | 65.6 – 78.6 |
| 45-<60 | 75.8 (94/124) | 67.3 – 83.0 |
| 60-90 | 74.2 (46/62) | 61.5 – 84.5 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in it exposes one to risk of hematuria | ||
| Age (years) | ||
| 20- <30 | 100.00 (41/41) | 91.4 – 100.0 |
| 30-<45 | 97.96 (192/196) | 94.9 – 99.4 |
| 45-<60 | 96.77 (120/124) | 91.9 – 99.1 |
| 60-90 | 98.36 (60/61) | 91.2 – 99.9 |
| Overall | 97.87 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Age (years) | ||
| 20- <30 | 4.8 (2/42) | 0.58 – 16.2 |
| 30-<45 | 0.5 (1/198) | 0.01 – 2.8 |
| 45-<60 | 7.2 (9/125) | 3.35 – 13.2 |
| 60-90 | 3.2 (2/62) | 0.39 – 11.2 |
| Overall | 3.3 (14/427) | 1.96 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Age (years) | ||
| 20- <30 | 42.9 (18/42) | 27.7 – 59.0 |
| 30-<45 | 37.4 (74/198) | 30.6 – 44.5 |
| 45-<60 | 39.2 (49/125) | 30.6 – 48.3 |
| 60-90 | 50.0 (31/62) | 37.0 – 62.9 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Age (years) | ||
| 20- <30 | 26.2 (11/42) | 13.86 – 42.0 |
| 30-<45 | 41.9 (83/198) | 34.9 – 49.1 |
| 45-<60 | 44.8 (56/125) | 35.9 – 53.9 |
| 60-90 | 33.9 (21/62) | 22.3 – 47.0 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the education level of the heads of household | ||
| Hematuria is a disease | ||
| Never been at school | 99.5 (191/192) | 97.1 – 99.9 |
| Primary school | 100.0 (125/125) | 97.1 – 100.0 |
| Secondary school | 99.0 (95/96) | 94.3 – 99.9 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Never been at school | 70.4 (138/196) | 63.5 – 76.7 |
| Primary school | 72.7 (93/128) | 64.1 – 80.2 |
| Secondary school | 77.8 (77/99) | 68.3 – 85.5 |
| University | 50.0 (2/4) | 6.8 – 93.2 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Never been at school | 42.6 (80/188) | 35.4 – 49.9 |
| Primary school | 37.5 (45/120) | 28.8 – 46.8 |
| Secondary school | 52.1 (49/94) | 41.6 – 62.5 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Never been at school | 76.4 (149/195) | 69.8 – 82.2 |
| Primary school | 67.7 (86/127) | 58.9 – 75.7 |
| Secondary school | 75.5 (74/98) | 65.8 – 83.6 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Never been at school | 97.4 (188/193) | 94.1 – 99.2 |
| Primary school | 96.8 (122/126) | 92.1 – 99.1 |
| Secondary school | 100.0 (99/99) | 96.3 – 100.0 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Never been at school | 2.6 (5/196) | 0.8 – 5.9 |
| Primary school | 4.7 (6/128) | 1.7 – 9.9 |
| Secondary school | 3.0 (3/99) | 0.6 – 8.6 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Never been at school | 40.3 (79/196) | 33.4 – 47.5 |
| Primary school | 42.2 (54/128) | 33.5 – 51.2 |
| Secondary school | 38.4 (38/99) | 28.8 – 48.7 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Never been at school | 36.7 (72/196) | 56.1 – 70.0 |
| Primary school | 36.7 (47/128) | 28.4 – 45.7 |
| Secondary school | 50.5 (50/99) | 40.3 – 60.7 |
| University | 50.0 (2/4) | 6.76 – 93.2 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the profession of the heads of household | ||
| Hematuria is a disease | ||
| Artisan | 100.0 (30/30) | 88.4 – 100.0 |
| Farmer | 99.6 (266/267) | 97.9 – 99.9 |
| Housewife | 100.0 (51/51) | 93.0 – 100.0 |
| Seller | 97.7 (42/43) | 87.7 – 99.9 |
| Teacher | 100.0 (9/9) | 66.4 – 100.0 |
| Other | 100.0 (17/17) | 80.5 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Artisan | 70.0 (21/30) | 50.6 – 86.3 |
| Farmer | 78.8 (215/273) | 73.4 – 83.5 |
| Housewife | 71.2 (37/52) | 56.9 – 82.9 |
| Seller | 43.2 (19/44) | 28.4 – 58.9 |
| Teacher | 54.6 (6/11) | 23.4 – 83.3 |
| Other | 70.6 (12/17) | 44.0 – 89.7 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Artisan | 53.6 (15/28) | 33.9 – 72.5 |
| Farmer | 40.4 (105/260) | 34.4 – 46.6 |
| Housewife | 50.0 (25/50) | 35.5 – 64.5 |
| Seller | 38.1 (16/42) | 23.6 – 54.4 |
| Teacher | 63.6 (7/11) | 30.8 – 89.1 |
| Other | 60.0 (9/15) | 12.3 – 83.7 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Artisan | 66.7 (20/30) | 47.2 – 82.7 |
| Farmer | 71.1 (192/270) | 65.3 – 76.4 |
| Housewife | 75.0 (39/52) | 61.1 – 85.9 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 72.7 (8/11) | 39.0 – 93.9 |
| Other | 82.4 (14/17) | 52.6 – 96.2 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Artisan | 90.0 (27/30) | 73.5 – 98.0 |
| Farmer | 99.3 (268/270) | 97.4 – 99.9 |
| Housewife | 94.1 (48/51) | 83.8 – 98.8 |
| Seller | 97.7 (43/44) | 88.0 – 99.9 |
| Teacher | 100.0 (11/11) | 71.5 – 100.0 |
| Other | 100.0 (16/16) | 79.4 – 100.0 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any means of control of hematuria | ||
| Artisan | 6.7 (2/30) | 0.8 – 22.1 |
| Farmer | 3.3 (9/273) | 1.5 – 6.2 |
| Housewife | 3.9 (2/52) | 0.5 – 13.2 |
| Seller | 0.0 (0/44) | 0.0 – 8.0 |
| Teacher | 0.0 (0/11) | 0.0 – 28.5 |
| Other | 5.9 (1/17) | 0.2 – 28.7 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Artisan | 26.7 (8/30) | 12.3 – 45.9 |
| Farmer | 42.1 (115/273) | 36.2 – 48.2 |
| Housewife | 28.9 (15/52) | 17.1 – 43.1 |
| Seller | 47.7 (21/44) | 32.5 – 63.3 |
| Teacher | 81.8 (9/11) | 48.2 – 97.7 |
| Other | 23.5 (4/17) | 48.2 – 97.7 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Artisan | 60.0 (18/30) | 22.7 – 59.4 |
| Farmer | 37.0 (101/273) | 31.3 – 43.0 |
| Housewife | 36.5 (19/52) | 23.6 – 51.0 |
| Seller | 45.5 (20/44) | 30.4 – 61.2 |
| Teacher | 45.5 (5/11) | 16.8 – 76.6 |
| Other | 47.1 (8/17) | 22.9 – 72.2 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to residence zone of the heads of household | ||
| Hematuria is a disease | ||
| Rural | 99.4 (328/330) | 97.8 – 99.8 |
| Semi urban | 100.0 (87/87) | 95.9 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Rural | 79.3 (268/338) | 74.7 – 83.3 |
| Semi urban | 47.2 (42/89) | 36.5 – 58.1 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Rural | 42.6 (139/326) | 37.4 – 48.1 |
| Semi urban | 47.5 (38/80) | 36.2 – 58.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Rural | 71.6 (240/335) | 66.6 – 76.2 |
| Semi urban | 80.9 (72/89) | 71.2 – 88.5 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in is at risk of hematuria | ||
| Rural | 97.6 (325/333) | 95.3 – 98.8 |
| Semi urban | 98.9 (88/89) | 93.9 – 99.9 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Rural | 3.6 (12/338) | 2.0 – 6.1 |
| Semi urban | 2.3 (2/89) | 0.3 – 7.9 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Rural | 37.3 (126/338) | 32.3 – 42.6 |
| Semi urban | 51.7 (46/89) | 40.8 – 62.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Rural | 40.8 (138/338) | 35.7 – 46.1 |
| Semi urban | 37.1 (33/89) | 27.1 – 47.9 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the religion of the heads of household | ||
| Hematuria is a disease | ||
| Christian | 98.9 (192/194) | 96.3 – 99.9 |
| Muslim | 100.0 (33/33) | 89.4 – 100.0 |
| Traditional (Animism) | 100.0 (190/190) | 98.1 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of hematuria in the local language | ||
| Christian | 71.9 (143/199) | 65.1 – 78.0 |
| Muslim | 54.3 (19/35) | 36.7 – 71.2 |
| Traditional (Animism) | 76.7 (148/193) | 70.1 – 82.5 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Christian | 41.9 (81/193) | 34.9 – 49.3 |
| Muslim | 55.2 (16/29) | 35.7 – 73.6 |
| Traditional (Animism) | 43.5 (80/184) | 36.2 – 50.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Christian | 71.2 (141/198) | 64.4 – 77.4 |
| Muslim | 97.1 (34/35) | 85.1 – 99.9 |
| Traditional (Animism) | 71.7 (137/191) | 64.8 – 78.0 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Christian | 97.9 (193/197) | 94.9 – 99.4 |
| Muslim | 94.1 (32/34) | 80.3 – 99.3 |
| Traditional (Animism) | 98.4 (188/191) | 95.5 – 99.7 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Christian | 2.5 (5/199) | 0.8 – 5.8 |
| Muslim | 0.0 (0/35) | 0.0 – 10.0 |
| Traditional (Animism) | 4.7 (9/193) | 2.2 – 8.7 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Christian | 45.7 (91/199) | 38.7 – 52.9 |
| Muslim | 37.1 (13/35) | 21.5 – 55.1 |
| Traditional (Animism) | 35.2 (68/193) | 28.5 – 42.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Christian | 42.2 (84/199) | 35.3 – 49.4 |
| Muslim | 37.1 (13/35) | 21.4 – 55.1 |
| Traditional (Animism) | 38.3 (74/193) | 31.5 – 45.6 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
Table 3: Knowledge of the heads of household on hematuria in school aged children by sociodemographic characteristics, Wahala Health area, Haho Health District, Togo, March 2020.
| Variables | Proportion | 95% CI |
|---|---|---|
| Attitudes on hematuria in school aged children according to the gender of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Men | 92.9 (263/283) | 89.3 – 95.6 |
| Women | 93.7 (134/143) | 88.4 – 97.1 |
| Overall | 93.2 (415/417) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Men | 65.7 (186/283) | 59.9 – 71.2 |
| Women | 81.1 (116/143) | 73.7 – 87.2 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Men | 14.1 (40/283) | 10.3 – 18.8 |
| Women | 7.0 (10/143) | 3.4 – 12.5 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Men | 39.0 (110/282) | 33.3 – 44.9 |
| Women | 43.4 (62/143) | 35.1 – 51.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Men | 14.9 (42/282) | 11.0 – 19.6 |
| Women | 10.5 (15/143) | 6.0 – 16.7 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Men | 10.3 (29/282) | 7.0 – 14.4 |
| Women | 13.3 (19/143) | 8.2 – 20.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the age groups of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Age (years) | ||
| 20- <30 | 92.9 (39/42) | 80.5 – 98.5 |
| 30-<45 | 92.9 (184/198) | 88.4 – 96.1 |
| 45-<60 | 93.6 (117/125) | 87.8 – 97.2 |
| 60-90 | 93.6 (58/62) | 80.3 – 98.2 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Age (years) | ||
| 20- <30 | 73.8 (31/42) | 51.0 – 86.1 |
| 30-<45 | 71.2 (141/198) | 64.4 – 77.4 |
| 45-<60 | 68.8 (86/125) | 59.9 – 76.8 |
| 60-90 | 72.6 (45/62) | 59.8 – 83.2 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Age (years) | ||
| 20- <30 | 11.9 (5/42) | 4.0 – 25.6 |
| 30-<45 | 11.6 (23/198) | 7.5 – 16.9 |
| 45-<60 | 11.2 (14/125) | 6.2 – 18.1 |
| 60-90 | 12.9 (8/62) | 5.7 – 23.9 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Age (years) | ||
| 20- <30 | 40.5 (17/42) | 25.6 – 56.7 |
| 30-<45 | 44.4 (88/198) | 37.4 – 51.7 |
| 45-<60 | 29.8 (37/124) | 22.0 – 38.7 |
| 60-90 | 48.4 (30/62) | 35.5 – 61.4 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Age (years) | ||
| 20- <30 | 7.1 (3/42) | 1.5 – 19.5 |
| 30-<45 | 13.1 (26/198) | 8.8 – 18.7 |
| 45-<60 | 16.9 (21/124) | 10.8 – 24.7 |
| 60-90 | 12.9 (8/62) | 5.7 – 23.9 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Age (years) | ||
| 20- <30 | 16.7 (7/42) | 7.0 – 31.4 |
| 30-<45 | 7.6 (15/198) | 4.3 – 12.2 |
| 45-<60 | 16.9 (21/124) | 10.8 – 24.7 |
| 60-90 | 8.1 (5/62) | 2.7 – 17.8 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the education level of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Never been at school | 93.4 (183/196) | 88.9 – 96.4 |
| Primary school | 92.2 (118/128) | 86.1 – 96.2 |
| Secondary school | 93.9 (93/99) | 87.3 – 97.7 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Never been at school | 66.3 (130/196) | 59.3 – 72.9 |
| Primary school | 78.9 (101/128) | 70.8 – 85.6 |
| Secondary school | 69.7 (69/99) | 59.7 – 78.5 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Never been at school | 44.9 (88/196) | 37.8 – 52.2 |
| Primary school | 43.0 (55/128) | 34.3 – 52.0 |
| Secondary school | 27.6 (27/98) | 19.0 – 37.5 |
| University | 50.0 (2/4) | 6.8 – 93.2 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Never been at school | 13.3 (26/196) | 8.9 – 18.8 |
| Primary school | 12.5 (16/128) | 7.3 – 19.5 |
| Secondary school | 16.3 (16/98) | 9.6 – 25.2 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Never been at school | 11.7 (23/196) | 7.6 – 17.1 |
| Primary school | 10.9 (14/128) | 6.1 – 17.7 |
| Secondary school | 11.2 (11/98) | 5.7 – 19.2 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children by profession of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Artisan | 83.3 (25/30) | 65.3 – 94.4 |
| Farmer | 94.5 (258/273) | 91.1 – 96.9 |
| Housewife | 94.2 (49/52) | 84.1 – 98.8 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 100.0 (11/11) | 71.5 – 100.0 |
| Other | 94.1 (16/17) | 71.3 – 99.9 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Artisan | 73.3 (22/30) | 54.1 – 87.7 |
| Farmer | 67.0 (183/273) | 61.1 – 72.6 |
| Housewife | 73.1 (38/52) | 59.0 – 84.4 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 81.8 (9/11) | 48.2 – 97.7 |
| Other | 70.6 (12/17) | 44.0 – 89.7 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Artisan | 3.3 (1/30) | 0.1 – 17.2 |
| Farmer | 14.3 (39/273) | 10.4 – 19.0 |
| Housewife | 13.5 (7/52) | 5.6 – 25.8 |
| Seller | 0.0 (0/44) | 0.0 – 8.0 |
| Teacher | 9.1 (1/11) | 0.2 – 41.3 |
| Other | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Artisan | 46.7 (14/30) | 28.3 – 65.7 |
| Farmer | 41.0 (112/273) | 35.1 – 47.1 |
| Housewife | 36.5 (19/52) | 23.6 – 51.0 |
| Seller | 45.5 (20/44) | 30.4 – 61.2 |
| Teacher | 50.0 (5/10) | 18.7 – 81.3 |
| Other | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Artisan | 16.7 (5/30) | 5.6 – 34.7 |
| Farmer | 11.0 (30/273) | 7.5 – 15.3 |
| Housewife | 17.3 (9/52) | 8.2 – 30.3 |
| Seller | 13.6 (6/44) | 5.2 – 27.4 |
| Teacher | 30.0 (3/10) | 6.7 – 65.3 |
| Other | 29.4 (5/17) | 10.3 – 56.0 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Artisan | 13.3 (4/30) | 3.8 – 30.7 |
| Farmer | 8.4 (23/273) | 5.4 – 12.4 |
| Housewife | 21.2 (11/52) | 11.1 – 34.7 |
| Seller | 13.6 (6/44) | 5.2 – 27.4 |
| Teacher | 10.0 (1/10) | 0.3 – 44.5 |
| Other | 17.7 (3/17) | 3.8 – 43.4 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children by the residence zone of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Rural | 97.3 (329/338) | 95.0 – 98.6 |
| Semi urban | 77.5 (69/89) | 67.5 – 85.7 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Rural | 65.1 (220/338) | 59.9 – 70.0 |
| Semi urban | 92.3 (83/89) | 85.9 – 97.5 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Rural | 14.2 (48/338) | 10.9 – 18.3 |
| Semi urban | 2.3 (2/89) | 0.3 – 7.9 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Rural | 39.2 (132/337) | 34.1 – 44.5 |
| Semi urban | 44.9 (40/89) | 34.4 – 55.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Rural | 13.1 (44/337) | 9.9 – 17.1 |
| Semi urban | 15.7 (14/89) | 8.9 – 25.0 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Rural | 11.0 (37/337) | 8.1 – 14.8 |
| Semi urban | 12.4 (11/89) | 6.3 – 21.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the religion of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Christian | 93.5 (186/199) | 89.1 – 96.5 |
| Muslim | 85.7 (30/35) | 69.7 – 95.2 |
| Traditional (Animism) | 94.3 (182/193) | 90.0 – 97.1 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Christian | 72.4 (144/199) | 65.6 – 78.5 |
| Muslim | 77.1 (27/35) | 59.9 – 89.6 |
| Traditional (Animism) | 68.4 (132/193) | 61.3 – 74.9 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Christian | 14.1 (28/199) | 9.7 – 19.7 |
| Muslim | 2.9 (1/35) | 0.1 – 14.9 |
| Traditional (Animism) | 10.9 (21/193) | 6.9 – 16.2 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Christian | 39.7 (79/199) | 32.9 – 46.9 |
| Muslim | 48.6 (17/35) | 31.4 – 66.0 |
| Traditional (Animism) | 39.6 (76/192) | 32.6 – 46.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Christian | 14.1 (28/199) | 9.6 – 19.7 |
| Muslim | 22.9 (8/35) | 10.4 – 40.1 |
| Traditional (Animism) | 11.5 (22/192) | 7.3 – 16.8 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Christian | 9.6 (19/199) | 5.9 – 14.5 |
| Muslim | 14.3 (5/35) | 4.8 – 30.3 |
| Traditional (Animism) | 12.5 (24/192) | 8.2 – 18.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
Table 4: Attitudes of the heads of household on hematuria in school aged children by sociodemographic characteristics, Wahala Health area, Haho Health District, Togo, March 2020.
| Variables | Proportion | 95% CI |
|---|---|---|
| Practices on hematuria in school aged children according to the gender of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Men | 31.6 (18/57) | 19.9 – 45.2 |
| Women | 46.7 (14/30) | 28.3 – 65.7 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Men | 24.6 (14/57) | 14.1 – 37.8 |
| Women | 6.7 (2/30) | 0.8 – 22.1 |
| Overall | 18.2 (16/88) | 10.7 – 27.8 |
| Did nothing when a child got hematuria | ||
| Men | 19.3 (11/57) | 10.1 – 31.9 |
| Women | 23.3 (7/30) | 9.9 – 42.3 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Self-medicated the child when he got hematuria | ||
| Men | 15.8 (9/57) | 7.5 – 27.9 |
| Women | 20.0 (6/30) | 7.7 – 38.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river or pond | ||
| Men | 48.5 (37/130) | 39.6 – 57.4 |
| Women | 45.8 (33/72) | 34.0 – 58.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished the children when they were seen swimming in rivers or ponds | ||
| Men | 19.2 (25/130) | 12.9 – 27.1 |
| Women | 19.4 (14/72) | 11.1 – 30.5 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Practices on hematuria in school aged children by age groups of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 20.0 (1/5) | 0.5 – 71.6 |
| 30-<45 | 36.6 (15/41) | 22.1 – 53.1 |
| 45-<60 | 25.9 (7/27) | 11.1 – 46.3 |
| 60-90 | 60.0 (9/15) | 32.3 – 83.7 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 40.0 (2/5) | 5.3 – 85.3 |
| 30-<45 | 19.5 (8/41) | 8.8 – 34.9 |
| 45-<60 | 14.8 (4/27) | 4.2 – 33.7 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 0.0 (0/5) | 0.0 – 52.2 |
| 30-<45 | 14.6 (6/41) | 5.6 – 29.2 |
| 45-<60 | 29.6 (8/27) | 13.8 – 50.2 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river or pond | ||
| Age (years) | ||
| 20- <30 | 45.0 (9/20) | 23.1 – 68.5 |
| 30-<45 | 44.7 (38/85) | 33.9 – 55.9 |
| 45-<60 | 52.2 (36/69) | 39.8 – 64.4 |
| 60-90 | 44.8 (13/29) | 26.5 – 64.3 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Age (years) | ||
| 20- <30 | 15.0 (3/20) | 3.2 – 37.9 |
| 30-<45 | 17.7 (15/85) | 10.2 – 27.4 |
| 45-<60 | 14.5 (10/69) | 7.2 – 25.0 |
| 60-90 | 37.9 (11/29) | 20.7 – 57.7 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Age (years) | ||
| 20- <30 | 40.0 (2/5) | 5.3 – 85.3 |
| 30-<45 | 22.0 (9/41) | 10.6 – 37.6 |
| 45-<60 | 18.5 (5/27) | 6.3 – 38.1 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the education level of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Never been at school | 41.6 (22/53) | 28.1 – 55.9 |
| Primary school | 29.6 (8/27) | 13.8 – 50.2 |
| Secondary school | 28.6 (2/7) | 3.7 – 71.0 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Never been at school | 15.1 (8/53) | 6.8 – 27.6 |
| Primary school | 11.1 (3/27) | 2.4 – 29.2 |
| Secondary school | 57.1 (4/7) | 18.4 – 90.1 |
| University | 100.0 (1/1) | 2.5 – 100.0 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Never been at school | 15.1 (8/53) | 6.8 – 27.6 |
| Primary school | 29.6 (8/27) | 15.8 – 50.2 |
| Secondary school | 0.0 (0/7) | 0.0 – 41.0 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Never been at school | 50.0 (48/96) | 39.6 – 60.4 |
| Primary school | 45.3 (29/64) | 32.8 – 58.3 |
| Secondary school | 42.9 (18/42) | 27.7 – 59.0 |
| University | 100.0 (1/1) | 2.5 – 100.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Never been at school | 21.9 (21/96) | 14.1 – 31.4 |
| Primary school | 17.2 (11/64) | 8.9 – 28.7 |
| Secondary school | 16.7 (7/42) | 7.0 – 31.4 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Never been at school | 17.0 (5/53) | 8.07 – 29.8 |
| Primary school | 29.6 (8/27) | 13.75 – 50.2 |
| Secondary school | 14.3 (1/7) | 0.36 – 57.9 |
| University | 0.0 (0/1) | 0.00 – 97.5 |
| Overall | 20.5 (18/88) | 12.60 – 30.4 |
| Practices on hematuria in school aged children according to the profession of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 39.3 (22/56) | 26.5 – 53.3 |
| Housewife | 33.3 (3/9) | 7.5 – 70.0 |
| Seller | 41.7 (5/12) | 15.2 – 72.3 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 21.4 (12/56) | 11.6 – 34.4 |
| Housewife | 0.0 (0/9) | 0.0 – 33.6 |
| Seller | 16.7 (2/12) | 2.1 – 48.4 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Artisan | 57.1 (4/7) | 18.4 – 90.1 |
| Farmer | 10.7 (6/56) | 4.0 – 21.9 |
| Housewife | 11.1 (1/9) | 0.3 – 48.3 |
| Seller | 33.3 (4/12) | 9.9 – 65.1 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Artisan | 28.9 (4/14) | 8.4 – 58.1 |
| Farmer | 50.4 (69/137) | 41.7 – 59.0 |
| Housewife | 50.0 (13/26) | 29.9 – 70.1 |
| Seller | 31.6 (6/19) | 12.6 – 56.6 |
| Teacher | 50.0 (1/2) | 1.3 – 98.7 |
| Other | 60.0 (3/5) | 14.7 – 94.7 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Artisan | 7.1 (1/14) | 0.2 – 33.9 |
| Farmer | 18.2 (25/137) | 12.2 – 25.8 |
| Housewife | 19.2 (5/26) | 6.6 – 39.4 |
| Seller | 36.8 (7/19) | 16.3 – 61.6 |
| Teacher | 0.0 (0/2) | 0.0 – 84.2 |
| Other | 20.0 (1/5) | 0.5 – 71.6 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 19.6 (11/56) | 10.2 – 32.4 |
| Housewife | 44.4 (4/9) | 13.7 – 78.8 |
| Seller | 8.3 (1/12) | 0.2 – 38.5 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the zone of residence of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Rural | 38.0 (27/71) | 26.8 – 50.3 |
| Semi urban | 29.4 (5/17) | 10.3 – 56.0 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Rural | 19.7 (14/71) | 11.2 – 30.9 |
| Semi urban | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Rural | 14.1 (10/71) | 7.0 – 24.4 |
| Semi urban | 35.3 (6/17) | 14.2 – 61.7 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Rural | 49.4 (87/176) | 41.8 – 57.1 |
| Semi urban | 33.3 (9/27) | 16.5 – 54.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Did nothing when a child got hematuria | ||
| Rural | 19.7 (14/71) | 11.2 – 30.9 |
| Semi urban | 23.5 (14/17) | 6.8 – 49.9 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the religion of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Christian | 20.0 (8/40) | 9.1 – 35.7 |
| Muslim | 27.3 (3/11) | 6.0 – 61.0 |
| Traditional (Animism) | 56.8 (21/37) | 39.5 – 72.9 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Christian | 25.0 (10/40) | 12.7 – 41.2 |
| Muslim | 9.1 (1/11) | 0.2 – 41.3 |
| Traditional (Animism) | 13.5 (5/37) | 4.5 – 28.8 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Christian | 22.5 (9/40) | 10.8 – 38.5 |
| Muslim | 36.4 (4/11) | 10.9 – 69.2 |
| Traditional (Animism) | 8.1 (3/37) | 1.7 – 21.9 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Christian | 51.6 (50/97) | 41.2 – 61.8 |
| Muslim | 33.3 (4/12) | 9.9 – 65.1 |
| Traditional (Animism) | 44.7 (42/94) | 34.4 – 55.3 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Christian | 14.4 (14/97) | 8.1 – 23.0 |
| Muslim | 50.0 (6/12) | 21.1 – 78.9 |
| Traditional (Animism) | 20.2 (19/94) | 12.6 – 29.8 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Christian | 27.5 (11/40) | 14.6 – 41.9 |
| Muslim | 27.3 (3/11) | 6.0 – 61.0 |
| Traditional (Animism) | 10.8 (4/37) | 3.0 – 25.4 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
Table 5: Practices of the heads of household on hematuria in school aged children by sociodemographic characteristics, Wahala Health area, Haho Health District, Togo, March 2020.
Figures

Keywords
- Prevalence
- Knowledge
- Attitudes
- Practices
- Schistosomiasis
- Wahala
1Direction Préfectorale de la Santé de Haho, Notsè, Togo, 2African Field Epidemiology Network (AFENET). Regional Office for West Africa, Ouagadougou, Burkina Faso, 3Health and Development International (HDI), Bureau de Lomé, Togo, 4Direction de la Lutte contre la Maladie et des Programmes de Santé, Lomé, Togo, 5African Field Epidemiology Network, Kampala, Uganda
&Corresponding author: Agballa Mébiny–Essoh Tchalla Abalo, African Field Epidemiology Network, Regional office for Central Africa and Indian Ocean, Kinshasa, Avenue 24, Imeuble Infinity, 2ème étage, Bureau 204, Email: tchanaldinio@yahoo.fr ORCID: https://orcid.org/0000-0003-1896-0272
Received: 02 Dec 2024, Accepted: 09 Mar 2026, Published: 11 Mar 2026
Domain: Neglected Tropical Diseases
Keywords: Prevalence, Knowledge, Attitudes, Practices, Schistosomiasis, Wahala
©Agballa Mébiny–Essoh Tchalla Abalo et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Agballa Mébiny – Essoh Tchalla Abalo et al. Prevalence of urinary schistosomiasis in school-aged children, knowledge, attitude and practice of household heads regarding hematuria, Haho Health District, Togo. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):44. https://doi.org/10.37432/jieph-d-24-02000
Abstract
Introduction: In March 2020, the Wahala Health area in Haho Health District notified 47 confirmed cases of urinary schistosomiasis in school-aged children, despite 10 years of Praziquantel Mass Drug Administration (MDA), one of the WHO-recommended strategies for ending schistosomiasis by 2030. The district coordination team investigated the prevalence of schistosomiasis among school-aged children and assessed the heads of household’s knowledge, attitudes, and practices regarding hematuria.
Methods: This was a cross-sectional study conducted in Wahala Health area in March 2020. It included heads of households and their school-aged children. Schwartz’s formula, adjusted for a 10% non-response rate, determined the sample size. We interviewed the heads of households and collected data on their knowledge, attitudes, and practices on hematuria. Urine samples from all school-aged children found in the selected households were collected after a moderate exercise of 5 to 10 minutes for parasitological examinations at the Wahala Health Centre laboratory and the National Reference Laboratory to determine the prevalence. The laboratory performed direct microscopy of urine sediment after centrifugation to identify bilharzia eggs. Descriptive analysis was performed using Epi Info 7 with the results summarised as medians and interquartile ranges (IQR) and proportions with 95% confidence interval (95%CI).
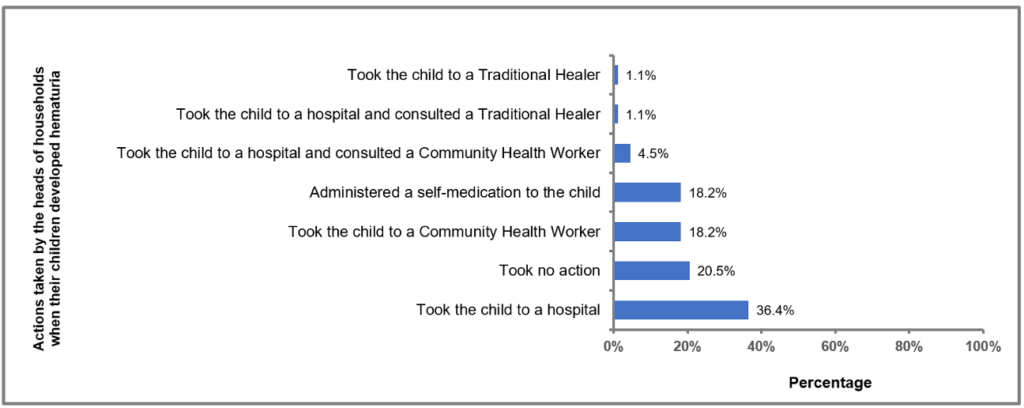
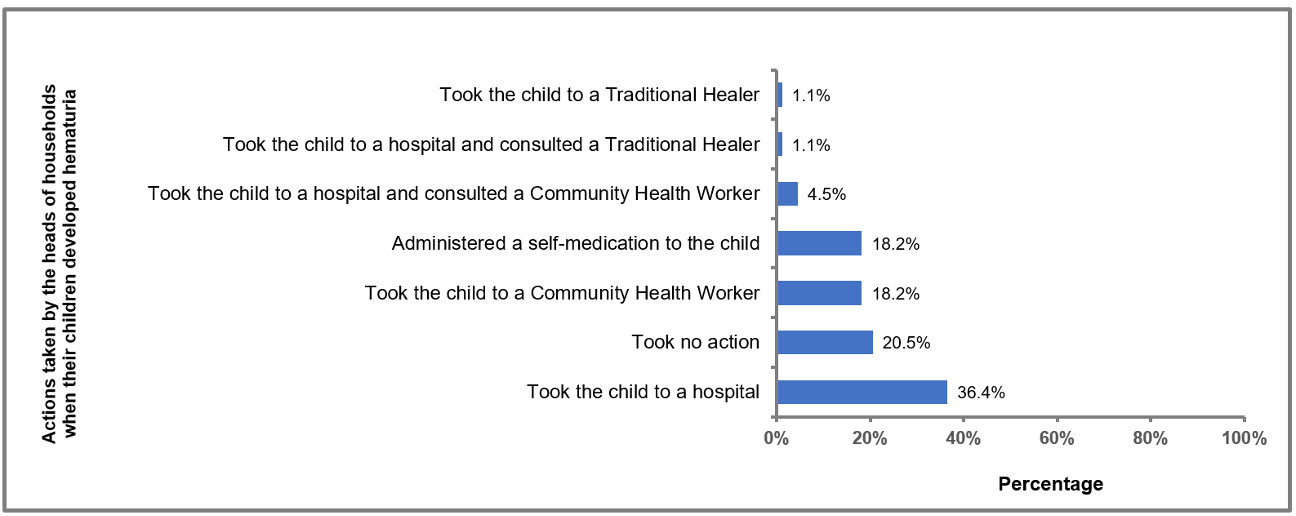
Results: A total of 427 heads of households were interviewed, of whom 33.6% were women, and 969 urine samples were collected. The overall prevalence of schistosomiasis was 31.8%(95%CI:28.9-34.8). Prevalence by gender and residence zone was 29.3%(95%CI:25.3-33.8) in girls, 33.8%(95% CI:29.9-37.9) in boys, 30.9%(95%CI:27.7 – 34.3) in rural and 34.7%(95%CI:28.4 – 41.3) in semi urban zones. Hematuria was reported as a disease by 80.6%(95%CI:76.6-84.1) of heads of households, and 81.5%(95%CI:77.8-84.90) considered swimming in rivers as a risk. They thought MDA: 40.1%(95%CI:35.5-44.8) and medical consultation: 41.7%(95%CI:37.1-46.4) contribute to preventing hematuria. Regarding what they would do in case a child of their household got hematuria, 83.1%(95%CI:79.3-86.4) and 11.7%(95%CI:9.0 – 11.2) declared they would take their child to a hospital or a Community Health Worker (CHW), respectively. Out of the 23.2%(95%CI:19.4–27.4) heads of households whose children experienced hematuria, 36.4%(95%CI:26.4 – 47.3) sought care at a hospital, 18.2%(95%CI:10.8–27.8) consulted a CHW, 1.4%(95%CI:0.04–7.6) consulted a traditional healer, 18.2%(95%CI:10.8–27.8) reported self-medication and 20.5%(95%CI:12.6–30.4) took no action. During the last MDA, 2.1%(95%CI:0.9-5.1) expressed resistance, and 1.4%(95% CI:0.7–3.0) refused to welcome CHWs into their households.
Conclusion: Schistosomiasis magnitude remains high despite 10 years of MDA, jeopardising progress towards the 2030 elimination target. Most heads of households knew what hematuria represents and how to prevent it, but there still exist practices contributing to its persistence. MDA and communication strategies should be redefined and combined with improved water and sanitation conditions and permanent free administration of Praziquantel in routine medical consultations.
Introduction
Schistosomiasis is a chronic Neglected Tropical Disease (NTD) caused by a parasite of the genus Schistosoma. Among the five species infecting humans, Schistosoma haematobium and Schistosoma mansoni are the predominant in Africa [1-4] and are responsible for urogenital and intestinal forms. At the infectious stage of the parasite, the larva, infects humans through skin contact with infected water. The main clinical features of the urogenital and intestinal forms are terminal hematuria and dysenteriform syndrome respectively. At least 230 million people worldwide are affected by schistosomiasis, of whom more than 90% live in sub-Saharan Africa [2-5]. Schistosomiasis was responsible for 2.12 million, 2.60 million, and 1.43 million Disability-Adjusted Life Years (DALY) lost in 1990, 2015, and 2017, respectively [6-8], and approximately 800,000 deaths each year worldwide [1]. Tropical and subtropical regions are the most affected by the disease, especially poor populations living in defective sanitation conditions and the lack of access to safe drinking water [5, 9, 10].
Schistosomiasis as part of the NTDs group was considered in the global strategic plan for NTDs control 2014 – 2020 [11] approved at the World Health Assembly in 2013 to eliminate schistosomiasis by 2020 [12]. The ambition of this plan has been reaffirmed and incorporated in the Sustainable Development Goals [13] and an updated roadmap was designed to ensure the end of NTDs by 2030 [14]. The recommended strategies to control schistosomiasis are the Mass Drug Administration (MDA) using Praziquantel, the therapeutic management of cases admitted in routine medical consultation, as well as communications supported by an epidemiological surveillance [14]. Between 2006 and 2016, implementation of the MDA contributed to reducing schistosomiasis-related mortality worldwide by 22% [5].
The national survey on NTDs carried out in Togo in 2009 reported a prevalence of urinary schistosomiasis of 23% nationwide and 42% in Haho Health District [15]. To reduce this prevalence, the MDA strategy using Praziquantel [11-13] as recommended by the World Health Organization (WHO) has been implemented since 2010. In 2015, the evaluation of the impact of this strategy revealed a prevalence of schistosomiasis of 4.20% nationwide, 11% to 20% in Haho Health District, and 0% in Wahala health area [16]. These findings led to a change in the frequency of MDA from one round every year to one round every two years. For all the Praziquantel MDA implemented, the targets were achieved in all health areas, including Wahala’s.
Schistosomiasis surveillance in Togo, is a passive system, and routine data are from hospitals and health facilities registers. These data are collected and compiled monthly in District Health Information System, version 2 (DHIS2) software and used to prepare the National Statistical Yearbook. According to the national statistics, the total number of cases of Schistosoma haematobium and Schistosoma mansoni recorded in Togo was 394 and 1,059, respectively, in 2017 and 2018, while in the Plateaux region, where the Haho Health District is located, it was 145 and 248, respectively, in the same periods [17, 18]. In March 2020, Wahala Health Centre notified 50 cases of hematuria in school-aged children, of which 47 were parasitologically confirmed as urinary schistosomiasis. This situation raised concerns about a possible reinfestation, prompting an investigation to determine the extent of the phenomenon and assess community understanding, with the aim of strengthening control strategies. The study determined the prevalence of urinary schistosomiasis among school-aged children, and described the knowledge, attitudes and practices of heads of households regarding hematuria in the Wahala Health area in 2020.
Methods
Study setting
Wahala Health area is a semi-urban and rural zone located in Haho Health District in southern Togo (Figure 1) with a population consisting mainly of farmers. The geographical accessibility to health facilities, estimated by the proportion of the population residing within five kilometres from a health center was 40% in 2020. This health area is crossed by two rivers used by people, including school-aged children, for drinking, bathing, washing, and farming. Wahala Health Centre has facilities to confirm schistosomiasis diagnosis. Patients diagnosed with schistosomiasis at the healthcare unit were prescribed Praziquantel, the recommended treatment. However, Praziquantel was not available at Wahaha Health Centre and patients could only get it from a private pharmacy located 16.5 miles from Wahaha. The price of one pack of Praziquantel containing six tablets was 9,185 XOF (about $15; exchange rate on 30th April 2020).
Study design and population
This study was a descriptive cross-sectional study carried out in Wahala Health area in March 2020. It included heads of households and school-aged children (5-14 years old). The sample size of heads of households was calculated using the Schwartz formula, adjusted for a 10% non-response rate, at 95% Confidence Interval (95% CI) and a margin of error of 5%. Assuming that 50% of household heads had knowledge of hematuria with a 95% CI for a Zα value equal to 1.96, the sample size of household heads was 422. Households were selected using a systematic sampling method with the sampling interval determined by the quotient between the total number of households in the health zone and the sample size. For a total number of 4,846 households, the sampling step size was 11. School-aged children were selected by a simple random method, considering all those living in the households and found at home during the survey.
Case definition
Urinary schistosomiasis was defined as the presence of S. haematobium eggs in a urine sample, as determined by microscopic examination of the urine sediment after centrifugation.
Variable assessment
Socio-demographic variables data of the heads of households were age, sex, residence zone, education level and religion. Knowledge of the heads of households was assessed by examining their response to what is hematuria, haematuria’s name in the native language, what they know about its transmission, the existence of a risk for people swimming, bathing, or walking in rivers, the existence and the complications of hematuria, the causes of hematuria, the existence of control strategies and the target population for MDA. Attitudes of the heads of households were assessed by the presence of the heads of households at home during the last MDA, measures taken to ensure the MDA target children were compliant in taking the medication, measures taken to control hematuria, and what they would do in case a child of the household gets hematuria. Practices of the heads of households were assessed by the actions taken when a child experienced hematuria, the measures taken when they saw or had evidence that children swim in rivers, and reluctance and resistance in the household to participate during the last MDA prior to the survey. Urine macroscopic aspect was described as clear, turbulent, and bloody, while its microscopic assessment was described as positive or negative.
Data collection and techniques
Data were collected by trained and supervised teams after the data collection forms had been pretested. Heads of households were interviewed about their knowledge, attitudes and practices regarding hematuria using a questionnaire. Urine samples were collected from school-aged children after a moderate exercise for 5 to 10 minutes.
Laboratory methods
The samples were examined the day of collection at Wahala Health Center laboratory by two laboratory scientists using a binocular optical microscope branded “SCOT” (110 series). The macroscopic and microscopic results were transcribed on a designed survey form. Urine collected after 5 to 10 minutes of moderate physical effort was first decanted and later centrifuged. The urine sediment was examined by microscopy to identify terminal spurred bilharzia eggs. All the urine samples with a sticker ranked “20” or “20” multiple, after being analyzed at Wahala Health Center’s laboratory, were conditioned and sent within 72 hours to the Institut National d’Hygiène, the national reference laboratory in Lomé, for external quality control.
Data processing and analysis
The survey forms were reviewed by the supervisors daily to check the quality of the data collected. Data were first processed in Microsoft Excel version 97–2003 using a predesigned spreadsheet, and thereafter imported into Epi-Info 7.2.1.0. Descriptive analysis was performed, and the results were presented using proportions at 95% CI, median with an interquartile range (IQR). Prevalence was determined by the proportion of samples with a positive laboratory result out of the total number of samples tested. The p-value of Chi-square or Fisher Exact test was used to assess the statistical difference between proportions.
Ethical considerations
Approval was obtained from the National Directorate for Disease Control and Public Health Programmes in accordance with note N°0023/2020/MSHPAUS/CAB/SG/DGAS/DLMPSP prior to conducting the study. The heads of households gave their free and informed oral consent before the interviews took place and the urine samples were collected from school-aged children. Data were collected and processed anonymously and with strict confidentiality. Children with macroscopic hematuria, or who were diagnosed with schistosomiasis following a laboratory examination, were given a free dose of praziquantel according to the MDA guidelines.
Results
Characteristics of study participants
A total of 427 heads of households were interviewed, and 969 urine samples were collected from school-aged children. The median age of the heads of households was 40 years (IQR: 35 – 50). The median age of the children was 9 years (IQR: 7 – 11) and was the same in both boys and girls. Women accounted for 33.6% (95% CI: 29.3 – 38.2) of the heads of households, and boys represented 54.3% (95% CI: 51.1 – 57.4) of the children. Regarding the place of residence, occupation, and education level, 20.8% (95%CI: 17.3 – 25.0) heads of households were from semi-urban area, 79.8% (95% CI: 75.1 – 82.7) from rural area, 63.8% (95%CI: 59.3 – 68.4) were farmers and 45.9% (95% CI: 41.2 – 50.6) have never been to school.
Prevalence of urinary schistosomiasis
A total of 969 urine samples were tested, 47 had external quality control, out of which 18 were positive for schistosomiasis, aligning with the same 18 samples that tested positive at Wahala Health Centre laboratory, making it 100% match of quality result. Urine macroscopic aspect was clear: 48.1% (95% CI: 45.0 – 51.2), turbulent: 44.4% (95%CI: 41.3 – 47.5) and bloody: 7.3% (95%CI: 6.0 – 9.4). The overall prevalence of urinary schistosomiasis was 31.8% (95%CI: 28.9 – 34.8) and varied from 29.3% (95%CI: 25.3 – 33.8) in girls to 33.8% (95%CI: 29.9 – 37.9) in boys and 30.0% (95% CI: 26.3 – 34.0) in children less than 10 years old to 34.0% (95%CI: 29.8 – 38.6) in those aged over 10 years. Schistosomiasis was positive in 30.9% (95%CI: 27.1 – 34.3) of children tested in the rural area, while 34.7% (95%CI: 28.4 – 41.3) were positive in the semi-urban area. Prevalence of schistosomiasis in children of heads of household who have never been to school was 37.1% (95%CI: 32.7 – 41.7) and 27.4% (95% CI:23.8 – 31.4) in children of heads of household who attended at least the primary school (Table 1). Considering the urine macroscopic aspect, the prevalence was 25.8% (95%CI: 22.0 – 29.9) in children with clear urine, 30.7% (95%CI: 26.5 – 35.2) in children with turbulent urine and 76.7% (65.4 – 85.8) in those with bloody urine.
Knowledge of heads of households
Almost all 99.5% (95%CI: 98.0 – 100.0) of heads of household interviewed recognised hematuria as a sign of disease. The proportion remained the same even among the heads of households who have never been to school, 99.5% (95%CI: 97.1 – 100.0). Only 6.2% (95%CI: 3.6 – 9.6) of men and 3.5% (95%CI: 1.2 – 8.0) of women knew the name of the disease as “Bilharzia”; however, 72.6% (95%CI: 68.2 – 76.6) of heads of household were able to provide the name of hematuria in their native language. Two heads of household, 0.4% (95% CI: 0.1 – 1.7) reported hematuria as witchcraft (Table 2).
About 73.6% (95%CI: 69.2 – 77.6) knew that people of any age group can be affected. Heads of households who knew that swimming or walking in rivers or ponds was a risk were 97.9% (95%CI: 96.0 – 98.9) and 81.5% (95%CI: 74.0 – 92.4) reported that “urinating blood” is one of the consequences of swimming or walking in rivers or ponds. About 86.4% (95%CI: 82.84 – 89.34) knew that hematuria could be treated or controlled. The medical consultation: 40.3% (95%CI: 35.7 – 45.0) and MDA: 40.1% (95%CI: 35.5– 44.8) were cited as the main strategies for prevention, treatment and control (Table 3).
Attitudes
Out of the heads of household interviewed, 93.2% (95%CI: 90.4 – 95.2) planned to be at home on the day the CHW visited their house during the last MDA. When heads of household were asked about what they would do in case a child of their household got hematuria, 71.0% (95%CI: 66.5 – 75.1) and 11.7% (95%CI: 9.0 – 11.2) declared they would take the child to the hospital or consult a CHW, respectively. When stratified by zone of residence, 65.1% (95%CI: 59.9 – 70.0) and 14.20% (95%CI: 10.9 – 18.3) would take their child to hospital and CHW in rural area, while 92.3% (95%CI: 85.9 – 97.5) and 2.3% (95%CI: 0.3 – 7.9) will take their child to hospital and CHW in semi-urban area (Table 4).
Practices
From January 2019 to March 2020, 23.2% (95%CI: 19.4 – 27.4) of households experienced at least one episode of hematuria. When it occurred, 36.4% (95%CI: 26.4 – 47.3) of heads of households took the children to the hospital, 18.2% (95%CI: 10.8–27.8) took the children to a CHW, 1.4% (95%CI: 0.04 – 7.6) consulted a traditional healer, 18.2% (95%CI: 10.8 – 27.8) self-medicated the children and 20.5% (95%CI: 12.6 – 30.4) did nothing (Figure 2). During the last MDA, 0.7% (95%CI: 0.2 – 2.1) heads of households hid the MDA target children to avoid them taking medication, and 1.4% (95%CI:0.7 – 3.0) refused to welcome CHWs in their household (Table 5).
Discussion
The study revealed the prevalence of urinary schistosomiasis in school-aged children and assessed the knowledge, attitudes, and practices of heads of households in Wahala Health Center area. Nearly a third of children were infected. The progression in the schistosomiasis prevalence from 0% in 2015 to the current value is consistent with a reinfestation and confirms the endemicity of urinary schistosomiasis in the Wahala Health area.
Urinary schistosomiasis remains a public health concern in that area despite several years of successful MDA. Such persistence of a high prevalence after several years of MDA implementation was also reported in Senegal [19]. The prevalence indicates the magnitude of the disease, classifying the Wahala Health area as a moderate prevalence zone [20]. This reinfestation is a critical challenge for the control and elimination of schistosomiasis [21] and had been predicted in the MDA impact study conducted in Togo in 2015 [22].
This reinfestation may have multiple, interdependent causes, including a change in the frequency of MDA from once a year between 2010 and 2015, to once every two years after 2015; non-implementation of MDA in the Wahala area in 2019 due to the unavailability of Praziquantel; lack of therapeutic management of cases detected during routine medical consultations; low access to drinking water and sanitation; and poor outreach communication for schistosomiasis control. Such reinfestation was also reported in Ghana in 2019 [23]. Other studies carried out in Côte d’Ivoire [24], Mali [25] in a context of several years of MDA implementation also reported prevalence between 10% and 50%, classifying these areas as moderate-risk zones like Wahala. However, in Ethiopia [26] and Kenya [27] lower prevalence of 0.3% had been reported after several years of MDA.
The heads of households had the required knowledge to understand what schistosomiasis is and how to control it. The impact of this knowledge is reflected in the measures taken, as well as the various attitudes adopted by households to prevent and control bilharzia. If these attitudes were implemented, their combination with MDA performed since 2010 would have had a sustained impact on the prevalence among school-aged children. For most heads of households, hematuria was a sign of pathological condition, and they knew what attitude or practice can be responsible for it. This shows that most of them have a good knowledge of the mechanisms of the occurrence of hematuria in school-aged children despite the reinfestation. Some studies conducted in Nigeria, Gabon, Kenya, Uganda, South Africa, Yemen, and Philippines also reported such high proportions of heads of households who had a good knowledge [28-34]. Conversely, other studies carried out in Togo, Cameroon, Ethiopia, Côte d’Ivoire and Mauritania reported a lower level of knowledge of heads of households about schistosomiasis and its associated signs [35-38]. This difference could be explained by the epidemiological context, the quality of the communication strategies used to raise community awareness of the disease and its modes of contamination and the criteria used to categorize the respondents’ knowledge.
Having as high as 20% of heads of households who could not state or reported inappropriate causes of hematuria among the study population, suggests that more educational strategies and public awareness campaigns need to be put in place. The plurality of names attributed to hematuria in native languages are due to the multiplicity of different linguistic groups residing in Wahala zone. For the great majority of the linguistic groups living in Wahala, the names translate the emission of bloody urine: Him Ni Tchalim and Himah in Kabyè-Lamba language, Ehoudoudo and Adoudove in Adja-Ewé, Hawem djim and Ahoran djim in Naouda. Such names as “Sugunèbilen or Gnèguènèbilen” in Bambara or Dioula language spoken in Mali, Burkina Faso, Côte d’Ivoire and Guinea also express the presence of blood in the urine [39]. The name “Eyawou” is more related to the dysuria and pain associated with hematuria. Similar translating names for signs and symptoms have been reported in Côte d’Ivoire and Mauritania [38].
The majority of heads of households were found in their respective homes during the last MDA with PRZ and they reported having implemented appropriate measures in their households to control schistosomiasis. These findings corroborate knowledge they have regarding hematuria, considered as a sign of pathological condition making it as one of priority events under surveillance in their households. This attitude also showed that a high proportion of heads of households in Wahala Health area believed that hematuria is a medical condition for which there are means of prevention and treatment. Several studies reported similar proportions of appropriate attitudes among heads of households [28-30, 34].
Only one in ten households that experienced an episode of hematuria undertook an appropriate action. This low proportion of appropriate practices regarding hematuria, contrasted with the knowledge and attitudes revealed in this study. More than a third of heads of households had an inappropriate action, reflecting insufficiencies in the adoption of good practices for hematuria control. The reasons for these inappropriate practices may be related to the poor geographic access to health care facilities by communities. Such a paradox between good knowledge and practices has earlier been also reported [28-30]. The very low proportion of reluctance and resistance during the last MDA revealed by the study is not uncommon during NTDs’ mass drug campaigns and other health-related mass activities such as immunisation campaigns. This illustrates the persistence of communities that have not yet adopted this control strategy, as was reported in several studies [40-44] and calls for the implementation of specific strategies.
The biological diagnostic technique for schistosomiasis, based on the search for bilharzia eggs in a urine sample using direct microscopy, could underestimate the prevalence and be a limitation of this study. However, the external quality control carried out at Togo’s national reference laboratory confirmed the quality of the results obtained at the Wahala Health Center laboratory. In addition, the parasite load could have provided information on the intensity of the infestation, but this information does not interfere with the determination of prevalence and the criteria for the MDA strategy to be implemented, nor the treatment dosage to be prescribed.
Conclusion
This study revealed a high prevalence of urinary schistosomiasis in school-aged children in the Wahala Health area despite the MDA undertaken since 2010, and this is compatible with reinfestation. Heads of households have a good knowledge of schistosomiasis, its clinical manifestations as well as its means of control, and most of them have ensured that all target children benefit from the MDA interventions. To increase and sustain the impact of schistosomiasis control interventions in this health area, some actions need to be undertaken. The frequency of MDA should be reversed to one round every year and involve all eligible people in the Wahala Health area. It’s also critical to make the Praziquantel tablets permanently available at the Wahala Health Center for free administration to people with hematuria or tested positive for schistosomiasis during routine and outreach medical consultations.
To track the trends, it will be important to conduct periodic surveys to monitor and update the mapping of schistosomiasis prevalence in the Haho Health District. The schistosomiasis indicators-based surveillance system currently in place should be strengthened by combining it with the event-based one, especially community-based surveillance. Wahala Health Center, with the support of the Haho Health District coordination team, should also develop and implement a communication plan for schistosomiasis control and conduct advocacy to improve the population’s access to safe drinking water, hygiene and sanitation.
- Schistosomiasis is a Neglected Tropical Disease, and Africa hosts 90% of the disease burden.
- Prevalence is declining in Togo with the Praziquantel Mass Distribution campaigns.
- Reinfestation with high prevalence despite a decade of Praziquantel MDA implementation.
- Knowledge and perception of the population on hematuria in a health area.
- Better knowledge of the epidemiological aspects of schistosomiasis in Wahala Health area.
- Identification and implementation of control strategies specific to the context of Wahala Health area.
Acknowledgements
The authors thank the Ministry of Health of Togo, the National Directorate for Disease Control and Health Programs, the National Program for NTDs control, the National Institute of Hygiene, the Directorate of Plateau Health Region, the Directorate of Haho Health District, the mayor of Haho 4 Commune and the personnel of Wahala Health Center.
Authors´ contributions
Agballa Mébiny – Essoh Tchalla Abalo developed the protocol of the study, participated in data collection, performed data processing and analysis, developed the manuscript, and its review.
Akara Essona Matatom participated in the protocol preparation, supervised the survey, and participated in data processing and analysis, manuscript development, and its review.
Edem Kpeglo, Koffi Messa Novinyo Edoh and Eyabo Balouki participated in data collection, supervision and the manuscript review.
Assane Hamadi, Bernard Sawadogo, Yao Kassankognon and Simon Antara reviewed the study protocol, the study analysis and the manuscript. All authors read and approved the final manuscript.
| Variable | Proportion (%) | 95% CI |
|---|---|---|
| Children age (years) | ||
| 5–9 | 30.0 (162/540) | 26.3 – 34.0 |
| 10–14 | 34.0 (146/429) | 29.8 – 38.6 |
| Children sex | ||
| Male | 32.8 (210/641) | 29.2 – 36.5 |
| Female | 29.9 (98/328) | 25.2 – 35.0 |
| Place of residence of heads of households | ||
| Rural | 30.9 (231/747) | 27.7 – 34.3 |
| Semi-urban | 34.7 (77/222) | 28.4 – 41.3 |
| Education level of the heads of households | ||
| Never been in school | 37.1 (163/440) | 32.7 – 41.7 |
| Primary school | 31.1 (97/312) | 26.2 – 36.4 |
| Secondary school | 22.4 (47/210) | 16.9 – 28.6 |
| University | 14.3 (1/6) | 0.4 – 57.9 |
| Names of hematuria in local languages | Number | Percentage | 95% CI |
|---|---|---|---|
| Community of Kabyè-Lamba (Respondents: 165/221) | |||
| Eyawou | 128 | 77.6 | 70.4 – 83.7 |
| Himah | 18 | 10.9 | 6.6 – 16.7 |
| Him Ni Tchalim | 17 | 10.3 | 6.1 – 16.0 |
| Kpètrè | 1 | 0.6 | 0.02 – 3.3 |
| Ouma | 1 | 0.6 | 0.02 – 3.3 |
| Community of Naouda (Respondents: 75/86) | |||
| Hawem djim | 39 | 52.0 | 40.2 – 63.7 |
| Ahoran djim | 36 | 48.0 | 36.3 – 59.9 |
| Community of Adja-Ewé-Fon (Respondents: 51/84) | |||
| Ehoudoudo | 39 | 76.5 | 62.5 – 87.2 |
| Adoudove | 12 | 23.5 | 12.8 – 37.5 |
| Community of Kotocoli (Respondents: 8/18) | |||
| Kpédéyou | 4 | 50.0 | 15.7 – 84.3 |
| Tchesso | 3 | 37.5 | 8.5 – 75.5 |
| Fim nazima | 1 | 12.5 | 0.3 – 52.7 |
| Community of Moba (Respondents: 6/7) | |||
| Fida | 1 | 16.7 | 0.4 – 64.1 |
| Hogna ninesoum | 1 | 16.7 | 0.4 – 64.1 |
| Ngnani Nassou | 1 | 16.7 | 0.4 – 64.1 |
| Oninyiam nissan | 1 | 16.7 | 0.4 – 64.1 |
| Ogmoriouso | 1 | 16.7 | 0.4 – 64.1 |
| Song | 1 | 16.7 | 0.4 – 64.1 |
| Community of Ifè (Respondents: 3/3) | |||
| Nto Owoun Onbarz | 2 | 66.7 | 9.43 – 99.16 |
| Toembra | 1 | 33.3 | 0.84 – 90.57 |
| Community of Ana (Respondents: 2/2) | |||
| Etraba | 2 | 100 | 15.8 – 100.0 |
| Variables | Proportion | 95% CI |
|---|---|---|
| Knowledge on hematuria in school aged children by gender of the head of household | ||
| Hematuria is sign of a disease | ||
| Men | 99.6 (274/275) | 97.99 – 99.99 |
| Women | 99.3 (140/141) | 96.1 – 99.98 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Knew the name of hematuria in the local language | ||
| Men | 80.2 (227/283) | 75.1 – 84.7 |
| Women | 57.3 (82/143) | 48.8 – 65.6 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Men | 48.7 (130/267) | 42.6 – 54.9 |
| Women | 33.3 (46/138) | 25.5 – 41.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Men | 73.7 (207/281) | 68.1 – 78.7 |
| Women | 73.2 (104/142) | 65.2 – 80.8 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in them exposes one to risk of hematuria | ||
| Men | 98.9 (278/281) | 96.9 – 99.8 |
| Women | 95.7 (134/140) | 90.9 – 98.4 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any means of control of hematuria | ||
| Men | 4.2 (12/283) | 2.2 – 7.3 |
| Women | 1.4 (2/143) | 0.2 – 5.0 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Men | 38.9 (110/283) | 33.2 – 44.8 |
| Women | 42.7 (82/143) | 34.4 – 51.2 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Men | 44.2 (125/283) | 38.3 – 50.2 |
| Women | 32.2 (46/143) | 26.6 – 40.5 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children by age groups of the heads of household | ||
| Hematuria is a sign of a disease | ||
| Age (years) | ||
| 20- <30 | 97.6 (41/42) | 87.4 – 99.9 |
| 30-<45 | 100.0 (193/193) | 98.1 – 100.0 |
| 45-<60 | 100.0 (121/121) | 97.0 – 100.0 |
| 60-90 | 98.4 (60/61) | 91.2 – 99.9 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of hematuria in the local language | ||
| Age (years) | ||
| 20- <30 | 66.7 (28/42) | 50.5 – 80.4 |
| 30-<45 | 74.8 (148/198) | 68.1 – 80.6 |
| 45-<60 | 72.8 (91/125) | 64.1 – 80.9 |
| 60-90 | 69.4 (43/62) | 56.4 – 80.4 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Age (years) | ||
| 20- <30 | 40.50 (19/40) | 31.5 – 63.9 |
| 30-<45 | 44.68 (84/188) | 37.4 – 52.1 |
| 45-<60 | 43.33 (52/120) | 34.3 – 52.7 |
| 60-90 | 37.93 (22/58) | 25.5 – 51.6 |
| Overall | 43.60 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Age (years) | ||
| 20- <30 | 74.4 (30/42) | 55.4 – 84.3 |
| 30-<45 | 72.5 (142/196) | 65.6 – 78.6 |
| 45-<60 | 75.8 (94/124) | 67.3 – 83.0 |
| 60-90 | 74.2 (46/62) | 61.5 – 84.5 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in it exposes one to risk of hematuria | ||
| Age (years) | ||
| 20- <30 | 100.00 (41/41) | 91.4 – 100.0 |
| 30-<45 | 97.96 (192/196) | 94.9 – 99.4 |
| 45-<60 | 96.77 (120/124) | 91.9 – 99.1 |
| 60-90 | 98.36 (60/61) | 91.2 – 99.9 |
| Overall | 97.87 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Age (years) | ||
| 20- <30 | 4.8 (2/42) | 0.58 – 16.2 |
| 30-<45 | 0.5 (1/198) | 0.01 – 2.8 |
| 45-<60 | 7.2 (9/125) | 3.35 – 13.2 |
| 60-90 | 3.2 (2/62) | 0.39 – 11.2 |
| Overall | 3.3 (14/427) | 1.96 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Age (years) | ||
| 20- <30 | 42.9 (18/42) | 27.7 – 59.0 |
| 30-<45 | 37.4 (74/198) | 30.6 – 44.5 |
| 45-<60 | 39.2 (49/125) | 30.6 – 48.3 |
| 60-90 | 50.0 (31/62) | 37.0 – 62.9 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Age (years) | ||
| 20- <30 | 26.2 (11/42) | 13.86 – 42.0 |
| 30-<45 | 41.9 (83/198) | 34.9 – 49.1 |
| 45-<60 | 44.8 (56/125) | 35.9 – 53.9 |
| 60-90 | 33.9 (21/62) | 22.3 – 47.0 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the education level of the heads of household | ||
| Hematuria is a disease | ||
| Never been at school | 99.5 (191/192) | 97.1 – 99.9 |
| Primary school | 100.0 (125/125) | 97.1 – 100.0 |
| Secondary school | 99.0 (95/96) | 94.3 – 99.9 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Never been at school | 70.4 (138/196) | 63.5 – 76.7 |
| Primary school | 72.7 (93/128) | 64.1 – 80.2 |
| Secondary school | 77.8 (77/99) | 68.3 – 85.5 |
| University | 50.0 (2/4) | 6.8 – 93.2 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Never been at school | 42.6 (80/188) | 35.4 – 49.9 |
| Primary school | 37.5 (45/120) | 28.8 – 46.8 |
| Secondary school | 52.1 (49/94) | 41.6 – 62.5 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Never been at school | 76.4 (149/195) | 69.8 – 82.2 |
| Primary school | 67.7 (86/127) | 58.9 – 75.7 |
| Secondary school | 75.5 (74/98) | 65.8 – 83.6 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Never been at school | 97.4 (188/193) | 94.1 – 99.2 |
| Primary school | 96.8 (122/126) | 92.1 – 99.1 |
| Secondary school | 100.0 (99/99) | 96.3 – 100.0 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Never been at school | 2.6 (5/196) | 0.8 – 5.9 |
| Primary school | 4.7 (6/128) | 1.7 – 9.9 |
| Secondary school | 3.0 (3/99) | 0.6 – 8.6 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Never been at school | 40.3 (79/196) | 33.4 – 47.5 |
| Primary school | 42.2 (54/128) | 33.5 – 51.2 |
| Secondary school | 38.4 (38/99) | 28.8 – 48.7 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Never been at school | 36.7 (72/196) | 56.1 – 70.0 |
| Primary school | 36.7 (47/128) | 28.4 – 45.7 |
| Secondary school | 50.5 (50/99) | 40.3 – 60.7 |
| University | 50.0 (2/4) | 6.76 – 93.2 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the profession of the heads of household | ||
| Hematuria is a disease | ||
| Artisan | 100.0 (30/30) | 88.4 – 100.0 |
| Farmer | 99.6 (266/267) | 97.9 – 99.9 |
| Housewife | 100.0 (51/51) | 93.0 – 100.0 |
| Seller | 97.7 (42/43) | 87.7 – 99.9 |
| Teacher | 100.0 (9/9) | 66.4 – 100.0 |
| Other | 100.0 (17/17) | 80.5 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Artisan | 70.0 (21/30) | 50.6 – 86.3 |
| Farmer | 78.8 (215/273) | 73.4 – 83.5 |
| Housewife | 71.2 (37/52) | 56.9 – 82.9 |
| Seller | 43.2 (19/44) | 28.4 – 58.9 |
| Teacher | 54.6 (6/11) | 23.4 – 83.3 |
| Other | 70.6 (12/17) | 44.0 – 89.7 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Artisan | 53.6 (15/28) | 33.9 – 72.5 |
| Farmer | 40.4 (105/260) | 34.4 – 46.6 |
| Housewife | 50.0 (25/50) | 35.5 – 64.5 |
| Seller | 38.1 (16/42) | 23.6 – 54.4 |
| Teacher | 63.6 (7/11) | 30.8 – 89.1 |
| Other | 60.0 (9/15) | 12.3 – 83.7 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Artisan | 66.7 (20/30) | 47.2 – 82.7 |
| Farmer | 71.1 (192/270) | 65.3 – 76.4 |
| Housewife | 75.0 (39/52) | 61.1 – 85.9 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 72.7 (8/11) | 39.0 – 93.9 |
| Other | 82.4 (14/17) | 52.6 – 96.2 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Artisan | 90.0 (27/30) | 73.5 – 98.0 |
| Farmer | 99.3 (268/270) | 97.4 – 99.9 |
| Housewife | 94.1 (48/51) | 83.8 – 98.8 |
| Seller | 97.7 (43/44) | 88.0 – 99.9 |
| Teacher | 100.0 (11/11) | 71.5 – 100.0 |
| Other | 100.0 (16/16) | 79.4 – 100.0 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any means of control of hematuria | ||
| Artisan | 6.7 (2/30) | 0.8 – 22.1 |
| Farmer | 3.3 (9/273) | 1.5 – 6.2 |
| Housewife | 3.9 (2/52) | 0.5 – 13.2 |
| Seller | 0.0 (0/44) | 0.0 – 8.0 |
| Teacher | 0.0 (0/11) | 0.0 – 28.5 |
| Other | 5.9 (1/17) | 0.2 – 28.7 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Artisan | 26.7 (8/30) | 12.3 – 45.9 |
| Farmer | 42.1 (115/273) | 36.2 – 48.2 |
| Housewife | 28.9 (15/52) | 17.1 – 43.1 |
| Seller | 47.7 (21/44) | 32.5 – 63.3 |
| Teacher | 81.8 (9/11) | 48.2 – 97.7 |
| Other | 23.5 (4/17) | 48.2 – 97.7 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Artisan | 60.0 (18/30) | 22.7 – 59.4 |
| Farmer | 37.0 (101/273) | 31.3 – 43.0 |
| Housewife | 36.5 (19/52) | 23.6 – 51.0 |
| Seller | 45.5 (20/44) | 30.4 – 61.2 |
| Teacher | 45.5 (5/11) | 16.8 – 76.6 |
| Other | 47.1 (8/17) | 22.9 – 72.2 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to residence zone of the heads of household | ||
| Hematuria is a disease | ||
| Rural | 99.4 (328/330) | 97.8 – 99.8 |
| Semi urban | 100.0 (87/87) | 95.9 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of the hematuria in the local language | ||
| Rural | 79.3 (268/338) | 74.7 – 83.3 |
| Semi urban | 47.2 (42/89) | 36.5 – 58.1 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Rural | 42.6 (139/326) | 37.4 – 48.1 |
| Semi urban | 47.5 (38/80) | 36.2 – 58.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Rural | 71.6 (240/335) | 66.6 – 76.2 |
| Semi urban | 80.9 (72/89) | 71.2 – 88.5 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swiming in rivers, ponds or walking in is at risk of hematuria | ||
| Rural | 97.6 (325/333) | 95.3 – 98.8 |
| Semi urban | 98.9 (88/89) | 93.9 – 99.9 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Rural | 3.6 (12/338) | 2.0 – 6.1 |
| Semi urban | 2.3 (2/89) | 0.3 – 7.9 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Rural | 37.3 (126/338) | 32.3 – 42.6 |
| Semi urban | 51.7 (46/89) | 40.8 – 62.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Rural | 40.8 (138/338) | 35.7 – 46.1 |
| Semi urban | 37.1 (33/89) | 27.1 – 47.9 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Knowledge on hematuria in school aged children according to the religion of the heads of household | ||
| Hematuria is a disease | ||
| Christian | 98.9 (192/194) | 96.3 – 99.9 |
| Muslim | 100.0 (33/33) | 89.4 – 100.0 |
| Traditional (Animism) | 100.0 (190/190) | 98.1 – 100.0 |
| Overall | 99.5 (415/417) | 98.3 – 99.9 |
| Know the name of hematuria in the local language | ||
| Christian | 71.9 (143/199) | 65.1 – 78.0 |
| Muslim | 54.3 (19/35) | 36.7 – 71.2 |
| Traditional (Animism) | 76.7 (148/193) | 70.1 – 82.5 |
| Overall | 72.6 (310/427) | 68.2 – 76.6 |
| Hematuria is contagious | ||
| Christian | 41.9 (81/193) | 34.9 – 49.3 |
| Muslim | 55.2 (16/29) | 35.7 – 73.6 |
| Traditional (Animism) | 43.5 (80/184) | 36.2 – 50.9 |
| Overall | 43.6 (177/406) | 38.9 – 48.5 |
| Hematuria can affect people of any age group | ||
| Christian | 71.2 (141/198) | 64.4 – 77.4 |
| Muslim | 97.1 (34/35) | 85.1 – 99.9 |
| Traditional (Animism) | 71.7 (137/191) | 64.8 – 78.0 |
| Overall | 73.6 (312/424) | 69.2 – 77.6 |
| Swimming in rivers, ponds or walking in is at risk of hematuria | ||
| Christian | 97.9 (193/197) | 94.9 – 99.4 |
| Muslim | 94.1 (32/34) | 80.3 – 99.3 |
| Traditional (Animism) | 98.4 (188/191) | 95.5 – 99.7 |
| Overall | 97.9 (413/422) | 96.0 – 98.9 |
| Don’t know any mean of control of hematuria | ||
| Christian | 2.5 (5/199) | 0.8 – 5.8 |
| Muslim | 0.0 (0/35) | 0.0 – 10.0 |
| Traditional (Animism) | 4.7 (9/193) | 2.2 – 8.7 |
| Overall | 3.3 (14/427) | 2.0 – 5.4 |
| Medical consultation is one of the means to control hematuria | ||
| Christian | 45.7 (91/199) | 38.7 – 52.9 |
| Muslim | 37.1 (13/35) | 21.5 – 55.1 |
| Traditional (Animism) | 35.2 (68/193) | 28.5 – 42.4 |
| Overall | 40.3 (172/427) | 35.7 – 45.0 |
| Mass Drug Administration is one of the means to control hematuria | ||
| Christian | 42.2 (84/199) | 35.3 – 49.4 |
| Muslim | 37.1 (13/35) | 21.4 – 55.1 |
| Traditional (Animism) | 38.3 (74/193) | 31.5 – 45.6 |
| Overall | 40.1 (171/427) | 35.5 – 44.8 |
| Variables | Proportion | 95% CI |
|---|---|---|
| Attitudes on hematuria in school aged children according to the gender of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Men | 92.9 (263/283) | 89.3 – 95.6 |
| Women | 93.7 (134/143) | 88.4 – 97.1 |
| Overall | 93.2 (415/417) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Men | 65.7 (186/283) | 59.9 – 71.2 |
| Women | 81.1 (116/143) | 73.7 – 87.2 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Men | 14.1 (40/283) | 10.3 – 18.8 |
| Women | 7.0 (10/143) | 3.4 – 12.5 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Men | 39.0 (110/282) | 33.3 – 44.9 |
| Women | 43.4 (62/143) | 35.1 – 51.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Men | 14.9 (42/282) | 11.0 – 19.6 |
| Women | 10.5 (15/143) | 6.0 – 16.7 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Men | 10.3 (29/282) | 7.0 – 14.4 |
| Women | 13.3 (19/143) | 8.2 – 20.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the age groups of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Age (years) | ||
| 20- <30 | 92.9 (39/42) | 80.5 – 98.5 |
| 30-<45 | 92.9 (184/198) | 88.4 – 96.1 |
| 45-<60 | 93.6 (117/125) | 87.8 – 97.2 |
| 60-90 | 93.6 (58/62) | 80.3 – 98.2 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Age (years) | ||
| 20- <30 | 73.8 (31/42) | 51.0 – 86.1 |
| 30-<45 | 71.2 (141/198) | 64.4 – 77.4 |
| 45-<60 | 68.8 (86/125) | 59.9 – 76.8 |
| 60-90 | 72.6 (45/62) | 59.8 – 83.2 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Age (years) | ||
| 20- <30 | 11.9 (5/42) | 4.0 – 25.6 |
| 30-<45 | 11.6 (23/198) | 7.5 – 16.9 |
| 45-<60 | 11.2 (14/125) | 6.2 – 18.1 |
| 60-90 | 12.9 (8/62) | 5.7 – 23.9 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Age (years) | ||
| 20- <30 | 40.5 (17/42) | 25.6 – 56.7 |
| 30-<45 | 44.4 (88/198) | 37.4 – 51.7 |
| 45-<60 | 29.8 (37/124) | 22.0 – 38.7 |
| 60-90 | 48.4 (30/62) | 35.5 – 61.4 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Age (years) | ||
| 20- <30 | 7.1 (3/42) | 1.5 – 19.5 |
| 30-<45 | 13.1 (26/198) | 8.8 – 18.7 |
| 45-<60 | 16.9 (21/124) | 10.8 – 24.7 |
| 60-90 | 12.9 (8/62) | 5.7 – 23.9 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Age (years) | ||
| 20- <30 | 16.7 (7/42) | 7.0 – 31.4 |
| 30-<45 | 7.6 (15/198) | 4.3 – 12.2 |
| 45-<60 | 16.9 (21/124) | 10.8 – 24.7 |
| 60-90 | 8.1 (5/62) | 2.7 – 17.8 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the education level of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Never been at school | 93.4 (183/196) | 88.9 – 96.4 |
| Primary school | 92.2 (118/128) | 86.1 – 96.2 |
| Secondary school | 93.9 (93/99) | 87.3 – 97.7 |
| University | 100.0 (4/4) | 39.8 – 100.0 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Never been at school | 66.3 (130/196) | 59.3 – 72.9 |
| Primary school | 78.9 (101/128) | 70.8 – 85.6 |
| Secondary school | 69.7 (69/99) | 59.7 – 78.5 |
| University | 75.0 (3/4) | 19.4 – 99.4 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Never been at school | 44.9 (88/196) | 37.8 – 52.2 |
| Primary school | 43.0 (55/128) | 34.3 – 52.0 |
| Secondary school | 27.6 (27/98) | 19.0 – 37.5 |
| University | 50.0 (2/4) | 6.8 – 93.2 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Never been at school | 13.3 (26/196) | 8.9 – 18.8 |
| Primary school | 12.5 (16/128) | 7.3 – 19.5 |
| Secondary school | 16.3 (16/98) | 9.6 – 25.2 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Never been at school | 11.7 (23/196) | 7.6 – 17.1 |
| Primary school | 10.9 (14/128) | 6.1 – 17.7 |
| Secondary school | 11.2 (11/98) | 5.7 – 19.2 |
| University | 0.0 (0/4) | 0.0 – 60.2 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children by profession of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Artisan | 83.3 (25/30) | 65.3 – 94.4 |
| Farmer | 94.5 (258/273) | 91.1 – 96.9 |
| Housewife | 94.2 (49/52) | 84.1 – 98.8 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 100.0 (11/11) | 71.5 – 100.0 |
| Other | 94.1 (16/17) | 71.3 – 99.9 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Artisan | 73.3 (22/30) | 54.1 – 87.7 |
| Farmer | 67.0 (183/273) | 61.1 – 72.6 |
| Housewife | 73.1 (38/52) | 59.0 – 84.4 |
| Seller | 88.6 (39/44) | 75.4 – 96.2 |
| Teacher | 81.8 (9/11) | 48.2 – 97.7 |
| Other | 70.6 (12/17) | 44.0 – 89.7 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Artisan | 3.3 (1/30) | 0.1 – 17.2 |
| Farmer | 14.3 (39/273) | 10.4 – 19.0 |
| Housewife | 13.5 (7/52) | 5.6 – 25.8 |
| Seller | 0.0 (0/44) | 0.0 – 8.0 |
| Teacher | 9.1 (1/11) | 0.2 – 41.3 |
| Other | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Artisan | 46.7 (14/30) | 28.3 – 65.7 |
| Farmer | 41.0 (112/273) | 35.1 – 47.1 |
| Housewife | 36.5 (19/52) | 23.6 – 51.0 |
| Seller | 45.5 (20/44) | 30.4 – 61.2 |
| Teacher | 50.0 (5/10) | 18.7 – 81.3 |
| Other | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Artisan | 16.7 (5/30) | 5.6 – 34.7 |
| Farmer | 11.0 (30/273) | 7.5 – 15.3 |
| Housewife | 17.3 (9/52) | 8.2 – 30.3 |
| Seller | 13.6 (6/44) | 5.2 – 27.4 |
| Teacher | 30.0 (3/10) | 6.7 – 65.3 |
| Other | 29.4 (5/17) | 10.3 – 56.0 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Artisan | 13.3 (4/30) | 3.8 – 30.7 |
| Farmer | 8.4 (23/273) | 5.4 – 12.4 |
| Housewife | 21.2 (11/52) | 11.1 – 34.7 |
| Seller | 13.6 (6/44) | 5.2 – 27.4 |
| Teacher | 10.0 (1/10) | 0.3 – 44.5 |
| Other | 17.7 (3/17) | 3.8 – 43.4 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children by the residence zone of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Rural | 97.3 (329/338) | 95.0 – 98.6 |
| Semi urban | 77.5 (69/89) | 67.5 – 85.7 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Rural | 65.1 (220/338) | 59.9 – 70.0 |
| Semi urban | 92.3 (83/89) | 85.9 – 97.5 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Rural | 14.2 (48/338) | 10.9 – 18.3 |
| Semi urban | 2.3 (2/89) | 0.3 – 7.9 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Rural | 39.2 (132/337) | 34.1 – 44.5 |
| Semi urban | 44.9 (40/89) | 34.4 – 55.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Rural | 13.1 (44/337) | 9.9 – 17.1 |
| Semi urban | 15.7 (14/89) | 8.9 – 25.0 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Rural | 11.0 (37/337) | 8.1 – 14.8 |
| Semi urban | 12.4 (11/89) | 6.3 – 21.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Attitudes on hematuria in school aged children according to the religion of the heads of household | ||
| Was at home during the last Mass Drug Administration | ||
| Christian | 93.5 (186/199) | 89.1 – 96.5 |
| Muslim | 85.7 (30/35) | 69.7 – 95.2 |
| Traditional (Animism) | 94.3 (182/193) | 90.0 – 97.1 |
| Overall | 93.2 (398/427) | 90.4 – 95.2 |
| Will take the child to the hospital in case he gets hematuria | ||
| Christian | 72.4 (144/199) | 65.6 – 78.5 |
| Muslim | 77.1 (27/35) | 59.9 – 89.6 |
| Traditional (Animism) | 68.4 (132/193) | 61.3 – 74.9 |
| Overall | 71.0 (303/427) | 66.5 – 75.1 |
| Will take the child to the Community Health Worker in case he gets hematuria | ||
| Christian | 14.1 (28/199) | 9.7 – 19.7 |
| Muslim | 2.9 (1/35) | 0.1 – 14.9 |
| Traditional (Animism) | 10.9 (21/193) | 6.9 – 16.2 |
| Overall | 11.7 (50/427) | 9.0 – 15.1 |
| Interdiction of swimming in ponds and rivers to avoid the occurrence of hematuria in a household member | ||
| Christian | 39.7 (79/199) | 32.9 – 46.9 |
| Muslim | 48.6 (17/35) | 31.4 – 66.0 |
| Traditional (Animism) | 39.6 (76/192) | 32.6 – 46.9 |
| Overall | 40.4 (172/426) | 35.8 – 45.1 |
| Oversees medication intake during Mass Drug Distribution to prevent the occurrence of hematuria in a household member | ||
| Christian | 14.1 (28/199) | 9.6 – 19.7 |
| Muslim | 22.9 (8/35) | 10.4 – 40.1 |
| Traditional (Animism) | 11.5 (22/192) | 7.3 – 16.8 |
| Overall | 13.6 (58/426) | 10.7 – 17.2 |
| No measures in place in the household to prevent the occurrence of hematuria | ||
| Christian | 9.6 (19/199) | 5.9 – 14.5 |
| Muslim | 14.3 (5/35) | 4.8 – 30.3 |
| Traditional (Animism) | 12.5 (24/192) | 8.2 – 18.0 |
| Overall | 11.3 (48/426) | 8.6 – 14.6 |
| Variables | Proportion | 95% CI |
|---|---|---|
| Practices on hematuria in school aged children according to the gender of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Men | 31.6 (18/57) | 19.9 – 45.2 |
| Women | 46.7 (14/30) | 28.3 – 65.7 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Men | 24.6 (14/57) | 14.1 – 37.8 |
| Women | 6.7 (2/30) | 0.8 – 22.1 |
| Overall | 18.2 (16/88) | 10.7 – 27.8 |
| Did nothing when a child got hematuria | ||
| Men | 19.3 (11/57) | 10.1 – 31.9 |
| Women | 23.3 (7/30) | 9.9 – 42.3 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Self-medicated the child when he got hematuria | ||
| Men | 15.8 (9/57) | 7.5 – 27.9 |
| Women | 20.0 (6/30) | 7.7 – 38.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river or pond | ||
| Men | 48.5 (37/130) | 39.6 – 57.4 |
| Women | 45.8 (33/72) | 34.0 – 58.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished the children when they were seen swimming in rivers or ponds | ||
| Men | 19.2 (25/130) | 12.9 – 27.1 |
| Women | 19.4 (14/72) | 11.1 – 30.5 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Practices on hematuria in school aged children by age groups of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 20.0 (1/5) | 0.5 – 71.6 |
| 30-<45 | 36.6 (15/41) | 22.1 – 53.1 |
| 45-<60 | 25.9 (7/27) | 11.1 – 46.3 |
| 60-90 | 60.0 (9/15) | 32.3 – 83.7 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 40.0 (2/5) | 5.3 – 85.3 |
| 30-<45 | 19.5 (8/41) | 8.8 – 34.9 |
| 45-<60 | 14.8 (4/27) | 4.2 – 33.7 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Age (years) | ||
| 20- <30 | 0.0 (0/5) | 0.0 – 52.2 |
| 30-<45 | 14.6 (6/41) | 5.6 – 29.2 |
| 45-<60 | 29.6 (8/27) | 13.8 – 50.2 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river or pond | ||
| Age (years) | ||
| 20- <30 | 45.0 (9/20) | 23.1 – 68.5 |
| 30-<45 | 44.7 (38/85) | 33.9 – 55.9 |
| 45-<60 | 52.2 (36/69) | 39.8 – 64.4 |
| 60-90 | 44.8 (13/29) | 26.5 – 64.3 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Age (years) | ||
| 20- <30 | 15.0 (3/20) | 3.2 – 37.9 |
| 30-<45 | 17.7 (15/85) | 10.2 – 27.4 |
| 45-<60 | 14.5 (10/69) | 7.2 – 25.0 |
| 60-90 | 37.9 (11/29) | 20.7 – 57.7 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Age (years) | ||
| 20- <30 | 40.0 (2/5) | 5.3 – 85.3 |
| 30-<45 | 22.0 (9/41) | 10.6 – 37.6 |
| 45-<60 | 18.5 (5/27) | 6.3 – 38.1 |
| 60-90 | 13.3 (2/15) | 1.7 – 40.5 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the education level of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Never been at school | 41.6 (22/53) | 28.1 – 55.9 |
| Primary school | 29.6 (8/27) | 13.8 – 50.2 |
| Secondary school | 28.6 (2/7) | 3.7 – 71.0 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Never been at school | 15.1 (8/53) | 6.8 – 27.6 |
| Primary school | 11.1 (3/27) | 2.4 – 29.2 |
| Secondary school | 57.1 (4/7) | 18.4 – 90.1 |
| University | 100.0 (1/1) | 2.5 – 100.0 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Never been at school | 15.1 (8/53) | 6.8 – 27.6 |
| Primary school | 29.6 (8/27) | 15.8 – 50.2 |
| Secondary school | 0.0 (0/7) | 0.0 – 41.0 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Never been at school | 50.0 (48/96) | 39.6 – 60.4 |
| Primary school | 45.3 (29/64) | 32.8 – 58.3 |
| Secondary school | 42.9 (18/42) | 27.7 – 59.0 |
| University | 100.0 (1/1) | 2.5 – 100.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Never been at school | 21.9 (21/96) | 14.1 – 31.4 |
| Primary school | 17.2 (11/64) | 8.9 – 28.7 |
| Secondary school | 16.7 (7/42) | 7.0 – 31.4 |
| University | 0.0 (0/1) | 0.0 – 97.5 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Never been at school | 17.0 (5/53) | 8.07 – 29.8 |
| Primary school | 29.6 (8/27) | 13.75 – 50.2 |
| Secondary school | 14.3 (1/7) | 0.36 – 57.9 |
| University | 0.0 (0/1) | 0.00 – 97.5 |
| Overall | 20.5 (18/88) | 12.60 – 30.4 |
| Practices on hematuria in school aged children according to the profession of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 39.3 (22/56) | 26.5 – 53.3 |
| Housewife | 33.3 (3/9) | 7.5 – 70.0 |
| Seller | 41.7 (5/12) | 15.2 – 72.3 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 21.4 (12/56) | 11.6 – 34.4 |
| Housewife | 0.0 (0/9) | 0.0 – 33.6 |
| Seller | 16.7 (2/12) | 2.1 – 48.4 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Artisan | 57.1 (4/7) | 18.4 – 90.1 |
| Farmer | 10.7 (6/56) | 4.0 – 21.9 |
| Housewife | 11.1 (1/9) | 0.3 – 48.3 |
| Seller | 33.3 (4/12) | 9.9 – 65.1 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Artisan | 28.9 (4/14) | 8.4 – 58.1 |
| Farmer | 50.4 (69/137) | 41.7 – 59.0 |
| Housewife | 50.0 (13/26) | 29.9 – 70.1 |
| Seller | 31.6 (6/19) | 12.6 – 56.6 |
| Teacher | 50.0 (1/2) | 1.3 – 98.7 |
| Other | 60.0 (3/5) | 14.7 – 94.7 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Artisan | 7.1 (1/14) | 0.2 – 33.9 |
| Farmer | 18.2 (25/137) | 12.2 – 25.8 |
| Housewife | 19.2 (5/26) | 6.6 – 39.4 |
| Seller | 36.8 (7/19) | 16.3 – 61.6 |
| Teacher | 0.0 (0/2) | 0.0 – 84.2 |
| Other | 20.0 (1/5) | 0.5 – 71.6 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Artisan | 14.3 (1/7) | 0.4 – 57.9 |
| Farmer | 19.6 (11/56) | 10.2 – 32.4 |
| Housewife | 44.4 (4/9) | 13.7 – 78.8 |
| Seller | 8.3 (1/12) | 0.2 – 38.5 |
| Other | 25.0 (1/4) | 0.6 – 80.6 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the zone of residence of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Rural | 38.0 (27/71) | 26.8 – 50.3 |
| Semi urban | 29.4 (5/17) | 10.3 – 56.0 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Rural | 19.7 (14/71) | 11.2 – 30.9 |
| Semi urban | 11.8 (2/17) | 1.5 – 36.4 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Rural | 14.1 (10/71) | 7.0 – 24.4 |
| Semi urban | 35.3 (6/17) | 14.2 – 61.7 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Rural | 49.4 (87/176) | 41.8 – 57.1 |
| Semi urban | 33.3 (9/27) | 16.5 – 54.0 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Did nothing when a child got hematuria | ||
| Rural | 19.7 (14/71) | 11.2 – 30.9 |
| Semi urban | 23.5 (14/17) | 6.8 – 49.9 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |
| Practices on hematuria in school aged children according to the religion of the heads of household | ||
| Took the child to the hospital when he got hematuria | ||
| Christian | 20.0 (8/40) | 9.1 – 35.7 |
| Muslim | 27.3 (3/11) | 6.0 – 61.0 |
| Traditional (Animism) | 56.8 (21/37) | 39.5 – 72.9 |
| Overall | 36.4 (32/88) | 26.4 – 47.3 |
| Took the child to the Community Health Worker when he got hematuria | ||
| Christian | 25.0 (10/40) | 12.7 – 41.2 |
| Muslim | 9.1 (1/11) | 0.2 – 41.3 |
| Traditional (Animism) | 13.5 (5/37) | 4.5 – 28.8 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Self-medicated the child when he got hematuria | ||
| Christian | 22.5 (9/40) | 10.8 – 38.5 |
| Muslim | 36.4 (4/11) | 10.9 – 69.2 |
| Traditional (Animism) | 8.1 (3/37) | 1.7 – 21.9 |
| Overall | 18.2 (16/88) | 10.8 – 27.8 |
| Advised the child when he saw him swimming in a river | ||
| Christian | 51.6 (50/97) | 41.2 – 61.8 |
| Muslim | 33.3 (4/12) | 9.9 – 65.1 |
| Traditional (Animism) | 44.7 (42/94) | 34.4 – 55.3 |
| Overall | 47.3 (96/203) | 40.3 – 54.4 |
| Punished their children when they were seen swimming in rivers and ponds | ||
| Christian | 14.4 (14/97) | 8.1 – 23.0 |
| Muslim | 50.0 (6/12) | 21.1 – 78.9 |
| Traditional (Animism) | 20.2 (19/94) | 12.6 – 29.8 |
| Overall | 19.2 (39/203) | 14.0 – 25.3 |
| Did nothing when a child got hematuria | ||
| Christian | 27.5 (11/40) | 14.6 – 41.9 |
| Muslim | 27.3 (3/11) | 6.0 – 61.0 |
| Traditional (Animism) | 10.8 (4/37) | 3.0 – 25.4 |
| Overall | 20.5 (18/88) | 12.6 – 30.4 |


References
- Aubry P, Gaüzère AB. [Schistosomiasis or bilharzia: News 2023]. Bordeaux (France): Medecine Tropicale; 2001. 10 p. French.
- Kokaliaris C, Garba A, Matuska M, Bronzan RN, Colley DG, Dorkenoo AM, Ekpo UF, Fleming FM, French MD, Kabore A, Mbonigaba JB, Midzi N, Mwinzi PNM, N’Goran EK, Polo MR, Sacko M, Tchuem Tchuenté LA, Tukahebwa EM, Uvon PA, Yang G, Wiesner L, Zhang Y, Utzinger J, Vounatsou P. Effect of preventive chemotherapy with praziquantel on schistosomiasis among school-aged children in sub-Saharan Africa: a spatiotemporal modelling study. The Lancet Infectious Diseases [Internet]. 2021 Dec 2 [cited 2026 Mar 11];22(1):136–49. doi: 10.1016/S1473-3099(21)00090-6 Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(21)00090-6/fulltext Erratum in: Correction to Lancet Infect Dis 2022; 22: 136–49. The Lancet Infectious Diseases [Internet]. 2022 Jan [cited 2026 Mar 11];22(1):e1. doi: 10.1016/S1473-3099(21)00771-4 Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(21)00771-4/fulltext
- Knowles SCL, Webster BL, Garba A, Sacko M, Diaw OT, Fenwick A, Rollinson D, Webster JP. Epidemiological Interactions between Urogenital and Intestinal Human Schistosomiasis in the Context of Praziquantel Treatment across Three West African Countries. Raso G, editor. PLoS Negl Trop Dis [Internet]. 2015 Oct 15 [cited 2026 Mar 11];9(10):e0004019. doi: 10.1371/journal.pntd.0004019 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0004019
- Adenowo AF, Oyinloye BE, Ogunyinka BI, Kappo AP. Impact of human schistosomiasis in sub-Saharan Africa. The Brazilian Journal of Infectious Diseases [Internet]. 2015 Jan 27 [cited 2026 Mar 11];19(2):196–205. doi: 10.1016/j.bjid.2014.11.004 Available from: https://www.sciencedirect.com/science/article/pii/S1413867015000264?via%3Dihub
- Torres-Vitolas CA, Dhanani N, Fleming FM. Factors affecting the uptake of preventive chemotherapy treatment for schistosomiasis in Sub-Saharan Africa: A systematic review. LoVerde PT, editor. PLoS Negl Trop Dis [Internet]. 2021 Jan 19 [cited 2026 Mar 11];15(1):e0009017. doi: 10.1371/journal.pntd.0009017 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0009017
- Murray CJL, Vos T, Lozano R, Naghavi M, Flaxman AD, Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V, Abraham J, Ackerman I, Aggarwal R, Ahn SY, Ali MK, AlMazroa MA, Alvarado M, Anderson HR, Anderson LM, Andrews KG, Atkinson C, Baddour LM, Bahalim AN, Barker-Collo S, Barrero LH, Bartels DH, Basáñez MG, Baxter A, Bell ML, Benjamin EJ, Bennett D, Bernabé E, Bhalla K, Bhandari B, Bikbov B, Abdulhak AB, Birbeck G, Black JA, Blencowe H, Blore JD, Blyth F, Bolliger I, Bonaventure A, Boufous S, Bourne R, Boussinesq M, Braithwaite T, Brayne C, Bridgett L, Brooker S, Brooks P, Brugha TS, Bryan-Hancock C, Bucello C, Buchbinder R, Buckle G, Budke CM, Burch M, Burney P, Burstein R, Calabria B, Campbell B, Canter CE, Carabin H, Carapetis J, Carmona L, Cella C, Charlson F, Chen H, Cheng ATA, Chou D, Chugh SS, Coffeng LE, Colan SD, Colquhoun S, Colson KE, Condon J, Connor MD, Cooper LT, Corriere M, Cortinovis M, De Vaccaro KC, Couser W, Cowie BC, Criqui MH, Cross M, Dabhadkar KC, Dahiya M, Dahodwala N, Damsere-Derry J, Danaei G, Davis A, Leo DD, Degenhardt L, Dellavalle R, Delossantos A, Denenberg J, Derrett S, Des Jarlais DC, Dharmaratne SD, Dherani M, Diaz-Torne C, Dolk H, Dorsey ER, Driscoll T, Duber H, Ebel B, Edmond K, Elbaz A, Ali SE, Erskine H, Erwin PJ, Espindola P, Ewoigbokhan SE, Farzadfar F, Feigin V, Felson DT, Ferrari A, Ferri CP, Fèvre EM, Finucane MM, Flaxman S, Flood L, Foreman K, Forouzanfar MH, Fowkes FGR, Fransen M, Freeman MK, Gabbe BJ, Gabriel SE, Gakidou E, Ganatra HA, Garcia B, Gaspari F, Gillum RF, Gmel G, Gonzalez-Medina D, Gosselin R, Grainger R, Grant B, Groeger J, Guillemin F, Gunnell D, Gupta R, Haagsma J, Hagan H, Halasa YA, Hall W, Haring D, Haro JM, Harrison JE, Havmoeller R, Hay RJ, Higashi H, Hill C, Hoen B, Hoffman H, Hotez PJ, Hoy D, Huang JJ, Ibeanusi SE, Jacobsen KH, James SL, Jarvis D, Jasrasaria R, Jayaraman S, Johns N, Jonas JB, Karthikeyan G, Kassebaum N, Kawakami N, Keren A, Khoo JP, King CH, Knowlton LM, Kobusingye O, Koranteng A, Krishnamurthi R, Laden F, Lalloo R, Laslett LL, Lathlean T, Leasher JL, Lee YY, Leigh J, Levinson D, Lim SS, Limb E, Lin JK, Lipnick M, Lipshultz SE, Liu W, Loane M, Ohno SL, Lyons R, Mabweijano J, MacIntyre MF, Malekzadeh R, Mallinger L, Manivannan S, Marcenes W, March L, Margolis DJ, Marks GB, Marks R, Matsumori A, Matzopoulos R, Mayosi BM, McAnulty JH, McDermott MM, McGill N, McGrath J, Medina-Mora ME, Meltzer M, Memish ZA, Mensah GA, Merriman TR, Meyer AC, Miglioli V, Miller M, Miller TR, Mitchell PB, Mock C, Mocumbi AO, Moffitt TE, Mokdad AA, Monasta L, Montico M, Moradi-Lakeh M, Moran A, Morawska L, Mori R, Murdoch ME, Mwaniki MK, Naidoo K, Nair MN, Naldi L, Narayan KMV, Nelson PK, Nelson RG, Nevitt MC, Newton CR, Nolte S, Norman P, Norman R, O’Donnell M, O’Hanlon S, Olives C, Omer SB, Ortblad K, Osborne R, Ozgediz D, Page A, Pahari B, Pandian JD, Rivero AP, Patten SB, Pearce N, Padilla RP, Perez-Ruiz F, Perico N, Pesudovs K, Phillips D, Phillips MR, Pierce K, Pion S, Polanczyk GV, Polinder S, Pope CA, Popova S, Porrini E, Pourmalek F, Prince M, Pullan RL, Ramaiah KD, Ranganathan D, Razavi H, Regan M, Rehm JT, Rein DB, Remuzzi G, Richardson K, Rivara FP, Roberts T, Robinson C, De Leòn FR, Ronfani L, Room R, Rosenfeld LC, Rushton L, Sacco RL, Saha S, Sampson U, Sanchez-Riera L, Sanman E, Schwebel DC, Scott JG, Segui-Gomez M, Shahraz S, Shepard DS, Shin H, Shivakoti R, Silberberg D, Singh D, Singh GM, Singh JA, Singleton J, Sleet DA, Sliwa K, Smith E, Smith JL, Stapelberg NJ, Steer A, Steiner T, Stolk WA, Stovner LJ, Sudfeld C, Syed S, Tamburlini G, Tavakkoli M, Taylor HR, Taylor JA, Taylor WJ, Thomas B, Thomson WM, Thurston GD, Tleyjeh IM, Tonelli M, Towbin JA, Truelsen T, Tsilimbaris MK, Ubeda C, Undurraga EA, Van Der Werf MJ, Van Os J, Vavilala MS, Venketasubramanian N, Wang M, Wang W, Watt K, Weatherall DJ, Weinstock MA, Weintraub R, Weisskopf MG, Weissman MM, White RA, Whiteford H, Wiebe N, Wiersma ST, Wilkinson JD, Williams HC, Williams SR, Witt E, Wolfe F, Woolf AD, Wulf S, Yeh PH, Zaidi AK, Zheng ZJ, Zonies D, Lopez AD. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. The Lancet [Internet]. 2012 Dec 14 [cited 2026 Mar 11];380(9859):2197–223. doi: 10.1016/S0140-6736(12)61689-4 Available from: https://www.sciencedirect.com/science/article/abs/pii/S0140673612616894 Erratum in: Department of Error. The Lancet [Internet]. 2013 Feb 22 [cited 2026 Mar 11];381(9867):628. doi: 10.1016/S0140-6736(13)60352-9 Available from: https://www.sciencedirect.com/science/article/pii/S0140673613603529; Department of Error. The Lancet [Internet]. 2014 Aug 14 [cited 2026 Mar 11];384(9943):582. doi: 10.1016/S0140-6736(14)61363-5 Available from: https://www.sciencedirect.com/science/article/pii/S0140673614613635
- Kassebaum NJ, Arora M, Barber RM, Bhutta ZA, Brown J, Carter A, Casey DC, Charlson FJ, Coates MM, Coggeshall M, Cornaby L, Dandona L, Dicker DJ, Erskine HE, Ferrari AJ, Fitzmaurice C, Foreman K, Forouzanfar MH, Fullman N, Gething PW, Goldberg EM, Graetz N, Haagsma JA, Hay SI, Johnson CO, Kemmer L, Khalil IA, Kinfu Y, Kutz MJ, Kyu HH, Leung J, Liang X, Lim SS, Lozano R, Mensah GA, Mikesell J, Mokdad AH, Mooney MD, Naghavi M, Nguyen G, Nsoesie E, Pigott DM, Pinho C, Rankin Z, Reinig N, Salomon JA, Sandar L, Smith A, Sorensen RJD, Stanaway J, Steiner C, Teeple S, Troeger C, Truelsen T, VanderZanden A, Wagner JA, Wanga V, Whiteford HA, Zhou M, Zoeckler L, Abajobir AA, Abate KH, Abbafati C, Abbas KM, Abd-Allah F, Abraham B, Abubakar I, Abu-Raddad LJ, Abu-Rmeileh NME, Achoki T, Ackerman IN, Adebiyi AO, Adedeji IA, Adsuar JC, Afanvi KA, Afshin A, Agardh EE, Agarwal A, Agarwal SK, Ahmed MB, Kiadaliri AA, Ahmadieh H, Akseer N, Al-Aly Z, Alam K, Alam NKM, Aldhahri SF, Alegretti MA, Aleman AV, Alemu ZA, Alexander LT, Ali R, Alkerwi A, Alla F, Allebeck P, Allen C, Alsharif U, Altirkawi KA, Martin EA, Alvis-Guzman N, Amare AT, Amberbir A, Amegah AK, Amini H, Ammar W, Amrock SM, Anderson GM, Anderson BO, Antonio CAT, Anwari P, Ärnlöv J, Arsenijevic VSA, Artaman A, Asayesh H, Asghar RJ, Avokpaho EFGA, Awasthi A, Quintanilla BPA, Azzopardi P, Bacha U, Badawi A, Balakrishnan K, Banerjee A, Barac A, Barker-Collo SL, Bärnighausen T, Barregard L, Barrero LH, Basu S, Bayou TA, Beardsley J, Bedi N, Beghi E, Bell B, Bell ML, Benjet C, Bennett DA, Bensenor IM, Berhane A, Bernabé E, Betsu BD, Beyene AS, Bhala N, Bhansali A, Bhatt S, Biadgilign S, Bienhoff K, Bikbov B, Abdulhak AAB, Biryukov S, Bisanzio D, Bjertness E, Blore JD, Borschmann R, Boufous S, Bourne RRA, Brainin M, Brazinova A, Breitborde NJK, Brenner H, Briant PS, Briko AN, Britton G, Brugha TS, Buchbinder R, Buckle GC, Butt ZA, Cahuana-Hurtado L, Campuzano JC, Cano J, Cárdenas R, Carrero JJ, Carter A, Carvalho F, Castañeda-Orjuela CA, Rivas JC, Catalá-López F, Cavalleri F, Chang JC, Chiang PPC, Chibalabala M, Chibueze CE, Chisumpa VH, Choi JYJ, Choudhury L, Christensen H, Ciobanu LG, Colistro V, Colomar M, Colquhoun SM, Cortinovis M, Crump JA, Damasceno A, Dandona R, Dandona L, Dargan PI, Das Neves J, Davey G, Davis AC, Leo DD, Degenhardt L, Gobbo LCD, Derrett S, Jarlais DCD, deVeber GA, Dharmaratne SD, Dhillon PK, Ding EL, Doyle KE, Driscoll TR, Duan L, Dubey M, Duncan BB, Ebrahimi H, Ellenbogen RG, Elyazar I, Endries AY, Ermakov SP, Eshrati B, Esteghamati A, Estep K, Fahimi S, Farid TA, Farinha CSES, Faro A, Farvid MS, Farzadfar F, Feigin VL, Fereshtehnejad SM, Fernandes JG, Fernandes JC, Fischer F, Fitchett JRA, Foigt N, Fowkes FGR, Franklin RC, Friedman J, Frostad J, Fürst T, Futran ND, Gabbe B, Gankpé FG, Garcia-Basteiro AL, Gebrehiwot TT, Gebremedhin AT, Geleijnse JM, Gibney KB, Gillum RF, Ginawi IAM, Giref AZ, Giroud M, Gishu MD, Giussani G, Godwin WW, Gomez-Dantes H, Gona P, Goodridge A, Gopalani SV, Gotay CC, Goto A, Gouda HN, Gugnani H, Guo Y, Gupta R, Gupta R, Gupta V, Gutiérrez RA, Hafezi-Nejad N, Haile D, Hailu AD, Hailu GB, Halasa YA, Hamadeh RR, Hamidi S, Hammami M, Handal AJ, Hankey GJ, Harb HL, Harikrishnan S, Haro JM, Hassanvand MS, Hassen TA, Havmoeller R, Hay RJ, Hedayati MT, Heredia-Pi IB, Heydarpour P, Hoek HW, Hoffman DJ, Horino M, Horita N, Hosgood HD, Hoy DG, Hsairi M, Huang H, Huang JJ, Iburg KM, Idrisov BT, Innos K, Inoue M, Jacobsen KH, Jauregui A, Jayatilleke AU, Jeemon P, Jha V, Jiang G, Jiang Y, Jibat T, Jimenez-Corona A, Jin Y, Jonas JB, Kabir Z, Kajungu DK, Kalkonde Y, Kamal R, Kan H, Kandel A, Karch A, Karema CK, Karimkhani C, Kasaeian A, Katibeh M, Kaul A, Kawakami N, Kazi DS, Keiyoro PN, Kemp AH, Kengne AP, Keren A, Kesavachandran CN, Khader YS, Khan AR, Khan EA, Khang YH, Khoja TAM, Khubchandani J, Kieling C, Kim C il, Kim D, Kim YJ, Kissoon N, Kivipelto M, Knibbs LD, Knudsen AK, Kokubo Y, Kolte D, Kopec JA, Koul PA, Koyanagi A, Defo BK, Kuchenbecker RS, Bicer BK, Kuipers EJ, Kumar GA, Kwan GF, Lalloo R, Lallukka T, Larsson A, Latif AA, Lavados PM, Lawrynowicz AEB, Leasher JL, Leigh J, Leung R, Li Y, Li Y, Lipshultz SE, Liu PY, Liu Y, Lloyd BK, Logroscino G, Looker KJ, Lotufo PA, Lucas RM, Lunevicius R, Lyons RA, Razek HMAE, Mahdavi M, Majdan M, Majeed A, Malekzadeh R, Malta DC, Marcenes W, Martinez-Raga J, Masiye F, Mason-Jones AJ, Matzopoulos R, Mayosi BM, McGrath JJ, McKee M, Meaney PA, Mehari A, Melaku YA, Memiah P, Memish ZA, Mendoza W, Meretoja A, Meretoja TJ, Mesfin YM, Mhimbira FA, Millear A, Miller TR, Mills EJ, Mirarefin M, Mirrakhimov EM, Mitchell PB, Mock CN, Mohammad KA, Mohammadi A, Mohammed S, Monasta L, Hernandez JCM, Montico M, Moradi-Lakeh M, Mori R, Mueller UO, Mumford JE, Murdoch ME, Murthy GVS, Nachega JB, Naheed A, Naldi L, Nangia V, Newton CR, Ng M, Ngalesoni FN, Nguyen QL, Nisar MI, Pete PMN, Nolla JM, Norheim OF, Norman RE, Norrving B, Obermeyer CM, Ogbo FA, Oh IH, Oladimeji O, Olivares PR, Olusanya BO, Olusanya JO, Oren E, Ortiz A, Ota E, Oyekale AS, Pa M, Park EK, Parsaeian M, Patten SB, Patton GC, Pedro JM, Pereira DM, Perico N, Pesudovs K, Petzold M, Phillips MR, Piel FB, Pillay JD, Pishgar F, Plass D, Polinder S, Popova S, Poulton RG, Pourmalek F, Prasad NM, Qorbani M, Rabiee RHS, Radfar A, Rafay A, Rahimi K, Rahimi-Movaghar V, Rahman M, Rahman MHU, Rahman SU, Rai D, Rai RK, Rajsic S, Raju M, Ram U, Ranganathan K, Refaat AH, Reitsma MB, Remuzzi G, Resnikoff S, Reynolds A, Ribeiro AL, Ricci S, Roba HS, Rojas-Rueda D, Ronfani L, Roshandel G, Roth GA, Roy A, Sackey BB, Sagar R, Sanabria JR, Sanchez-Niño MD, Santos IS, Santos JV, Sarmiento-Suarez R, Sartorius B, Satpathy M, Savic M, Sawhney M, Schmidt MI, Schneider IJC, Schutte AE, Schwebel DC, Seedat S, Sepanlou SG, Servan-Mori EE, Shahraz S, Shaikh MA, Sharma R, She J, Sheikhbahaei S, Shen J, Sheth KN, Shibuya K, Shigematsu M, Shin MJ, Shiri R, Sigfusdottir ID, Silva DAS, Silverberg JI, Simard EP, Singh A, Singh JA, Singh PK, Skirbekk V, Skogen JC, Soljak M, Søreide K, Sorensen RJD, Sreeramareddy CT, Stathopoulou V, Steel N, Stein DJ, Stein MB, Steiner TJ, Stovner LJ, Stranges S, Stroumpoulis K, Sunguya BF, Sur PJ, Swaminathan S, Sykes BL, Szoeke CEI, Tabarés-Seisdedos R, Tandon N, Tanne D, Tavakkoli M, Taye B, Taylor HR, Ao BJT, Tegegne TK, Tekle DY, Terkawi AS, Tessema GA, Thakur JS, Thomson AJ, Thorne-Lyman AL, Thrift AG, Thurston GD, Tobe-Gai R, Tonelli M, Topor-Madry R, Topouzis F, Tran BX, Tran KB, Troeger C, Tsadik AG, Tsoi D, Tudor Car L, Tyrovolas S, Ukwaja KN, Ullah I, Undurraga EA, Updike RL, Usman MS, Uthman OA, Vaduganathan M, Vaezi A, Valdez PR, Varavikova E, Varughese S, Vasankari TJ, Vasconcelos AMN, Venketasubramanian N, Villafaina S, Violante FS, Vladimirov SK, Vlassov V, Vollset SE, Vos T, Vosoughi K, Vujcic IS, Wagnew FS, Waheed Y, Wang Y, Wang YP, Weiderpass E, Weintraub RG, Weiss DJ, Weldegebreal F, Weldegwergs KG, Werdecker A, West TE, Westerman R, Whiteford HA, Widecka J, Wijeratne T, Williams HC, Wilner LB, Wilson S, Winkler AS, Wiyeh AB, Wiysonge CS, Wolfe CDA, Woolf AD, Wyper GMA, Xavier D, Xu G, Yadgir S, Yahyazadeh Jabbari SH, Yamada T, Yan LL, Yano Y, Yaseri M, Yasin YJ, Yeshaneh A, Yimer EM, Yip P, Yisma E, Yonemoto N, Yoon SJ, Yotebieng M, Younis MZ, Yousefifard M, Yu C, Zadnik V, Zaidi Z, Zaman SB, Zamani M, Zandian H, Zar HJ, Zenebe ZM, Zhou M, Zipkin B, Zodpey S, Zucker I, Zuhlke LJ, Murray CJL. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet [Internet]. 2018 Nov 10 [cited 2026 Mar 11];392(10159):1859–922. doi: 10.1016/S0140-6736(18)32335-3 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(18)32335-3/fulltext Erratum in: Department of Error. The Lancet [Internet]. 2019 Jun 22 [cited 2026 Mar 11];393(10190):e44. doi: 10.1016/S0140-6736(19)31043-8 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)31043-8/fulltext
- Kyu HH, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, Abbastabar H, Abd-Allah F, Abdela J, Abdelalim A, Abdollahpour I, Abdulkader RS, Abebe M, Abebe Z, Abil OZ, Aboyans V, Abrham AR, Abu-Raddad LJ, Abu-Rmeileh NME, Accrombessi MMK, Acharya D, Acharya P, Ackerman IN, Adamu AA, Adebayo OM, Adekanmbi V, Ademi Z, Adetokunboh OO, Adib MG, Adsuar JC, Afanvi KA, Afarideh M, Afshin A, Agarwal G, Agesa KM, Aggarwal R, Aghayan SA, Agrawal A, Ahmadi A, Ahmadi M, Ahmadieh H, Ahmed MB, Ahmed S, Aichour AN, Aichour I, Aichour MTE, Akinyemiju T, Akseer N, Al-Aly Z, Al-Eyadhy A, Al-Mekhlafi HM, Al-Raddadi RM, Alahdab F, Alam K, Alam T, Alashi A, Alavian SM, Alene KA, Alijanzadeh M, Alizadeh-Navaei R, Aljunid SM, Alkerwi A, Alla F, Allebeck P, Alonso J, Alsharif U, Altirkawi K, Alvis-Guzman N, Aminde LN, Amini E, Amiresmaili M, Ammar W, Amoako YA, Anber NH, Andrei CL, Androudi S, Animut MD, Anjomshoa M, Ansha MG, Antonio CAT, Anwari P, Arabloo J, Aremu O, Ärnlöv J, Arora A, Arora M, Artaman A, Aryal KK, Asayesh H, Ataro Z, Ausloos M, Avila-Burgos L, Avokpaho EFGA, Awasthi A, Ayala Quintanilla BP, Ayer R, Azzopardi PS, Babazadeh A, Badali H, Balakrishnan K, Bali AG, Banach M, Banoub JAM, Barac A, Barboza MA, Barker-Collo SL, Bärnighausen TW, Barquera S, Barrero LH, Bazargan-Hejazi S, Bedi N, Beghi E, Behzadifar M, Behzadifar M, Bekele BB, Bekru ET, Belachew AB, Belay YA, Bell ML, Bello AK, Bennett DA, Bensenor IM, Berhane A, Bernabe E, Bernstein RS, Beuran M, Beyranvand T, Bhala N, Bhatt S, Bhaumik S, Bhutta ZA, Biadgo B, Biehl MH, Bijani A, Bikbov B, Bilano V, Bililign N, Bin Sayeed MS, Bisanzio D, Bjørge T, Bleyer A, Bobasa EM, Bou-Orm IR, Boufous S, Bourne R, Brady OJ, Brant LC, Brayne C, Brazinova A, Breitborde NJK, Brenner H, Briant PS, Briko AN, Britton G, Brugha T, Buchbinder R, Busse R, Butt ZA, Cahuana-Hurtado L, Campuzano Rincon JC, Cano J, Cárdenas R, Carrero JJ, Carter A, Carvalho F, Castañeda-Orjuela CA, Castillo Rivas J, Castro F, Catalá-López F, Cercy KM, Cerin E, Chaiah Y, Chang JC, Charlson FJ, Chattu VK, Chiang PPC, Chitheer A, Choi JYJ, Christensen H, Christopher DJ, Chung SC, Cicuttini FM, Cirillo M, Collado-Mateo D, Cooper C, Cortesi PA, Cortinovis M, Cousin E, Criqui MH, Cromwell EA, Cross M, Crump JA, Daba AK, Dachew BA, Dadi AF, Dandona L, Dandona R, Dargan PI, Daryani A, Das Gupta R, Das Neves J, Dasa TT, Davitoiu DV, De La Hoz FP, De Leo D, De Neve JW, De Steur H, Degefa MG, Degenhardt L, Deiparine S, Demoz GT, Denova-Gutiérrez E, Deribe K, … (truncated in original document) … Vos T, Vosoughi K, Vujcic IS, Wagnew FS, Waheed Y, Wang Y, Wang YP, Weiderpass E, Weintraub RG, Weiss DJ, Weldegebreal F, Weldegwergs KG, Werdecker A, West TE, Westerman R, Whiteford HA, Widecka J, Wijeratne T, Williams HC, Wilner LB, Wilson S, Winkler AS, Wiyeh AB, Wiysonge CS, Wolfe CDA, Woolf AD, Wyper GMA, Xavier D, Xu G, Yadgir S, Yahyazadeh Jabbari SH, Yamada T, Yan LL, Yano Y, Yaseri M, Yasin YJ, Yeshaneh A, Yimer EM, Yip P, Yisma E, Yonemoto N, Yoon SJ, Yotebieng M, Younis MZ, Yousefifard M, Yu C, Zadnik V, Zaidi Z, Zaman SB, Zamani M, Zandian H, Zar HJ, Zenebe ZM, Zhou M, Zipkin B, Zodpey S, Zucker I, Zuhlke LJ, Murray CJL. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet [Internet]. 2018 Nov 10 [cited 2026 Mar 11];392(10159):1859–922. doi: 10.1016/S0140-6736(18)32335-3 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(18)32335-3/fulltext Erratum in: Department of Error. The Lancet [Internet]. 2019 Jun 22 [cited 2026 Mar 11];393(10190):e44. doi: 10.1016/S0140-6736(19)31043-8 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)31043-8/fulltext
- Boisson S, Engels D, Gordon BA, Medlicott KO, Neira MP, Montresor A, Solomon AW, Velleman Y. Water, sanitation and hygiene for accelerating and sustaining progress on neglected tropical diseases: a new Global Strategy 2015–20. Int Health [Internet]. 2016 Mar 3 [cited 2026 Mar 11];8(suppl 1):i19–21. doi: 10.1093/inthealth/ihv073 Available from: https://academic.oup.com/inthealth/article/8/suppl_1/i19/2488386
- Grimes JE, Croll D, Harrison WE, Utzinger J, Freeman MC, Templeton MR. The roles of water, sanitation and hygiene in reducing schistosomiasis: a review. Parasites Vectors [Internet]. 2015 Mar 13 [cited 2026 Mar 11];8(1):156. doi: 10.1186/s13071-015-0766-9 Available from: https://link.springer.com/article/10.1186/s13071-015-0766-9
- WHO AFRO. [Strategic Plan for the Fight Against Neglected Tropical Diseases in the African Region 2014-2020] [Internet]. Brazzaville (Republic of Congo): WHO AFRO; 2013 [cited 2026 Mar 11]. 26 p. French. Available from: https://iris.who.int/items/bc580497-7dee-4383-8daa-a13cb0e4d36c Download pdf to view full text.
- Molyneux DH, Savioli L, Engels D. Neglected tropical diseases: progress towards addressing the chronic pandemic. The Lancet [Internet]. 2016 Sep 14 [cited 2026 Mar 11];389(10066):312–25. doi: 10.1016/S0140-6736(16)30171-4 Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(16)30171-4/abstract Register to view full text.
- Global Compact Network France. [The 17 Sustainable Development Goals and their 169 targets] [Internet]. Paris (France): Global Compact Network France; c2022 [cited 2026 Mar 11]. [about 20 pages]. French. Available from: https://pactemondial.org/wp-content/uploads/2022/05/Liste_des_17_ODD_et_169_cibles-1.pdf
- World Health Organization. Ending the Neglect to Attain the Sustainable Development Goals: A Rationale for Continued Investment in Tackling Neglected Tropical Diseases 2021-2030 [Internet]. 1st ed. Geneva (Switzerland): World Health Organization; 2022 [cited 2026 Mar 11]. 35 p. Available from: https://www.who.int/publications/i/item/9789240052932 Download pdf to view full text.
- Ministry of Health (Togo). [National Master Plan for Integrated Control of Neglected Tropical Diseases 2016–2020] [Internet]. Lomé (Togo): Ministry of Health (Togo); 2015 May [cited 2026 Mar 11]. 86 p. French. Available from: https://espen.afro.who.int/sites/default/files/content/document/TOGO_NTD_Master_Plan_2016_2020.pdf
- Ministère de la Santé et de la Protection Sociale, Togo, Division de la Santé Communautaire. [Impact assessment in Togo 2015: Report on the national impact assessment of four to five years of TDM on the prevalence of schistosomiasis and soil-transmitted helminthiasis, coupled with a serological survey on onchocerciasis in Togo]. Lome (Togo): Ministère de la Santé et de la Protection Sociale, Togo, Division de la Santé Communautaire; 2015. 15 p. French.
- Ministère de la Santé et de la Protection Sociale, Togo, Direction Générale des études, de la Planification et de l’Information sanitaire, Division de l’information sanitaire. [Yearbook of Health Statistics of Togo 2017]. Lome (Togo): Ministère de la Santé et de la Protection Sociale; 2018. p.49–115. French.
- Ministère de la Santé et de la Protection Sociale, Togo, Direction Générale des études, de la Planification et de l’Information sanitaire, Division de l’information sanitaire. [Yearbook of health statistics for Togo 2018]. Lome (Togo): Ministère de la Santé et de la Protection Sociale; 2019. p.47–70. French.
- Abdellahi M, Ndir O, Niang S. [Evaluation of the prevalence of schistosomiasis in children aged 5 to 14 years after several years of mass treatment in the Senegal River basin]. Santé Publique [Internet]. 2016 Nov 7 [cited 2026 Mar 11];28(4):535–540. French. Available from: https://www.sfsp.fr/content-page/item/3091-evaluation-de-la-prevalence-des-bilharzioses-aupres-des-enfants-de-5-a-14-ans-apres-plusieurs-annees-de-traitement-de-masse-dans-le-bassin-du-fleuve-senegal Abstract only.
- World Health Organization. WHO guideline on control and elimination of human schistosomiasis [Internet]. Geneva (Switzerland): WHO; 2022 Feb 14 [cited 2026 Mar 11]. 118 p. Available from: https://www.who.int/publications/i/item/9789240041608 Download pdf to view full text.
- Aula OP, McManus DP, Jones MK, Gordon CA. Schistosomiasis with a Focus on Africa. TropicalMed [Internet]. 2021 Jun 22 [cited 2026 Mar 11];6(3):109. doi: 10.3390/tropicalmed6030109 Available from: https://www.mdpi.com/2414-6366/6/3/109
- Bronzan RN, Dorkenoo AM, Agbo YM, Halatoko W, Layibo Y, Adjeloh P, Teko M, Sossou E, Yakpa K, Tchalim M, Datagni G, Seim A, Sognikin KS. Impact of community-based integrated mass drug administration on schistosomiasis and soil-transmitted helminth prevalence in Togo. Steinmann P, editor. PLoS Negl Trop Dis [Internet]. 2018 Aug 20 [cited 2026 Mar 11];12(8):e0006551. doi: 10.1371/journal.pntd.0006551 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0006551
- Tetteh-Quarcoo PB, Forson PO, Amponsah SK, Ahenkorah J, Opintan JA, Ocloo JEY, Okine EN, Aryee R, Afutu E, Anang AK, Ayeh-Kumi PF. Persistent Urogenital Schistosomiasis and Its Associated Morbidity in Endemic Communities within Southern Ghana: Suspected Praziquantel Resistance or Reinfection? Medical Sciences [Internet]. 2020 Feb 10 [cited 2026 Mar 11];8(1):10. doi: 10.3390/medsci8010010 Available from: https://www.mdpi.com/2076-3271/8/1/10
- Angora EK, Boissier J, Menan H, Rey O, Tuo K, Touré AO, Coulibaly JT, Méité A, Raso G, N’Goran EK, Utzinger J, Balmer O. Prevalence and Risk Factors for Schistosomiasis among Schoolchildren in two Settings of Côte d’Ivoire. TropicalMed [Internet]. 2019 Jul 23 [cited 2026 Mar 11];4(3):110. doi: 10.3390/tropicalmed4030110 Available from: https://www.mdpi.com/2414-6366/4/3/110
- Bintou LY, Yaro AS, Sodio B, Sacko M. [Persistence of urinary schistosomiasis in endemic areas subjected to repeated mass treatments in Mali]. International Journal of Biological and Chemical Sciences [Internet]. 2019 May 24 [cited 2026 Mar 11];13(1):369–81. French. doi: 10.4314/ijbcs.v13i1.29 Available from: https://www.ifgdg.org/index.php?option=com_content&view=article&id=2781:persistance-de-la-schistosomiase-urinaire-en-zones-endemiques-soumises-aux-traitements-de-masse-repetes-au-mali&catid=199&Itemid=544 Download pdf to view full text.
- Leta GT, Mekete K, Wuletaw Y, Gebretsadik A, Sime H, Mekasha S, Woyessa A, Shafi O, Vercruysse J, Grimes JET, Gardiner I, French M, Levecke B, Drake L, Harrison W, Fenwick A. National mapping of soil-transmitted helminth and schistosome infections in Ethiopia. Parasites Vectors [Internet]. 2020 Sep 1 [cited 2026 Mar 11];13(1):437. doi: 10.1186/s13071-020-04317-6 Available from: https://link.springer.com/article/10.1186/s13071-020-04317-6
- Okoyo C, Campbell SJ, Williams K, Simiyu E, Owaga C, Mwandawiro C. Prevalence, intensity and associated risk factors of soil-transmitted helminth and schistosome infections in Kenya: Impact assessment after five rounds of mass drug administration in Kenya. Montresor A, editor. PLoS Negl Trop Dis [Internet]. 2020 Oct 7 [cited 2026 Mar 11];14(10):e0008604. doi: 10.1371/journal.pntd.0008604 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0008604 Erratum in: Correction: Prevalence, intensity and associated risk factors of soil-transmitted helminth and schistosome infections in Kenya: Impact assessment after five rounds of mass drug administration in Kenya. PLoS Negl Trop Dis [Internet]. 2022 Jun 15 [cited 2026 Mar 11];16(6):e0010550. doi: 10.1371/journal.pntd.0010550 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0010550
- Dawaki S, Al-Mekhlafi HM, Ithoi I, Ibrahim J, Abdulsalam AM, Ahmed A, Sady H, Nasr NA, Atroosh WM. The Menace of Schistosomiasis in Nigeria: Knowledge, Attitude, and Practices Regarding Schistosomiasis among Rural Communities in Kano State. Kumar N, editor. PLoS ONE [Internet]. 2015 Nov 25 [cited 2026 Mar 11];10(11):e0143667. doi: 10.1371/journal.pone.0143667 Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0143667
- Dejon-Agobé JC, Zinsou JF, Honkpehedji YJ, Edoa JR, Adegbité BR, Beh-Mba R, Kremsner PG, Adegnika AA, Grobusch MP. Knowledge, attitudes and practices pertaining to urogenital schistosomiasis in Lambaréné and surrounding areas, Gabon. Parasites Vectors [Internet]. 2021 Sep 22 [cited 2026 Mar 11];14(1):486. doi: 10.1186/s13071-021-04905-0 Available from: https://link.springer.com/article/10.1186/s13071-021-04905-0
- Adoka SO, Anyona DN, Abuom PO, Dida GO, Karanja D, Vulule JM, Okurut T, Matano AS, Gichere SK, Ofulla AVO. Community perceptions of schistosomiasis transmission, prevalence and control in relation to aquatic habitats in the Lake Victoria basin of Kenya. East Afr Med J. 2014 Jul [cited 2026 Mar 11];91(7):232–44. Available from: https://pubmed.ncbi.nlm.nih.gov/26862658/ Abstract only.
- Anyolitho MK, Poels K, Huyse T, Tumusiime J, Mugabi F, Tolo CU, Masquillier C, Nyakato VN. Knowledge, attitudes, and practices regarding schistosomiasis infection and prevention: A mixed-methods study among endemic communities of western Uganda. Dikomitis L, editor. PLoS Negl Trop Dis [Internet]. 2022 Feb 23 [cited 2026 Mar 11];16(2):e0010190. doi: 10.1371/journal.pntd.0010190 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0010190
- Mbereko A, Chimbari MJ, Manyangadze T, Mukaratirwa S. Knowledge and perceptions of schistosomiasis, a water-borne disease, in two semi-arid rural areas of South Africa (Ndumo) and Zimbabwe (Ntalale). Food and Waterborne Parasitology [Internet]. 2020 Oct 20 [version of record 2020 Nov 16; cited 2026 Mar 11];21:e00091. doi: 10.1016/j.fawpar.2020.e00091 Available from: https://www.sciencedirect.com/science/article/pii/S2405676620300202?via%3Dihub
- Sady H, Al-Mekhlafi HM, Atroosh WM, Al-Delaimy AK, Nasr NA, Dawaki S, Al-Areeqi MA, Ithoi I, Abdulsalam AM, Chua KH, Surin J. Knowledge, attitude, and practices towards schistosomiasis among rural population in Yemen. Parasites Vectors [Internet]. 2015 Aug 25 [cited 2026 Mar 11];8(1):436. doi: 10.1186/s13071-015-1050-8 Available from: https://link.springer.com/article/10.1186/s13071-015-1050-8
- Francisco I, Jiz M, Rosenbaum M, Baltazar P, Steele JA. Knowledge, attitudes and practices related to schistosomiasis transmission and control in Leyte, Philippines. Soares Magalhaes RJ, editor. PLoS Negl Trop Dis [Internet]. 2019 May 2 [cited 2026 Mar 11];13(5):e0007358. doi: 10.1371/journal.pntd.0007358 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0007358
- Djagadou KA, Tchamdja T, Némi KD, Balaka A, Djibril MA. [Knowledge, attitudes and practices of the population of the city of Lomé regarding the prevention of schistosomiasis: the case of the canton of Légbassito]. Pan Afr Med J [Internet]. 2019 Sep 10 [cited 2026 Mar 11];34:19. French. doi: 10.11604/pamj.2019.34.19.18918 Available from: https://www.panafrican-med-journal.com/content/article/34/19/full/
- Folefac LN, Nde-Fon P, Verla VS, Tangye MN, Njunda AL, Luma HN. Knowledge, attitudes and practices regarding urinary schistosomiasis among adults in the Ekombe Bonji Health Area, Cameroon. Pan Afr Med J [Internet]. 2018 Mar 19 [cited 2026 Mar 11];29:161. doi: 10.11604/pamj.2018.29.161.14980 Available from: https://www.panafrican-med-journal.com/content/article/29/161/full/
- Assefa A, Erko B, Gundersen SG, Medhin G, Berhe N. Low awareness and common misconceptions about schistosomiasis in endemic lowland areas in Western Ethiopia: a mixed-methods study. BMC Public Health [Internet]. 2021 Jun 4 [cited 2026 Mar 11];21(1):1064. doi: 10.1186/s12889-021-11106-y Available from: https://link.springer.com/article/10.1186/s12889-021-11106-y
- Koffi AJ d’Arc, Doumbia M, Fokou G, Keita M, Koné B, Abé NN. Community knowledge, attitudes and practices related to schistosomiasis and associated healthcare-seeking behaviours in northern Côte d’Ivoire and southern Mauritania. Infect Dis Poverty [Internet]. 2018 Jul 10 [cited 2026 Mar 11];7(1):70. doi: 10.1186/s40249-018-0453-0 Available from: https://link.springer.com/article/10.1186/s40249-018-0453-0 Erratum in: Correction to: Community knowledge, attitudes and practices related to schistosomiasis and associated healthcare-seeking behaviours in northern Côte d’Ivoire and southern Mauritania. Infect Dis Poverty [Internet]. 2018 Aug 23 [cited 2026 Mar 11];7(1):93. doi: 10.1186/s40249-018-0476-6 Available from: https://link.springer.com/article/10.1186/s40249-018-0476-6
- Bailleul C, Davydov A, Erman A, Maslinsky K, Méric JJ, Vydrin V. [Bamadaba: Bambara orthographic dictionary, with a French-Bambara index. 2011–2022] [Internet]. Paris (France): [publisher unknown]; 2022 Dec 22 [cited 2026 Mar 11]. French. Available from: http://cormand.huma-num.fr/Bamadaba/lexicon/index.htm
- Dicko I, Coulibaly YI, Sangaré M, Sarfo B, Nortey PA. Non-compliance to Mass Drug Administration Associated with the Low Perception of the Community Members About their Susceptibility to Lymphatic Filariasis in Ankobra, Ghana. IDDT [Internet]. 2020 May 13 [cited 2026 Mar 11];20(2):167–74. doi: 10.2174/1871526519666190206210808 Available from: https://www.eurekaselect.com/article/96445 Purchase or subscription required to view full text.
- Agboraw E, Sosu F, Dean L, Siakeh A, Thomson R, Kollie K, Worrall E. Factors influencing mass drug administration adherence and community drug distributor opportunity costs in Liberia: a mixed-methods approach. Parasites Vectors [Internet]. 2021 Oct 28 [cited 2026 Mar 11];14(1):557. doi: 10.1186/s13071-021-05058-w Available from: https://link.springer.com/article/10.1186/s13071-021-05058-w
- Chami GF, Kabatereine NB, Tukahebwa EM. Profiling the best-performing community medicine distributors for mass drug administration: a comprehensive, data-driven analysis of treatment for schistosomiasis, lymphatic filariasis, and soil-transmitted helminths in Uganda. BMC Med [Internet]. 2019 Mar 28 [cited 2026 Mar 11];17(1):69. doi: 10.1186/s12916-019-1303-z Available from: https://link.springer.com/article/10.1186/s12916-019-1303-z
- Inobaya MT, Chau TN, Ng SK, MacDougall C, Olveda RM, Tallo VL, Landicho JM, Malacad CM, Aligato MF, Guevarra JB, Ross AG. Mass drug administration and the sustainable control of schistosomiasis: an evaluation of treatment compliance in the rural Philippines. Parasites Vectors [Internet]. 2018 Jul 31 [cited 2026 Mar 11];11(1):441. doi: 10.1186/s13071-018-3022-2 Available from: https://link.springer.com/article/10.1186/s13071-018-3022-2
- Olveda DU, McManus DP, Ross AGP. Mass drug administration and the global control of schistosomiasis: successes, limitations and clinical outcomes. Current Opinion in Infectious Diseases [Internet]. 2016 Dec [cited 2026 Mar 11];29(6):595–608. doi: 10.1097/QCO.0000000000000312 Available from: https://journals.lww.com/co-infectiousdiseases/abstract/2016/12000/mass_drug_administration_and_the_global_control_of.8.aspx Purchase or subscription required to view full text.
