Research | Open Access | Volume 9 (Suppl 12): 1 | Published: 18 Mar 2026
Lassa fever trends and outcomes from 2018 to April 2025 in Ebonyi State, Nigeria: A descriptive analysis of surveillance data
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| VARIABLE | FREQUENCY (N=416) | PERCENTAGE (%) |
|---|---|---|
| Age (years) | ||
| <11 | 43 | 10.3 |
| 11-20 | 58 | 13.9 |
| 21-30 | 102 | 24.5 |
| 31-40 | 105 | 25.2 |
| 41-50 | 62 | 14.9 |
| ≥51 | 46 | 11.1 |
| Sex | ||
| Female | 204 | 49.0 |
| Male | 212 | 51.0 |
Table 1: Distribution of Confirmed Lassa Fever Cases by Age and Sex Reported in Ebonyi State Nigeria, from January 2018 to April 2025
| SOURCE STATE OF CASE | SOURCE LGA OF CASE | FREQUENCY (N=416) | PERCENTAGE (%) |
|---|---|---|---|
| Ebonyi (n=403) | Abakaliki | 199 | 47.8 |
| Izzi | 47 | 11.3 | |
| Ezza North | 46 | 11.1 | |
| Ikwo | 28 | 6.7 | |
| Ebonyi | 22 | 5.3 | |
| Ezza South | 17 | 4.1 | |
| Ohaukwu | 15 | 3.6 | |
| Afikpo North | 9 | 2.2 | |
| Onicha | 8 | 1.9 | |
| Ohaozara | 6 | 1.4 | |
| Ishielu | 5 | 1.2 | |
| Afikpo South | 1 | 0.2 | |
| Enugu (n=4) | Uzo-Uwani | 2 | 0.5 |
| Nkanu West | 1 | 0.2 | |
| Enugu East | 1 | 0.2 | |
| Abia (n=3) | Bende | 1 | 0.2 |
| Aba South | 1 | 0.2 | |
| Umuahia North | 1 | 0.2 | |
| Cross River (n=3) | Ogoja | 1 | 0.2 |
| Yala | 1 | 0.2 | |
| Obubara | 1 | 0.2 | |
| Anambra (n=1) | Aguata | 1 | 0.2 |
| Imo (n=1) | Owerri Municipal | 1 | 0.2 |
| Delta (n=1) | Ika North | 1 | 0.2 |
LGA: Local Government Area; n = Number of cases from each State, respectively
Table 2: Geographic Distribution of Confirmed Lassa Fever Cases in Ebonyi State, Nigeria, 2018–April 2025, by State and Local Government Area of Origin
Figures
Keywords
- Lassa Fever
- Epidemiological Trend
- Disease Surveillance
- Retrospective Studies
- Nigeria
Cosmas Kenan Onah1,&, Onyinyechukwu Uzoamaka Oka1, Chijioke Vitalus Iloke1, Marycynthia Nnenna Otta1, Nwambeke Ogbonna Nwambeke2, Robinson Chukwudi Onoh3, Benedict Ndubueze Azuogu1
1Department of Community Medicine, Alex Ekwueme Federal University/Teaching Hospital, Abakaliki, Ebonyi State, Nigeria, 2Epidemiology Unit, State Ministry of Health, Abakaliki, Ebonyi State, Nigeria, 3Department of Obstetrics and Gynaecology, Faculty of Clinical Medicine, College of Medicine, Alex Ekwueme Federal University, Ndufu-Alike Ikwo, Ebonyi State, Nigeria
&Corresponding author: Cosmas Kenan Onah, Department of Community Medicine, Alex Ekwueme Federal University Teaching Hospital, Abakaliki, Ebonyi State, Nigeria, Email: onahcosyo@gmail.com ORCID: https://orcid.org/0000-0002-3315-7851
Received: 23 Nov 2025, Accepted: 13 Mar 2026, Published: 18 Mar 2026
Domain: Infectious Disease Epidemiology
Keywords: Lassa Fever, Epidemiological Trend, Disease Surveillance, Retrospective Studies, Nigeria.
©Cosmas Kenan Onah et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Cosmas Kenan Onah et al., Lassa fever trends and outcomes from 2018 to April 2025 in Ebonyi State, Nigeria: A descriptive analysis of surveillance data. Journal of Interventional Epidemiology and Public Health. 2026; 9(Suppl 12):1. https://doi.org/10.37432/jieph-d-25-00300
Abstract
Introduction: Lassa fever remains a major public health threat in West Africa, with Nigeria bearing a substantial burden of the disease. Ebonyi State, in southeastern Nigeria, is a recognized hotspot, and the Alex Ekwueme Federal University Teaching Hospital Abakaliki (AEFUTHA) serves as the region’s principal treatment centre and a designated site for a Phase 2b Lassa fever vaccine clinical trial. This study described trends and outcomes among confirmed Lassa fever cases to generate epidemiological insights that can strengthen surveillance and inform the design and planning of future clinical research in endemic settings.
Methods: We conducted a retrospective descriptive study of confirmed Lassa fever cases reported in Ebonyi State from January 2018 to April 2025. Data was sourced from Ebonyi State Ministry of Health surveillance records and AEFUTHA’s Lassa fever treatment register. All 416 confirmed cases were analyzed using IBM-SPSS version 25 to assess temporal trends, age and sex distribution, seasonal patterns, geographic origin, survival and case fatality rates.
Results: Males accounted for 56.5% of cases. The most affected age groups were 31–40 years (105/416; 25.2%) and 21–30 years (102/416; 24.5%). The overall case fatality rate was 38.5% (160/416), with higher annual rates observed in 2025 (11/19; 57.9%) and 2023 (33/58; 56.9%). Seasonally, 83.7% (348/416) of cases occurred between January and March. Geographically, 96.9% (403/416) originated from Ebonyi State, with a few referrals from neighbouring states. Abakaliki Local Government Area accounted for nearly half of all cases (199/416; 47.8%), followed by Izzi (47/416; 11.3%) and Ezza North (46/416; 11.1%). The highest annual number of confirmed cases was recorded in 2020 (90/416; 21.6%), with lower counts observed in subsequent years.
Conclusion: This eight-year trend analysis describes key epidemiological patterns of confirmed Lassa fever in Ebonyi State and provides evidence to support the strengthening of surveillance and planning of clinical research in an endemic setting. By identifying high-burden age groups, seasonal peaks, and geographic hotspots, the study offers contextual insights for prioritizing prevention, improving clinical care, and informing future research in Lassa fever–endemic settings.
Introduction
Lassa fever (LF) is an acute viral haemorrhagic disease caused by Lassa virus, an arenavirus first identified in the town of Lassa, Nigeria, in 1969 and formally described in 1970 [1,2]. The natural reservoir is the multimammate rat – Mastomys natalensis, which sheds the virus in urine and faeces. Humans become infected through ingestion or inhalation of contaminated food or household materials, while secondary human-to-human transmission occurs via contact with blood, secretions, or contaminated medical equipment [2].
The disease is endemic in West Africa, particularly Nigeria, Sierra Leone, Liberia, and Guinea, with Nigeria carrying the heaviest burden. Lassa virus is estimated to cause 100,000 – 300,000 infections and about 5,000 deaths annually, though these are rough estimates due to diagnostic limitations and extrapolation from historical data [3–5]. Transmission shows a strong seasonal pattern, peaking in the dry months of December – April when rodents increasingly enter homes in search of food due to food scarcity [6]. Risk factors include poor housing, unsafe food storage, rodent infestation, and delayed healthcare access. Certain occupational groups, including farmers, traders, and healthcare workers face increased exposure risk [7,8]. Previous studies from Nigeria have shown that LF predominantly affects young and middle-aged adults (often 21–40 years), with a nearly equal sex distribution and a slight male predominance [9–11]. Case fatality rates (CFRs) vary: the World Health Organization (WHO) estimates 15–20% among hospitalized cases, but rates can exceed 50% during outbreaks or with late presentation, with pregnant women facing the highest mortality and fetal loss approaching 95% [2].
In Nigeria, frequent outbreaks and high fatality rates have been repeatedly documented across nearly all regions, underscoring the disease’s persistent public health threat [12]. Ebonyi State in southeast Nigeria has been a major hotspot, experiencing recurrent outbreaks since 2005, including hospital-based epidemics in Abakaliki with high fatality rates. Between 2005 and 2014, 82 cases were reported with an overall fatality rate of 30.5%, and subsequent outbreaks have reaffirmed Ebonyi as one of the most affected states in the country [13,14]. Outbreaks in Ebonyi and other endemic states follow the dry-season peak, further emphasizing the predictable epidemiology of the disease [6,12,15].
In response to the continuing high burden, experts and health authorities emphasize the urgent need for effective diagnostics, robust surveillance, and most crucially, the development of a protective LF vaccine [4,16]. Alex Ekwueme Federal University Teaching Hospital Abakaliki (AEFUTHA) has been designated as a Phase 2b vaccine trial site under the Coalition for Epidemic Preparedness and Innovation (CEPI)’s ENABLE Lassa programme, marking a critical step toward advancing vaccine development in West Africa [17–20].
This study analyzes LF surveillance data on confirmed cases from Ebonyi and neighbouring States managed at AEFUTHA between January 2018 and April 2025 to describe temporal, demographic, seasonal, and geographic patterns. The goal is to provide updated epidemiological context relevant to strengthening surveillance and planning clinical and public health research activities in this endemic setting.
Methods
Study area
This study was conducted in Ebonyi State, a LF endemic region in southeastern Nigeria. The AEFUTHA, located in the state capital, is the only designated LF treatment centre for Ebonyi State and also receives referrals from neighbouring states, including Enugu, Abia, Anambra, Cross River, and Benue. As a result, LF surveillance data for Ebonyi State are largely generated from cases managed at AEFUTHA. Although AEFUTHA has been selected as a site for a Phase 2b LF vaccine clinical trial, this study was based solely on routine surveillance and hospital records and did not involve any trial-related activities. However, the routine LF surveillance data from AEFUTHA were used to guide the selection of communities for participant enrollment into the ENABLE Lassa Research Programmes, an epidemiological study designed to generate baseline data to support subsequent LF vaccine trial activities [21].
Study design and population
The study utilized a retrospective descriptive design to examine temporal, demographic, geographic, and seasonal trends of confirmed LF cases reported over an eight-year period, from January 2018 to April 2025. The study population comprised all individuals with laboratory-confirmed LF managed at AEFUTHA during the study period, including residents of Ebonyi State and referrals from neighbouring states, as documented by the State Ministry of Health and AEFUTHA.
Sample size and sampling technique
A total of 416 confirmed LF cases were included in the final dataset. The study employed total population sampling, in which all eligible confirmed cases within the defined period were reviewed to provide a descriptive account of reported cases without applying any exclusion criteria.
Data collection
Data for the period between 2020 and April 2025 were obtained from the Epidemiology Division of the Ebonyi State Ministry of Health. Records for 2018 and 2019 were extracted from the treatment registers of the Virology Centre at AEFUTHA. Only laboratory-confirmed cases were included in the analysis. A confirmed LF case was defined as any patient with laboratory confirmation of Lassa virus infection by reverse transcription polymerase chain reaction (RT-PCR). Data on suspected or probable cases were not included in the analysis due to inconsistent documentation across the study period.
Data analysis
Both datasets were reviewed for completeness, cleaned, and harmonized using Microsoft Excel to eliminate duplicate entries and address inconsistencies. The harmonized dataset was imported into IBM SPSS Statistics version 25 for analysis. Descriptive statistics were used to explore temporal trends, demographic characteristics such as age and sex, geographic origin of the cases, seasonal distribution, and patient outcomes, including survival status and CFRs.
Ethical considerations
Ethical approval for this study was obtained from the Health Research Ethics Committee of AEFUTHA, with reference number: AEFUTHA/REC/VOL 3/2025/538. The study involved secondary analysis of existing health records and did not include direct contact with patients. All data were fully anonymized before analysis to ensure the privacy and confidentiality of affected individuals.
Results
A total of 416 confirmed LF cases were recorded in Ebonyi State between January 2018 and April 2025. Lassa infection predominantly affected young and middle-aged adults, with the 31–40-year age group contributing the largest proportion of cases (105/416, 25.2%; Table 1), closely followed by the 21–30-year age group (102/416; 24.5%). Children younger than 11 years accounted for 10.3% (43/416) of cases. Sex distribution was nearly equal, with a slight male predominance (51.0%), females (49.0%).
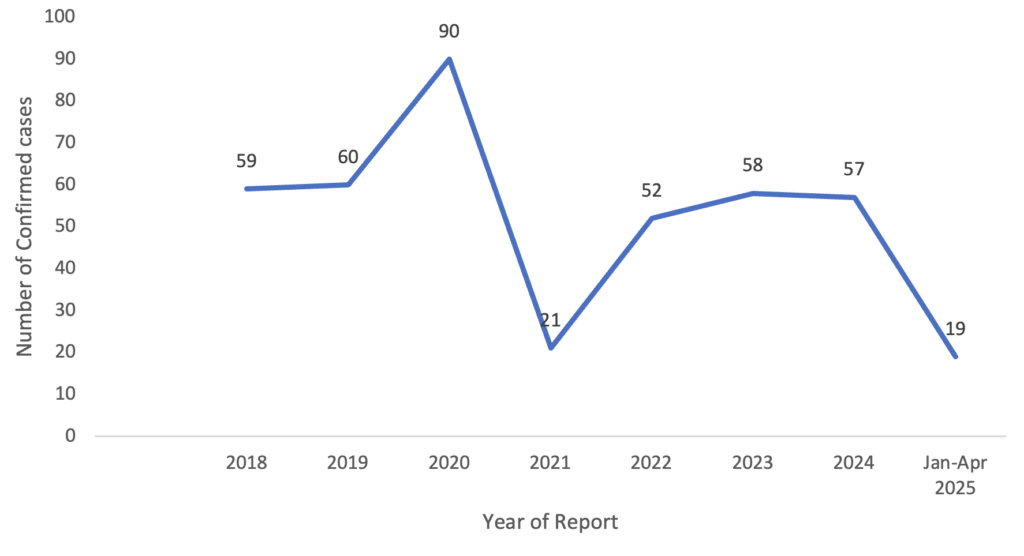
The annual distribution demonstrated variation in the number of reported cases across the years (Figure 1). The highest annual number of cases was recorded in 2020, with 90 cases (21.6%), followed by 2019 (60/416; 14.4%) and 2018 (59/416; 14.2%). Lower numbers of cases were recorded in 2021 (21/416; 5.0%) and in the first quarter of 2025 (19/416; 4.6%).
The majority (403/416; 96.9%) of cases originated from Ebonyi State, while 3.1% (13/416) were referrals from neighbouring states, including Enugu (4/416; 1.0%), Abia (3/416; 0.7%), Cross River (3/416; 0.7%), Imo (1/416; 0.2%), Delta (1/416; 0.2%), and Anambra (1/416; 0.2%; Table 2). At the Local Government Area (LGA) level, Abakaliki contributed nearly half of all cases (199/416; 47.8%), followed by Izzi (47/416; 11.3%) and Ezza North (46/416; 11.1%). These three LGAs collectively accounted for over 70% (292/416) of the cases within Ebonyi State. Other LGAs with a relatively high burden were Ikwo (28/416; 6.7%), Ebonyi (22/416; 5.3%), Ezza South (17/416; 4.1%), and Ohaukwu (15/416; 3.6%) (Table 2).
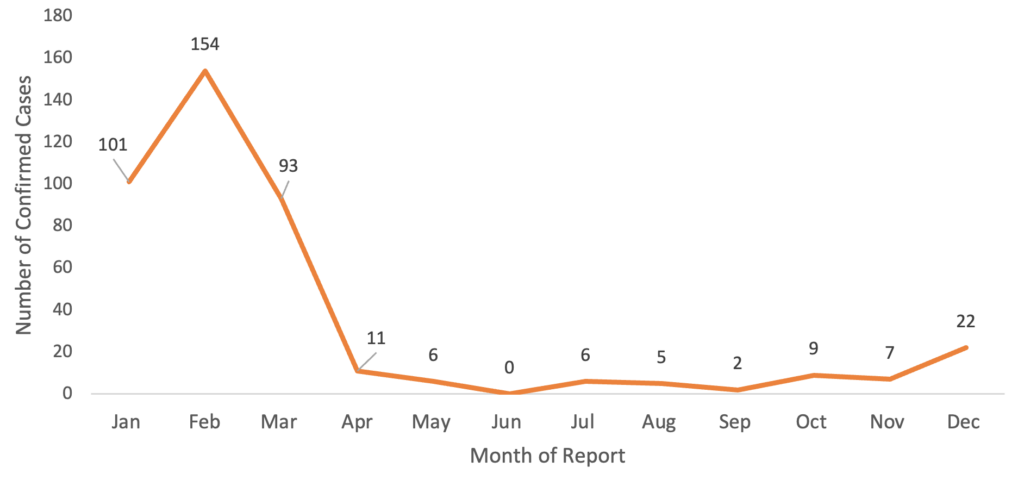
Seasonal distribution showed a distinct clustering during the dry season (Figure 2). Most cases occurred between January and March, with February contributing the highest proportion (154/416; 37.0%), followed by January (101/416; 24.3%) and March (93/416; 22.4%). Only 16.3% (68/416) of cases occurred outside this three-month window, with minimal reported transmission between June and September.
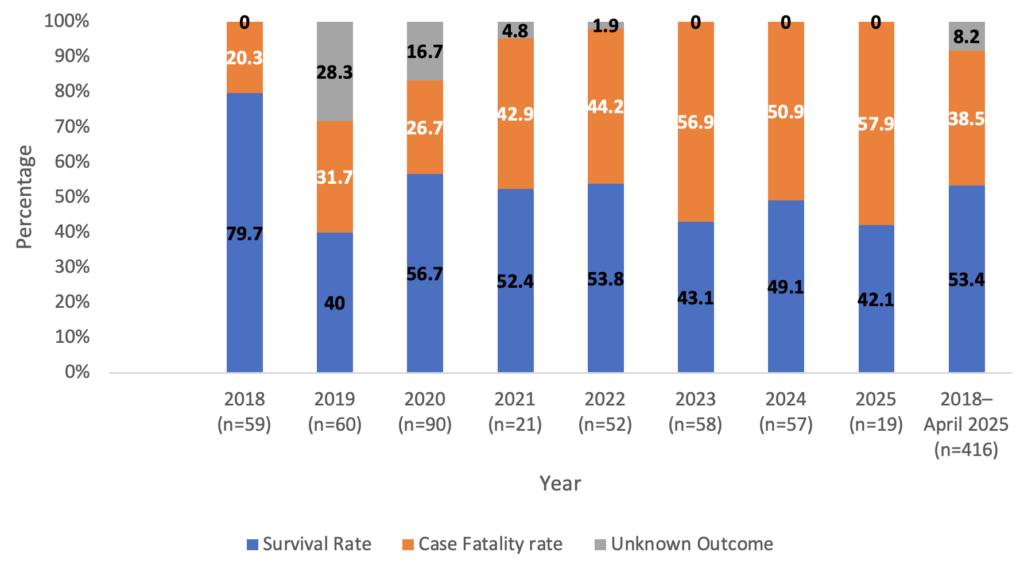
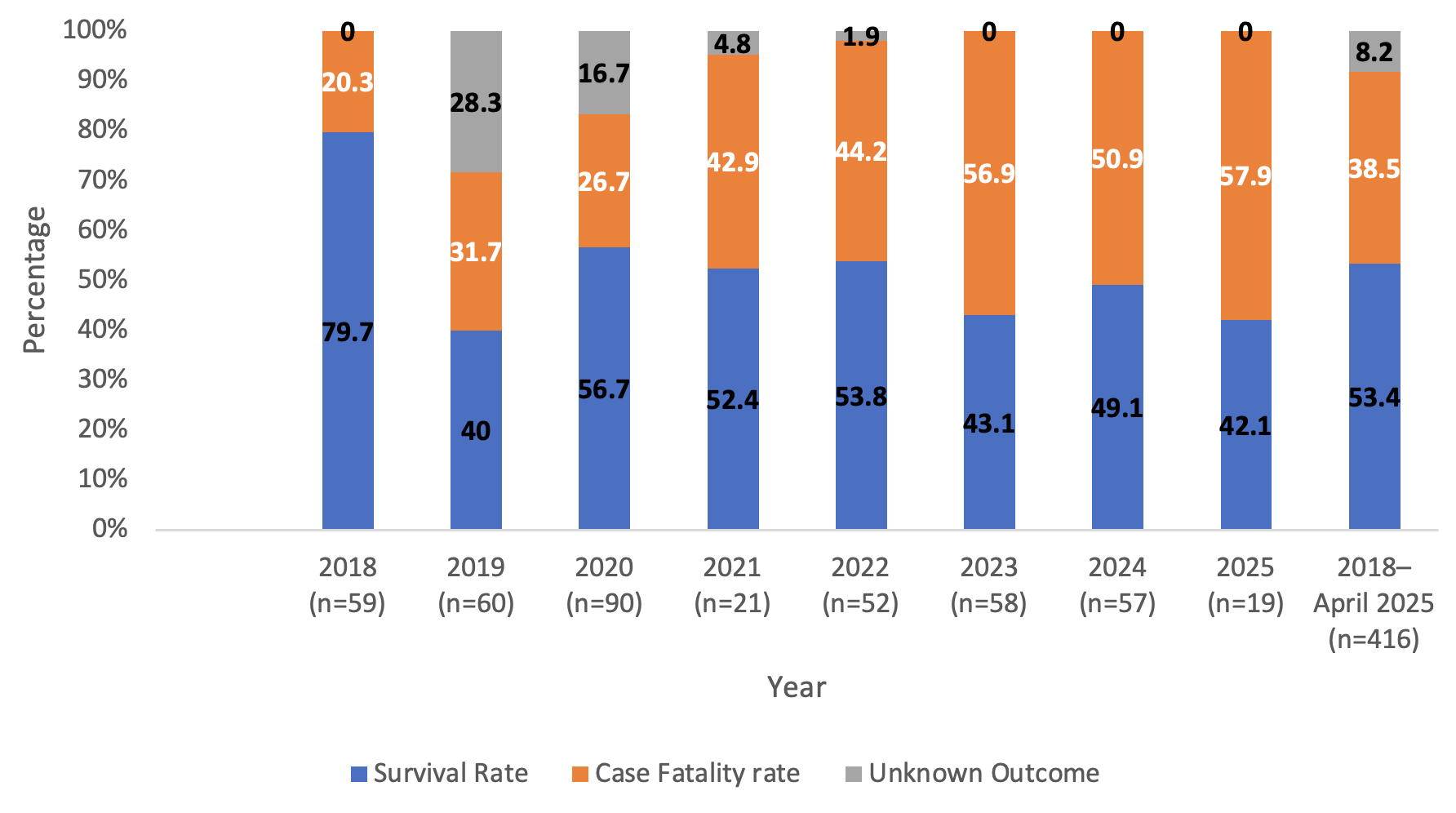
Clinical outcomes are presented in Figure 3. Overall, 53.4% (222/416) of the patients survived, 38.5% (160/416) died, and 8.2% (34/416) had unknown outcomes. Case fatality rates varied across the study period, with higher annual rates observed in 2025 (11/19; 57.9%) and 2023 (33/58; 56.9%), while the lowest was observed in 2018 (12/59; 20.3%). Annual survival proportions also varied, with the highest survival recorded in 2018 (47/59; 79.7%) and the lowest in 2025 (8/19; 42.1%).
Discussion
This eight-year retrospective analysis of LF in Ebonyi State provides an updated description of epidemiological patterns in an endemic setting, based on 416 confirmed cases recorded between January 2018 and April 2025. Reported case numbers varied over time, picking in 2020 and declining in subsequent years, with particularly low counts in 2021 and the first quarter of 2025. These trends parallel national and regional patterns, reaffirming the endemicity of LF in Nigeria [8,22,23]. Temporal variation may reflect changes in surveillance intensity, healthcare-seeking behavior, diagnostic capacity, or reporting completeness, as suggested by prior reviews [8,12].
Demographic analysis highlights that young and middle-aged adults, especially those aged 21–40 years, accounted for the largest proportion of reported cases, with the 31–40- and 21–30-year age groups together contributing nearly half of all cases. This is consistent with occupational and domestic exposures among farmers, traders, and domestic workers in endemic areas and mirrors findings from other Nigerian and West African cohorts [8,23]. The near-equal sex distribution with a slight male predominance indicates that both men and women face substantial exposure risks through shared household and workplace contact with rodents, aligning with national surveillance profiles [22,23].
The CFRs recorded in 2023 and 2025, exceeding 55%, are strikingly higher than both national averages and the WHO estimate of 15–20% among hospitalized patients [2,7]. These elevated rates should be interpreted cautiously, as they may reflect a combination of factors, including late presentation, referral bias to a tertiary treatment centre, limited availability of intensive supportive care, small annual case counts, and incomplete outcome documentation [12,24]. Rather than indicating a true increase in disease severity, these findings highlight ongoing challenges in clinical management and surveillance within endemic settings, and the need to strengthen surveillance systems and improve early case detection, diagnosis, and treatment. Previous studies have shown that survival outcomes improve substantially with early presentation, prompt diagnosis, and timely initiation of ribavirin and supportive care [25].
Geographically, clustering in Abakaliki, Izzi, and Ezza North LGAs suggests that a large proportion of reported cases occurred within a few LGAs, with these three together accounting for over 70% of all cases. Similar associations between environmental risk and outbreak clustering have been reported in other endemic states [25–27]. The high number of cases recorded in Abakaliki may also reflect the presence of AEFUTHA as the state’s main treatment and referral centre, which is likely to improve case detection and reporting. Nonetheless, these findings reinforce the importance of geographically targeted prevention and control measures in high-burden areas, including improved environmental sanitation, safer food storage practices, and other locally appropriate public health interventions [24,28].
The marked seasonality observed, peaking between January and March, is consistent with national and regional evidence. WHO outbreak reports and Nigeria Centre for Disease Control (NCDC) surveillance data consistently document LF spikes during the dry season [29–31]. Ecological studies attribute this to seasonal fluctuations in Mastomys populations and greater spillover during December–March [6], a finding reinforced by state-level analyses in Ebonyi [8]. From a planning perspective, this predictable seasonal pattern may be useful for timing enhanced surveillance, preparedness, and other public health response activities in endemic areas.
Epidemiological data on the timing of case occurrence, demographic characteristics, and geographic origin of cases provide contextual information that may inform targeted public health interventions and planning of future research activities in endemic settings. However, information on stakeholders’ knowledge, perspectives, needs, and the impact of the disease on health services and populations, which are important for planning clinical trials, was not assessed in this study and therefore remains an important areas for further research.
More broadly, these findings underscore the strategic value of routine surveillance data in strengthening the interface between public health practice and clinical research. Locally generated epidemiological evidence can support a clearer understanding of disease burden and transmission patterns in endemic areas. However, translating such epidemiological insights into operational research capacity requires strengthened laboratory systems, diagnostic infrastructure, and data management [10,32]. Strengthening existing health institutions may further facilitate the integration of routine surveillance, public health response, and future research activities [32,33].
Limitations
This study was based on secondary surveillance and treatment register data. Information on some clinically relevant variables, including pregnancy status, was not consistently recorded, making it impossible to estimate pregnancy-specific CFRs. Because the data were derived largely from referrals, referral bias is possible, and the findings may over-represent more severe cases. In addition, underreporting within routine surveillance systems and incomplete documentation of some variables in the register may have influenced the findings.
Conclusion
This eight-year analysis of LF in Ebonyi State describes a fluctuating but persistent burden, with the highest annual case count in 2020 and marked clustering in Abakaliki, Izzi, and Ezza North LGAs. Young and middle-aged adults were most affected, while CFRs in several years remained alarmingly high. The strong seasonal pattern, with most cases occurring between January and March, underscores the predictable epidemiology of the disease.
These findings highlight key epidemiological trends with implications for strengthening surveillance, supporting timely diagnosis, and improving clinical management in endemic settings. The seasonal peaks and geographic clustering described here provide contextual epidemiological information for public health interventions and for planning future clinical research in this endemic setting.
What is already known about the topic
- Lassa fever remains endemic in Nigeria, with Ebonyi State recognized as one of the major hotspots.
- Seasonal peaks typically occur during the dry season (December–April), aligning with increased rodent–human contact.
- Case fatality rates vary widely but can exceed 50% during severe outbreaks or delayed presentation.
- Surveillance and early diagnosis remain critical yet challenging in many endemic regions.
What this study adds
- Provides an updated eight-year epidemiological trend (2018–2025) of Lassa fever in Ebonyi State, using harmonized treatment and surveillance data.
- Identifies specific high-burden LGAs – Abakaliki, Izzi, and Ezza North – which account for more than 70% of reported cases.
- Highlights persistently high case fatality rates, emphasizing urgent needs to strengthen surveillance and clinical management.
- Offers contextually relevant epidemiological information to inform surveillance strengthening, public health planning, and future research activities in Ebonyi State.
Authors´ contributions
CKO: Conceptualization, study design, data acquisition, data analysis, manuscript drafting, and overall supervision.
OUO: Data acquisition, data cleaning, literature review, and manuscript review.
CVI: Data analysis support, results interpretation, and manuscript editing.
MNO: Literature review, results synthesis, and drafting of selected sections.
NON: Contribution to state surveillance data retrieval and interpretation.
RCO: Clinical insights, manuscript review, and advisory contribution.
BNA: Conceptualization, senior review, guidance, manuscript editing, and final approval.
All authors read and approved the final manuscript
| VARIABLE | FREQUENCY (N=416) | PERCENTAGE (%) |
|---|---|---|
| Age (years) | ||
| <11 | 43 | 10.3 |
| 11-20 | 58 | 13.9 |
| 21-30 | 102 | 24.5 |
| 31-40 | 105 | 25.2 |
| 41-50 | 62 | 14.9 |
| ≥51 | 46 | 11.1 |
| Sex | ||
| Female | 204 | 49.0 |
| Male | 212 | 51.0 |
| SOURCE STATE OF CASE | SOURCE LGA OF CASE | FREQUENCY (N=416) | PERCENTAGE (%) |
|---|---|---|---|
| Ebonyi (n=403) | Abakaliki | 199 | 47.8 |
| Izzi | 47 | 11.3 | |
| Ezza North | 46 | 11.1 | |
| Ikwo | 28 | 6.7 | |
| Ebonyi | 22 | 5.3 | |
| Ezza South | 17 | 4.1 | |
| Ohaukwu | 15 | 3.6 | |
| Afikpo North | 9 | 2.2 | |
| Onicha | 8 | 1.9 | |
| Ohaozara | 6 | 1.4 | |
| Ishielu | 5 | 1.2 | |
| Afikpo South | 1 | 0.2 | |
| Enugu (n=4) | Uzo-Uwani | 2 | 0.5 |
| Nkanu West | 1 | 0.2 | |
| Enugu East | 1 | 0.2 | |
| Abia (n=3) | Bende | 1 | 0.2 |
| Aba South | 1 | 0.2 | |
| Umuahia North | 1 | 0.2 | |
| Cross River (n=3) | Ogoja | 1 | 0.2 |
| Yala | 1 | 0.2 | |
| Obubara | 1 | 0.2 | |
| Anambra (n=1) | Aguata | 1 | 0.2 |
| Imo (n=1) | Owerri Municipal | 1 | 0.2 |
| Delta (n=1) | Ika North | 1 | 0.2 |
LGA: Local Government Area; n = Number of cases from each State, respectively



References
- Frame JD, Baldwin JM, Gocke DJ, Troup JM. Lassa Fever, a New Virus Disease of Man from West Africa: I. Clinical Description and Pathological Findings. The American Journal of Tropical Medicine and Hygiene [Internet]. 1970 Jul [cited 2026 Mar 18];19(4):670–6. doi:10.4269/AJTMH.1970.19.670 Available from: https://www.ajtmh.org/view/journals/tpmd/19/4/article-p670.xml
- World Health Organization. Lassa fever [Internet]. Geneva (Switzerland): WHO; 2024 Dec 5 [cited 2026 Mar 18]. Available from: https://www.who.int/news-room/fact-sheets/detail/lassa-fever
- Nigeria CDC (NCDC). Lassa Fever Situation Report Epi Week 33: 11th – 17th August 2025 [Internet]. Abuja (Nigeria): NCDC; 2025 Aug [cited 2026 Mar 18]. 7 p. Available from: https://ncdc.gov.ng/themes/common/files/sitreps/aa4bc98fb7a8256a02bfad73167c5575.pdf
- Besson ME, Pépin M, Metral PA. Lassa Fever: Critical Review and Prospects for Control. TropicalMed [Internet]. 2024 Aug 14 [cited 2026 Mar 18];9(8):178. doi:10.3390/tropicalmed9080178 Available from: https://www.mdpi.com/2414-6366/9/8/178
- Africa CDC. Lassa Fever [Internet]. Addis Ababa (Ethiopia): Africa CDC; c2026 [cited 2026 Mar 18]. Available from: https://africacdc.org/disease/lassa-fever/
- McKendrick JQ, Tennant WSD, Tildesley MJ. Modelling seasonality of Lassa fever incidences and vector dynamics in Nigeria. Nzelu C, editor. PLoS Negl Trop Dis [Internet]. 2023 Nov 13 [cited 2026 Mar 18];17(11):e0011543. doi:10.1371/journal.pntd.0011543 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0011543
- Al-Mustapha AI, Adesiyan IM, Orum TG, Ogundijo OA, Lawal AN, Nzedibe OE, Onyeka LO, Muhammad KU, Odetayo L, Oyewo M, Muhammad SO, Atadiose EO, Adebudo LI, Adetunji DA, Jantiku HJ, Akintule AO, Nwachukwu RC, Abubakar AT. Lassa fever in Nigeria: epidemiology and risk perception. Sci Rep [Internet]. 2024 Nov 12 [cited 2026 Mar 18];14(1):27669. doi:10.1038/s41598-024-78726-3 Available from: https://www.nature.com/articles/s41598-024-78726-3
- Ezenwa-Ahanene A, Salawu AT, Adebowale AS. Descriptive epidemiology of Lassa fever, its trend, seasonality, and mortality predictors in Ebonyi State, South-East, Nigeria, 2018—2022. BMC Public Health [Internet]. 2024 Dec 18 [cited 2026 Mar 18];24(1):3470. doi:10.1186/s12889-024-20840-y Available from: https://link.springer.com/article/10.1186/s12889-024-20840-y
- Grace JUA, Egoh IJ, Udensi N. Epidemiological trends of Lassa fever in Nigeria from 2015-2021: A review. Therapeutic Advances in Infection [Internet]. 2021 Jan [cited 2026 Mar 18];8:20499361211058252. doi:10.1177/20499361211058252 Available from: https://journals.sagepub.com/doi/10.1177/20499361211058252
- Asogun D, Arogundade B, Unuabonah F, Olugbenro O, Asogun J, Aluede F, Ehichioya D. A Review of the Epidemiology of Lassa Fever in Nigeria. Microorganisms [Internet]. 2025 Jun 18 [cited 2026 Mar 18];13(6):1419. doi:10.3390/microorganisms13061419 Available from: https://www.mdpi.com/2076-2607/13/6/1419
- Akindokun SS, Adeleye OO, Olorunlowu DR. The socioeconomic impact of Lassa fever in Nigeria. Discov Public Health [Internet]. 2024 Oct 14 [cited 2026 Mar 18];21(1):133. doi:10.1186/s12982-024-00265-z Available from: https://link.springer.com/article/10.1186/s12982-024-00265-z
- Eneh SC, Obi CG, Ephraim Ikpongifono U, Dauda Z, Udoewah SA, Anokwuru CC, Onukansi FO, Ikhuoria OV, Ojo TO, Madukaku CU, Orabueze IN, Chizoba AF. The resurgence of Lassa fever in Nigeria: economic impact, challenges, and strategic public health interventions. Front Public Health [Internet]. 2025 Jul 16 [cited 2026 Mar 18];13:1574459. doi:10.3389/fpubh.2025.1574459 Available from: https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2025.1574459/full
- Agboeze J, Onoh R, Eze J, Nwali M, Ukaegbe C. Clinical Profile of Lassa Fever Patients in Abakaliki, South-Eastern Nigeria, January – March 2018. Ann Med Health Sci Res [Internet]. 2019 [cited 2026 Mar 18];9:598–602. Available from: http://scholar.google.com/scholar?hl=en&q=+Clinical+Profile+of+Lassa+Fever+Patients+in+Abakaliki+South-+Eastern+Nigeria+January+-+March+2018
- Ehichioya DU, Hass M, Ölschläger S, Becker-Ziaja B, Chukwu COO, Coker J, Nasidi A, Ogugua OO, Günther S, Omilabu SA. Lassa Fever, Nigeria, 2005–2008. Emerg Infect Dis [Internet]. 2010 Jun [cited 2026 Mar 18];16(6):1040–1. doi:10.3201/eid1606.100080 Available from: https://wwwnc.cdc.gov/eid/article/16/6/10-0080_article
- Afeez Bolaji. Drier, hotter, rattier: as the weather shifts, Nigeria braces for Lassa fever 2023 [Internet]. Washington (DC): GAVI; 2023 Nov 29 [cited 2026 Mar 18]. Available from: https://www.gavi.org/vaccineswork/drier-hotter-rattier-weather-shifts-nigeria-braces-lassa-fever
- Moore KA, Ostrowsky JT, Mehr AJ, Johnson RA, Ulrich AK, Moua NM, Fay PC, Hart PJ, Golding JP, Benassi V, Preziosi MP, Adetifa IM, Akpede GO, Ampofo WK, Asogun DA, Barrett ADT, Bausch DG, De Coster I, Emperador DM, Feldmann H, Fichet-Calvet E, Formenty PBH, Garry RF, Grant DS, Günther S, Gupta SB, Jaspard M, Mazzola LT, Okogbenin SA, Roth C, Schmaljohn CS, Osterholm MT. Lassa fever research priorities: towards effective medical countermeasures by the end of the decade. The Lancet Infectious Diseases [Internet]. 2024 Nov [cited 2026 Mar 18];24(11):e696–706. doi:10.1016/S1473-3099(24)00229-9 Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(24)00229-9/abstract
- Udeze EK. Capacity Building Activity on Lassa Fever Vaccine Clinical Trials: Strengthening Research Capacity in West Africa [Internet]. Abuja (Nigeria): AEFUTHA; 2025 [cited 2026 Mar 18]. Available from: https://www.aefutha.gov.ng/index.php/news-and-events/capacity-building-activity-on-lassa-fever-vaccine-clinical-trials-strengthening-research-capacity-in-west-africa
- CEPI. Lassa fever [Internet]. Oslo (Norway): CEPI; c2026 [cited 2026 Mar 18]. Available from: https://cepi.net/lassa-fever
- CEPI. Participants in Nigeria vaccinated in first-ever Phase 2 Lassa fever vaccine clinical trial [Internet]. Oslo (Norway): CEPI; 2024 Apr 4 [cited 2026 Mar 18]. Available from: https://cepi.net/participants-nigeria-vaccinated-first-ever-phase-2-lassa-fever-vaccine-clinical-trial
- U.S. Embassy in Nigeria. New Lassa Vaccine Trial in Nigeria [Internet]. Abuja (Nigeria): U.S. Embassy in Nigeria; 2021 Jun 18 [cited 2026 Mar 18]. Available from: https://ng.usembassy.gov/new-lassa-vaccine-trial-in-nigeria/
- Penfold S, Adegnika AA, Asogun D, Ayodeji O, Azuogu BN, Fischer WA, Garry RF, Grant DS, Happi C, N’Faly M, Olayinka A, Samuels R, Sibley J, Wohl DA, Accrombessi M, Adetifa I, Annibaldis G, Camacho A, Dan-Nwafor C, Deha ARE, DeMarco J, Duraffour S, Goba A, Grais R, Günther S, Honvou ÉJJP, Ihekweazu C, Jacobsen C, Kanneh L, Momoh M, Ndiaye A, Nsaibirni R, Okogbenin S, Ochu C, Ogbaini E, Logbo ÉPMA, Sandi JD, Schieffelin JS, Verstraeten T, Vielle NJ, Yadouleton A, Yovo EK, on behalf of the Enable Protocol authorship group. A prospective, multi-site, cohort study to estimate incidence of infection and disease due to Lassa fever virus in West African countries (the Enable Lassa research programme)–Study protocol. Bhattacharya D, editor. PLoS ONE [Internet]. 2023 Mar 30 [cited 2026 Mar 18];18(3):e0283643. doi:10.1371/journal.pone.0283643 Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0283643
- NCDC. Lassa Fever Situation Report: Epi Week 24: 9th – 15th June 2025 [Internet]. Abuja (Nigeria): NCDC; 2025 Jun [cited 2026 Mar 18]. 7 p. Available from: https://ncdc.gov.ng/themes/common/files/sitreps/17f59c56d38345b9f7de4c38b5838e4d.pdf
- Emembolu CE, Ikeanyionwu IV, Obidike BA, Uchebo OA, Onwuegbuna AA, Ekwebene OC, Chijioke-Ofoma UC, Uzoewulu NG, Njoku CC, Eleje GU. Epidemiology, fatality rate, and geographic distribution of Lassa fever in Anambra state, Nigeria. Discov Public Health [Internet]. 2025 Jun 25 [cited 2026 Mar 18];22(1):360. doi:10.1186/s12982-025-00763-8 Available from: https://link.springer.com/article/10.1186/s12982-025-00763-8
- Sigia Consultancy. Evaluation of MSF’S lassa fever project in Abakaliki, Nigeria [Internet]. Porto (Portugal): Sigia Consultancy; 2024 Jan 29 [cited 2026 Mar 18]. 70 p. Available from: https://evaluation.msf.org/sites/default/files/2024-04/evl_2024_abala_dis_finalreport.pdf
- Wogu JO. Mass media awareness campaign and the prevention of the spread of Lassa fever in the rural communities of Ebonyi State, Nigeria: Impact evaluation. J Public Health Afr [Internet]. 2018 Dec 21 [cited 2026 Mar 18];9(3):a935. doi:10.4081/jphia.2018.882 Available from: https://publichealthinafrica.org/index.php/jphia/article/view/953
- Ekwueme AC, Asogwa FN. Social Media and Health Mobilization During Emergencies: The Case of Lassa Fever Outbreak in Ebonyi, Nigeria [Internet]. Nsukka (Nigeria): University of Nigeria; 2020 Dec [cited 2026 Mar 18]. 73 p. Available from: https://journals.covenantuniversity.edu.ng/index.php/cjoc/article/view/2428/1226
- Awofe KI. Halting an Infectious Disease Outbreak: Nigeria’s Centre for Disease Control Contains Lassa Fever and Sets a Prevention Plan, 2015 to 2017 [Internet]. Abuja (Nigeria): Global Delivery Initiative; 2020 Sep [cited 2026 Mar 18]. 10 p. Available from: https://www.effectivecooperation.org/sites/default/files/documents/GDI%20KDIS%20Case%20Study%20Nigeria%20Combating%20Lassa%20Fever.pdf
- Zubaida Baba Ibrahim, Ibukun Oguntola. Addressing Lassa Fever in Nigeria: The Case for Vaccines and Civil Society Engagement [Internet]. Abuja (Nigeria): Nigeria Health Watch; 2025 Feb 1 [cited 2026 Mar 18]. Available from: https://articles.nigeriahealthwatch.com/addressing-lassa-fever-in-nigeria-the-case-for-vaccines-and-civil-society-engagement/
- World Health Organization. Lassa fever – Nigeria 2023 [Internet]. Geneva (Switzerland): WHO; 2023 May 1 [cited 2026 Mar 18]. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2023-DON463
- Ilori EA, Frank C, Dan-Nwafor CC, Ipadeola O, Krings A, Ukponu W, Womi-Eteng OE, Adeyemo A, Mutbam SK, Musa EO, Lasuba CLP, Alemu W, Okogbenin S, Ogbaini E, Unigwe U, Ogah E, Onoh R, Abejegah C, Ayodeji O, Ihekweazu C. Increase in Lassa Fever Cases in Nigeria, January–March 2018. Emerg Infect Dis [Internet]. 2019 May [cited 2026 Mar 18];25(5):1026–7. doi:10.3201/eid2505.181247 Available from: https://wwwnc.cdc.gov/eid/article/25/5/18-1247_article
- NCDC. Lassa Fever 2019 [Internet]. Abuja (Nigeria): NCDC; 2019 Apr 4 [cited 2026 Mar 18]. Available from: https://ncdc.gov.ng/diseases/factsheet/47
- Aerie Em. Advancing research capacity and pandemic preparedness in West Africa [Internet]. Seoul (Republic of Korea): International Vaccine Institute; 2024 Dec 16 [cited 2026 Mar 18]. Available from: https://www.ivi.int/advancing-research-capacity-and-pandemic-preparedness-in-west-africa/
- Ibukun Oguntola, Oluomachukwu Omeje. Strengthening Lassa Fever Vaccine Research and Trial Capacity in West Africa [Internet]. Abuja (Nigeria): Nigeria Health Watch; 2025 Aug 13 [cited 2026 Mar 18]. Available from: https://articles.nigeriahealthwatch.com/strengthening-lassa-fever-vaccine-research-and-trial-capacity-in-west-africa/
