Research | Open Access | Volume 9 (1): Article 49 | Published: 23 Mar 2026
Coverage and determinants of intermittent preventive treatment for malaria in pregnancy with sulfadoxine-pyrimethamine in Guinea, 2021
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
Table 1: Sociodemographic characteristics of participants according to the coverage of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15 to 49 whose last live birth occurred within the past two years before the survey, malaria and anemia indicators survey, Guinea, 2021.
| Variables | Frequency n (%) | Women who received at least three doses of IPTp-SP | p-value | |
|---|---|---|---|---|
| Yes n (%) | No n (%) | |||
| Total | 1,511 | 760 (50.30) | 751 (49.70) | |
| Region | <0.001 | |||
| Boké | 184 (12.2) | 76 (41.30) | 108 (58.70) | |
| Conakry | 192 (12.71) | 104 (54.17) | 88 (45.83) | |
| Faranah | 142 (9.42) | 85 (59.85) | 58 (40.85) | |
| Kankan | 297 (19.68) | 113 (38.05) | 185 (61.95) | |
| Kindia | 233 (15.44) | 125 (53.65) | 108 (46.35) | |
| Labé | 129 (8.55) | 60 (46.51) | 69 (53.49) | |
| Mamou | 91 (6.02) | 40 (43.95) | 50 (54.95) | |
| N’Zérékoré | 241 (15.98) | 157 (65.15) | 84 (34.85) | |
| Place of residence | 0.002 | |||
| Rural | 1,099 (72.78) | 526 (47.86) | 573 (52.14) | |
| Urban | 411 (27.22) | 234 (56.93) | 177 (43.07) | |
| Sex of the head of household | 0.165 | |||
| Male | 1,323 (87.64) | 657 (49.65) | 666 (50.35) | |
| Female | 187 (12.36) | 103 (55.08) | 84 (44.92) | |
| Age of the head of household (years) | 0.506 | |||
| <26 | 38 (2.50) | 19 (50.00) | 19 (50.00) | |
| 26-34 | 347 (22.99) | 182 (52.40) | 165 (47.60) | |
| 35-44 | 437 (28.95) | 205 (46.80) | 233 (53.20) | |
| 45-54 | 296 (19.58) | 151 (51.00) | 145 (49.00) | |
| 55+ | 392 (25.98) | 204 (52.00) | 188 (48.00) | |
| Household size | 0.173 | |||
| Medium | 626 (41.45) | 312 (49.84) | 314 (50.16) | |
| Large | 506 (33.53) | 270 (53.36) | 236 (46.64) | |
| Small | 378 (25.02) | 178 (47.10) | 200 (52.90) | |
| Household wealth index | 0.021 | |||
| Poor | 652 (43.16) | 319 (48.91) | 333 (51.07) | |
| Middle | 314 (20.76) | 143 (45.50) | 171 (54.50) | |
| Rich | 545 (36.08) | 298 (54.68) | 247 (45.32) | |
| Level of woman’s education | 0.013 | |||
| None | 960 (63.56) | 468 (48.75) | 492 (51.25) | |
| Primary | 305 (20.17) | 148 (48.52) | 157 (51.48) | |
| Secondary and above | 246 (16.27) | 145 (58.94) | 101 (41.06) | |
| Religion | <0.001 | |||
| Muslim | 1317 (87.22) | 632 (48.00) | 685 (52.00) | |
| Animist/No religion | 30 (2.01) | 19 (63.33) | 11 (36.70) | |
| Christian | 163 (10.77) | 109 (66.87) | 54 (33.13) | |
| Age of women (years) | 0.681 | |||
| Under 20 | 206 (13.61) | 108 (52.43) | 98 (47.57) | |
| 20–34 | 1,096 (72.61) | 544 (49.64) | 552 (50.36) | |
| 35–49 | 208 (13.78) | 107 (51.44) | 101 (48.56) | |
| Relationship status with head of household | 0.035 | |||
| Wife | 1,022 (67.68) | 491 (48.04) | 531 (51.96) | |
| Other relationship | 150 (9.94) | 81 (54.00) | 69 (46.00) | |
| Daughter | 338 (22.38) | 188 (55.62) | 150 (44.38) | |
| Frequency of reading newspaper | 0.047 | |||
| Not at all | 1,419 (93.94) | 706 (49.75) | 713 (50.25) | |
| Less than once a week | 47 (3.10) | 23 (48.89) | 24 (51.11) | |
| At least once a week | 45 (2.96) | 31 (68.89) | 14 (31.11) | |
| Frequency of listening to radio | 0.046 | |||
| Not at all | 784 (51.93) | 393 (50.13) | 391 (49.87) | |
| Less than once a week | 392 (25.96) | 181 (46.17) | 211 (53.83) | |
| At least once a week | 334 (22.11) | 185 (55.39) | 149 (44.61) | |
| Frequency of watching television | 0.208 | |||
| Not at all | 837 (55.42) | 421 (50.30) | 416 (49.70) | |
| Less than once a week | 281 (18.6) | 130 (46.26) | 151 (53.74) | |
| At least once a week | 392 (25.9) | 209 (53.31) | 183 (46.69) | |
Table 1: Sociodemographic characteristics of participants according to the coverage of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15 to 49 whose last live birth occurred within the past two years before the survey, malaria and anemia indicators survey, Guinea, 2021
Table 2: Distribution of Obstetric history of women whose last live birth occurred within two years prior to the study period according to the number of intermittent preventive treatment doses received, Guinea Malaria and Anemia Indicators Survey, 2021
| Variables | Frequency n (%) | Women who received at least three doses of IPTp-SP | p-value | |
|---|---|---|---|---|
| Yes n (%) | No n (%) | |||
| Total participants | 1,511 | 760 (50.30) | 751 (49.70) | – |
| Parity | 0.038 | |||
| Multiparous | 895 (59.23) | 432 (48.26) | 463 (51.74) | |
| Grand multiparous | 287 (18.99) | 142 (49.47) | 145 (50.53) | |
| Primiparous | 329 (21.78) | 186 (56.53) | 143 (43.46) | |
| Profile of the person who performed the antenatal care | <0.001 | |||
| Not a health personnel | 83 (5.50) | 14 (16.87) | 69 (83.13) | |
| Health personnel qualified for Antenatal Care | 1,009 (66.78) | 560 (55.50) | 449 (44.50) | |
| Health personnel not qualified for Antenatal Care | 419 (27.72) | 186 (44.39) | 233 (55.61) | |
| Status of antenatal care visit | <0.001 | |||
| No antenatal care | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| Antenatal care within the first trimester | 557 (36.86) | 330 (59.25) | 227 (40.75) | |
| Antenatal care beyond the first trimester | 870 (57.61) | 416 (47.82) | 454 (52.18) | |
| Number of ANC visits | <0.001 | |||
| No antenatal care visit | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| 1–3 antenatal care visit | 530 (35.08) | 177 (33.40) | 353 (66.60) | |
| At least 4 antenatal care visit | 897 (59.39) | 569 (63.43) | 328 (36.57) | |
| Place of ANC | <0.001 | |||
| No antenatal care visit | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| Unofficial health facilities | 12 (0.82) | 5 (41.67) | 7 (58.33) | |
| Official health facilities | 1,414 (93.65) | 741 (52.40) | 673 (47.60) | |
Table 2: Distribution of Obstetric history of women whose last live birth occurred within two years prior to the study period according to the number of intermittent preventive treatment doses received, Guinea Malaria and Anemia Indicators survey, 2021
Table 3: Distribution of factors independently associated (bivariate analysis) with uptake of at least three doses of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15–49 whose last live birth occurred within the past two years before the study. Malaria and Anemia Indicators Survey, Guinea, 2021
| Variables | Uptake of Sulfadoxine-Pyrimethamine during pregnancy | Crude Prevalence Ratio | 95% CI | p-value | ||
|---|---|---|---|---|---|---|
| < 3 doses n (%) | ≥ 3 doses n (%) | Lower | Upper | |||
| Region | ||||||
| Boké | 108 (58.70) | 76 (41.30) | 2.41 | 1.38 | 4.22 | 0.002 |
| Conakry | 88 (45.83) | 104 (54.17) | 2.26 | 1.21 | 4.21 | 0.011 |
| Faranah | 58 (40.85) | 85 (59.85) | 1.18 | 0.70 | 2.01 | 0.532 |
| Kankan | 185 (61.95) | 113 (38.05) | 2.59 | 1.56 | 4.28 | 0.001 |
| Kindia | 108 (46.35) | 125 (53.65) | 1.71 | 0.96 | 3.03 | 0.067 |
| Labé | 69 (53.49) | 60 (46.51) | 2.28 | 1.20 | 4.36 | 0.012 |
| Mamou | 50 (54.95) | 40 (43.95) | 2.26 | 1.15 | 4.44 | 0.018 |
| N’Zérékoré | 84 (34.85) | 157 (65.15) | 1.00 | – | – | – |
| Place of residence | ||||||
| Rural | 573 (52.14) | 526 (47.86) | 1.58 | 1.10 | 2.65 | 0.048 |
| Urban | 177 (43.07) | 234 (56.93) | 1.00 | – | – | – |
| Sex of the head of household | ||||||
| Male | 666 (50.35) | 657 (49.65) | 1.05 | 0.72 | 1.51 | 0.067 |
| Female | 84 (44.92) | 103 (55.08) | 1.00 | – | – | – |
| Age groups of head of household (years) | ||||||
| <26 | 19 (50.00) | 19 (50.00) | 1.14 | 0.52 | 2.49 | 0.743 |
| 35-44 | 233 (53.20) | 205 (46.80) | 1.32 | 0.95 | 1.83 | 0.102 |
| 45-54 | 145 (49.00) | 151 (51.00) | 1.34 | 0.91 | 1.96 | 0.140 |
| 55-95 | 188 (48.00) | 204 (52.00) | 1.40 | 0.92 | 2.12 | 0.115 |
| 26-34 | 165 (47.60) | 182 (52.40) | 1.00 | – | – | – |
| Parity | ||||||
| Multiparous | 463 (51.74) | 432 (48.26) | 1.35 | 0.97 | 1.88 | 0.080 |
| Grand multiparous | 145 (50.53) | 142 (49.47) | 1.33 | 0.83 | 2.13 | 0.233 |
| Primiparous | 143 (43.46) | 186 (56.53) | 1.00 | – | – | – |
| Profile of ANC provider | ||||||
| Not a health personnel | 69 (83.13) | 14 (16.87) | 9.65 | 5.16 | 18.02 | <0.001 |
| Not qualified | 233 (55.61) | 186 (44.39) | 1.22 | 0.93 | 1.61 | 0.147 |
| Qualified | 449 (44.50) | 560 (55.50) | 1.00 | – | – | – |
Table 3: Distribution of factors independently associated (bivariate analysis) with uptake of at least three doses of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15–49 whose last live birth occurred within the past two years before the study. Malaria and Anemia Indicators Survey, Guinea, 2021
Table 4: Factors independently associated (multivariate analysis) with uptake of less than three doses of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15–49 whose last live birth occurred within the past two years before the study, Malaria and Anemia Indicators Survey, Guinea, 2021
| Variables | Parameter | Adjusted Prevalence Ratio | 95% CI | p-value | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| Socio-demographic characteristics | |||||
| Region | |||||
| Boké | 2.43 | 1.46 | 4.06 | <0.001 | |
| Conakry | 2.29 | 1.37 | 3.84 | 0.002 | |
| Faranah | 1.15 | 0.69 | 1.91 | 0.588 | |
| Kankan | 2.22 | 1.39 | 3.55 | <0.001 | |
| Kindia | 1.75 | 1.03 | 2.97 | 0.039 | |
| Labé | 2.34 | 1.27 | 4.31 | 0.006 | |
| Mamou | 2.38 | 1.25 | 4.54 | 0.009 | |
| N’Zérékoré (Ref) | 1.00 | – | – | – | |
| Frequency of reading newspaper | |||||
| Not at all | 1.69 | 0.87 | 3.28 | 0.120 | |
| Less than once a week | 2.61 | 1.07 | 6.35 | 0.035 | |
| At least once a week (Ref) | 1.00 | – | – | – | |
| Obstetric history | |||||
| Profile of the person who performed the antenatal | |||||
| Not a health personnel | 9.59 | 5.20 | 17.70 | <0.001 | |
| Health personnel not qualified for Antenatal Care | 1.28 | 0.99 | 1.66 | 0.062 | |
| Health personnel qualified for Antenatal Care (Ref) | 1.00 | – | – | – | |
| Number of antenatal care visits | |||||
| Less than 4 antenatal care visits | 3.55 | 2.77 | 4.54 | <0.001 | |
| At least 4 antenatal care visits (Ref) | 1.00 | – | – | – | |
Table 4: Factors independently associated (multivariate analysis) with uptake of less than three doses of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15–49 whose last live birth occurred within the past two years before the study, Malaria and Anemia Indicators Survey, Guinea, 2021
Figures

Keywords
- Intermittent preventive treatment
- Malaria
- Coverage
- Associated factors
- Guinea
Niouma Nestor Leno1,2,3*, Sidikiba Sidibé1,2, Aboubacar Sidiki Cherif4, Mohamed Fara Mara1, Djiki Camara1, Tamba Gaston Kambadouno1, Aissata Kolonga Bangoura1, Timothé Guilavogui1, Alioune Camara1,2,5
1Faculty of Health Sciences and Techniques, Gamal Abdel Nasser University of Conakry, Conakry, Guinea, 2African Center of Excellence for the Prevention and Control of Communicable Diseases (CEA-PCMT), Gamal Abdel Nasser University of Conakry, Conakry, Guinea, 3Expanded Program on Immunization, Ministry of Health and Public Hygiene, Conakry, Guinea, 4National Institute of Statistics, Ministry of Planning and Cooperation, Conakry, Guinea, 5National Malaria Control Program, Ministry of Health and Public Hygiene, Conakry, Guinea
&Corresponding author: Niouma Nestor Leno, Gamal Adbel Nasser University of Conakry, Guinea, Email: nnleno81@gmail.com, ORCID: https://orcid.org/0000-0002-2863-2267
Received: 25 Nov 2025, Accepted: 20 Mar 2026, Published: 23 Mar 2026
Domain: Maternal and Child Health, Infecious Disease Epidemiology
Keywords: Intermittent preventive treatment, malaria, coverage, associated factors, Guinea
©Niouma Nestor Leno et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Niouma Nestor Leno et al., Coverage and determinants of intermittent preventive treatment for malaria in pregnancy with sulfadoxine-pyrimethamine in Guinea, 2021. Journal of Interventional Epidemiology and Public Health. 2026; 9(1):49. https://doi.org/10.37432/jieph-d-25-00304
Abstract
Introduction: In Guinea, malaria continues to pose a major threat to maternal and child health, with an estimated national prevalence of 17%. Intermittent Preventive Treatment with Sulfadoxine-Pyrimethamine (IPTp-SP) is a priority intervention in the 2023–2027 strategic plan, only a limited proportion of pregnant women receive the recommended three or more doses. This study assessed IPTp-SP coverage and identified factors associated with not completing IPTp-SP treatment.
Methods: This analytical cross-sectional study, conducted from September 12 to October 5, 2025, used data from the 2021 Guinea Malaria and Anemia Indicators Survey among women aged 15–49 years with a live birth in the preceding two years. Data were analyzed using IBM SPSS Statistics 27. Weighted descriptive statistics described participant characteristics, and logistic regression identified determinants of not completing IPTp-SP treatment using adjusted Prevalence Ratio (aPR), with statistical significance defined by a p-value <0.05 and 95% confidence intervals excluding 1.
Results: The IPTp-SP-3+ overall coverage was 50.3%, 95% CI [47.8 % – 52.8%] and varied from 37.9%, 95% CI [35.8% – 39.9%] in the region of Kankan to 65.1% [63.3% – 66.9%] in the region of Nzérékoré. Not performing antenatal care with a qualified health professional (aPR = 9.59; 95%CI: 5.20–17.70, less than four antenatal care (ANC) visits (aPR=3.55, 95% CI: 2.77–4.54), and not reading newspaper weekly (aPR = 2.61; 95% CI: 1.07–6.35) were independently associated with not completing IPTp-SP treatment.
Conclusion: IPTp-SP coverage in Guinea remains below the 90% target and is influenced by antenatal care quality, visit frequency, and regional disparities. Improving outcomes requires strengthening the availability of qualified health personnel at ANC facilities, enhancing health education to promote timely attendance and adherence, and reinforcing community mobilization through active engagement of local leaders to increase awareness and uptake of antenatal services.
Introduction
Malaria continues to pose a significant global health challenge, with the heaviest toll borne by sub-Saharan Africa. The disease remains a leading cause of illness and death, particularly among vulnerable populations such as pregnant women and young children[1]. In 2023, an estimated 263 million malaria cases and approximately 597,000 malaria-related deaths were reported worldwide. The WHO African Region bears most of this burden, accounting for nearly 95% of global malaria cases and deaths, which underscores the disproportionate impact of the disease on populations across the continent[2,3]. Pregnant women are disproportionately affected, as pregnancy reduces immunity and increases susceptibility to severe malaria, maternal anemia, miscarriage, and adverse birth outcomes including low birth weight, preterm birth, stillbirth, and neonatal mortality [4–6].
To mitigate these risks, WHO recommends intermittent preventive treatment during pregnancy with sulfadoxine-pyrimethamine (IPTp-SP) as a cornerstone intervention in areas with moderate to high transmission of Malaria. This strategy has been shown to reduce complications for both mother and child [6,7]. Across sub-Saharan Africa, IPTp-SP is incorporated into national malaria control strategies and delivered through antenatal care (ANC) service from the second trimester of the pregnancy. Nevertheless, coverage remains uneven. While some countries have achieved notable improvements, particularly in reaching the target of three or more doses, others continue to face persistent obstacles such as inequities in access, health system weaknesses, drug supply interruptions, and inconsistent quality of ANC services [8–10]. Recognizing these challenges, the updated 2022 WHO recommendations emphasized initiation of IPTp-SP as early as possible in the second trimester, with doses administered at least monthly until delivery, and encouraged the involvement of community-level providers to extend access beyond formal ANC [6,11].
In Guinea, malaria continues to be a major contributor to maternal and child morbidity and mortality, with an estimated national prevalence of 17% among children under five years of age (aged 6–59 months) [12]. In health facilities in Guinea, 2,411,407 malaria cases were reported in 2022. In addition, malaria accounted for 12.8% of proportional mortality in health facilities across the country during the same year[13]. Transmission is intense across most of the country, with peaks during the rainy season. Plasmodium falciparum is the predominant species, accounting for more than 95% of malaria cases in Guinea. Other species, such as Plasmodium malariae and Plasmodium ovale, are also present but remain very rare [14]. The maternal mortality ratio, estimated at around 553 deaths per 100,000 live births in 2020, underscores the significant role of preventable causes such as malaria [15]. Intermittent Preventive Treatment with Sulfadoxine-Pyrimethamine (IPTp-SP) has been adopted by the National Malaria Control Programme (NMCP) as part of its maternal health strategy, and recent policy adjustments have aligned national protocols with WHO guidance. The national target is to achieve 90% coverage of pregnant women receiving at least three doses of sulfadoxine–pyrimethamine during pregnancy for malaria prevention. This preventive treatment targets pregnant women, regardless of their malaria parasitemia status, starting from the second trimester of pregnancy [16]. In Guinea, the 2012 and 2018 Demographic and Health Surveys (DHS) show that coverage of antenatal care remains relatively low (81%). In addition, these surveys reveal suboptimal uptake of IPTp. Consequently, only a limited proportion of pregnant women receive the full set of recommended doses[17]. For instance, a study across nine district hospitals reported that nearly 58% of parturients had received fewer than three doses of SP, and just 24% benefited from comprehensive malaria prevention combining insecticide-treated net use with full SP dosing [18].
Multiple factors have been linked to inadequate IPTp-SP uptake. Coverage of at least three doses varies considerably across the African Region, from less than 20% in some countries to over than 60–80% in others, reflecting differences in health infrastructure, financing, and programme performance [9,10,19]. Determinants identified in previous studies include socio-demographic characteristics (maternal age, education, parity, socio-economic status, and place of residence), ANC attendance patterns (timing and number of visits), as well as health system issues such as SP stock-outs, provider practices, clarity of guidelines, and community-level awareness of malaria risk [8,18,20].
Guinea malaria control program strategic 2023 – 2027 planned to achieve the WHO and national targets. The Guinean NMCP strategic plan 2023–2027 set to achieve the objective of at least 90% of pregnant women at risk receiving at least three IPTp-SP doses during the pregnancy[21]. To ensure the effectiveness of a program, it is essential to regularly collect data on the progress made toward achieving its objectives, as well as on factors that may explain suboptimal performance. In this context, in 2021, the National Malaria Control Program of Guinea, in collaboration with the National Institute of Statistics, conducted the “Guinea Malaria and Anemia Indicator Survey” (GMAIS). This survey described the coverage of intermittent preventive treatment for malaria among pregnant women but did not sufficiently explore the determinants of its use.
Understanding the factors influencing treatment completion is a strategic tool for health programs: it enables the adjustment of existing interventions and the identification of targeted actions to be implemented in a timely manner, thereby achieving set objectives and avoiding unsatisfactory outcomes at the end of implementation periods. The study objectives were to determine the IPTp-SP coverage and factors associated with not completing IPTp-SP treatment among women aged 15 – 49 whose last live birth occurred within two years prior to the study period. The findings will help identify gaps, inform programme adjustments, and guide policy decisions to strengthen malaria prevention in pregnancy, reduce disparities, and improve maternal, perinatal, and neonatal health outcomes in the country.
Methods
Study setting
Guinea is one of the West African littoral countries sharing borders in the north, east and west with six countries and the Atlantic Ocean in the south. It has eight health regions, and Conakry, the capital, has a special status. Each region is led by a governor; seven regions are divided into 33 prefectures, while Conakry is divided into communes. The country’s population was estimated at 14,363,931 inhabitants in 2025, of whom 25% were women of reproductive age (15–49 years). In Guinea, health outcomes remain poor: maternal mortality is 650 per 100,000 live births, and child mortality rates, though declining, remain high[22]. The country has 38 health districts, including 33 in the interior of the country and 5 in Conakry. Malaria prevalence in children dropped from 44% in 2012 to 17% in 2021, but it is still the leading cause of medical consultations in health facilities and over than one-third of hospital deaths [23].
In Guinea, IPTp-SP is primarily delivered through routine antenatal care (ANC) services in public health facilities. SP is administered under directly observed therapy (DOT) during ANC visits to ensure that the tablets are taken in the presence of a health worker. Pregnant women receive SP starting from the second trimester (from 13 weeks of gestation) and at each subsequent ANC visit, with doses spaced at least one month apart, in accordance with WHO recommendations.
SP is generally provided free of charge as part of the national malaria prevention strategy. Health facilities are supplied through the national pharmaceutical and malaria control supply system, coordinated by the National Malaria Control Program (NMCP). We have incorporated these clarifications into the revised manuscript to improve readers’ understanding of the IPTp-SP delivery context in Guinea [9,19,21].
Study design
This analytical cross-sectional study, conducted from September 12 to October 5, 2025, used data from the 2021 Guinea Malaria and Anemia Indicators Survey among women aged 15–49 years with a live birth in the preceding two years.
Population and data source
This study included women aged 15–49 years who were living in the selected households or who had been present there for at least one day prior to the survey date, and whose last live birth occurred no more than two years before the survey. The data were extracted from the 2021 Survey on Malaria and Anemia Indicators in Guinea. Field data collection for the primary survey was conducted from July 13 to September 10, 2021. Furthermore, the extraction of data from the primary database and its analysis for the present study were carried out between July 12 and August 10, 2025. The study also included the households in which the women selected for the analysis were residing.
Sampling
This study was a secondary data analysis of the 2021 Guinea Malaria and Anemia Indicator Survey (GMAIS). This survey provided nationally, regionally, and urban–rural representative data. The country was divided into 15 survey domains across eight regions, with urban and rural strata (except Conakry) [24]. Using a stratified, two-stage cluster design, 169 Primary Sampling Units were selected from 2014 census Enumeration Areas via probability proportional to size. Household listings created sampling frames, and 24 households per cluster were systematically selected, totalling 4,056 households included in the survey.
Definition of variables
The dependent variable in this study is the coverage of Intermittent Preventive Treatment with Sulfadoxine-Pyrimethamine (IPTp-SP) for malaria among pregnant women. It is defined as the percentage of women aged 15–49 years who had a live birth in the two years preceding the survey and received at least three doses of sulfadoxine–pyrimethamine for malaria prevention during pregnancy.
The independent variables included women’s sociodemographic characteristics (region, place of residence, sex, age, education level, religion, frequency of newspaper reading, frequency of radio listening and frequency of watching television), household wealth level, and obstetric history (parity, person consulted during ANC, timing of first antenatal visit, number of ANC visits and place of ANC).
Data processing and analysis
The data used in this study were obtained from the Guinea Malaria and Anemia Indicator Survey 2021 primary database. The final validated version of the dataset, stored in Excel format, was used for the analysis. Initial data quality checks, including the identification and correction of inconsistencies, outliers, and missing values, had already been conducted by the National Institute of Statistics (INS) during the management of the primary database.
For this secondary analysis, selected variables were recoded and transformed to align with the study objectives. Variables of interest were identified, and when necessary, derived variables were generated. The dependent variable and explanatory variables were defined and categorized according to the study’s analytical framework. After these data preparation steps, the final analytical dataset was exported to IBM SPSS Statistics version 27 for statistical analysis. Weighted descriptive statistics summarized participants’ sociodemographic and clinical characteristics.
The dependent variable used to identify factors associated with not completing IPTp-SP treatment was dichotomised as follows:
- Completed IPTp-S treatment: women who received at least three doses of sulfadoxine–pyrimethamine for malaria prevention during pregnancy, coded as “0”.
- Not completed IPTp-SP treatment: women who received fewer than three doses of sulfadoxine–pyrimethamine for malaria prevention during pregnancy, coded as “1.”
Logistic regression was used to identify the factors associated with not completing IPTp-SP treatment (receipt of fewer than three doses). The measure of association calculated was the prevalence ratio. In this regression process, first, a bivariate logistic regression analysis was conducted to identify candidate variables for inclusion in the multivariable model. Variables included in the bivariate analysis were selected based on evidence from the literature regarding factors associated with incomplete uptake of intermittent preventive treatment for malaria during pregnancy, as well as variables relevant to the Guinean context. The crude prevalence ratio (cPR) was used as the measure of association. Variables with a p-value ≤ 0.20 in the bivariate analysis were retained for inclusion in the multivariable analysis.
Subsequently, a multivariable logistic regression model was fitted using a backward stepwise selection procedure to identify independent factors associated with non-completion of IPTp-SP treatment. The adjusted prevalence ratio (aPR) was used to estimate the strength of the associations. Multicollinearity among explanatory variables was assessed using the Variance Inflation Factor (VIF). Variables were considered statistically significant determinants if their aPR differed from 1, the 95% confidence interval (CI) excluded 1, and the p-value was < 0.05.
Ethical consideration
The baseline study, carried out by the National Institute of Statistics for the external evaluation of Guinea’s National Malaria Control Program, received ethical approval (N° 123/CNERS/21). Participation was voluntary, with informed consent obtained after a clear explanation of the study objectives. Authorization for the secondary data analysis was granted by both the National Malaria Control Program and the Institute. All data were fully anonymized, ensuring the confidentiality and protection of participants throughout the analysis process.
Results
For this study, 4,056 households were selected, of which 4,023 were occupied. Among these occupied households, 3,985 were successfully interviewed. Within these households, 1,512 women aged 15–49 years whose last live birth occurred no more than two years prior to the survey were selected for the study. Households from rural areas accounted for 66.32% (95% CI: 65.18%–67.42%) of the interviewed households.
The overall median age was 27 years (20–35 years) and varied from 25 years (17 – 32 years) in rural area to 30 years (22 – 37 years) in urban area. The overall percentage of male heads of households was 87.29%, 95% CI [84.69%-89.89%] and varied from 92.34%, 95% CI [89.34%-95.34%] in rural area to 83.23%, 95% CI [80.83%-85.63%] in urban area (Table 1). Participants from rural area represented 72.78%, 95% CI [70.22% – 73.36%]. Overall, 63.56%, 95% CI [62.31%–63.89%) of participants had no formal education. Based on the wealth index, nearly half of the participants (43.16%, 95% CI [42.52% – 44.41%]) came from poor households, compared with 36.08%, 95% CI [35.56% – 37.22%] from wealthy or well-off households. In this study, 87.22%, 95% CI [86.43% – 88.21%] of participants were Muslim, while 10.77%, 95% CI [10.01% – 11.22%] were Christian. This study shows that 93.94%, 95% CI [92.67% – 94.35%] of participants did not read newspapers, 51.94%, 95% CI [49.54% – 52.03%] did not listen to the radio, and 55.41% did not watch television (Table 1).
Regarding the obstetric history of the women included in this study, 21.78%, 95% CI [20.28%–23.11%] were primiparous, compared with 18.99%, 95% CI [16.89%–20.23%] who were grand multiparous. We also observed that 5.53%, 95% CI [5.01%–6.13%] of the women had not attended any antenatal care visits. Women who attended at least four antenatal care visits accounted for 59.39%, 95% CI [58.45%–60.26%] of the sample. Furthermore, 93.65%, 95% CI [92.29%–94.37%] of the women received antenatal care in formal health facilities, and 66.78%, 95% CI [64.36%–67.43%] were attended by qualified health professionals (Table 2).
Coverage of intermittent preventive treatment with sulfadoxine-pyrimethamine (IPTp-SP)
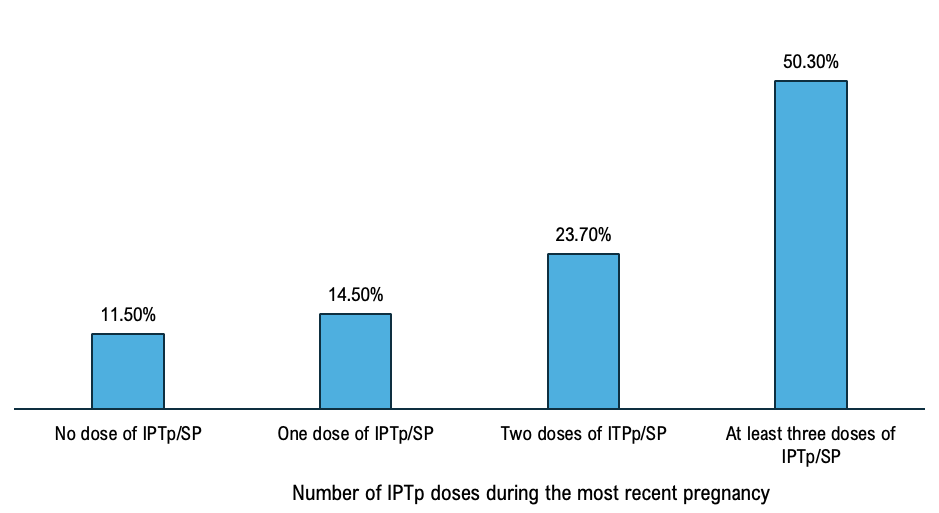
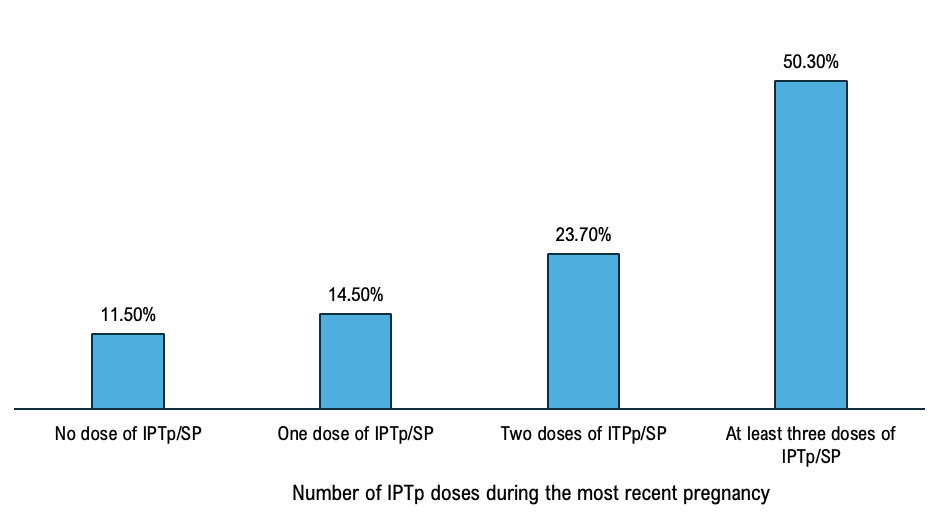
The overall complete IPTp-SP coverage represented 50.30%, 95% CI [48.10%- 251 52.50%] and not completing IPTp-SP treatment was 49.70%, 95% CI [47.40%-50.00%]. Among women who participated, 23.70%, 95% CI [22.2% – 25.20%] received two doses, 14.50% 95% CI 253 [13.4%-15.6%] received one dose and 11.50%, CI [10.4%12.1%] (Figure 1).
The IPTp-SP coverage varied from 38.05%, 95% CI [35.8% – 39.9%] in the region of Kankan to 65.15% [63.31% – 66.92%] in the region of Nzérékoré. Complete IPTp-SP rate was 47.86%, 95% CI [46.26% – 48.47%] in rural area and 56.93%, 95% CI [55.60% – 57.12%] in urban area. Male heads of households accounted for 87.64%, 95% CI [86.80% – 89.31%], and IPTp-SP coverage was 49.65%, 95% CI [48.56% – 50.21%] in households led by a man while it was 55.08%, 95% CI [53.67% – 55.87%] in those led by a woman (Table 1). The coverage of intermittent preventive treatment for malaria among pregnant women varied according to the wealth status of the surveyed women. It was 48.91% (95% CI [47.23%–49.56%]) among women from poor households, 45.50% (95% CI [44.30%–46.11%]) among those from middle-income households, and 54.68% (95% CI [53.22%–55.34%]) among women from wealthy households. In this study, we found that the coverage of intermittent preventive treatment for malaria among pregnant women with no formal education was 48.75%, 95% CI [ 47.38%–49.66%], and 58.94%, 95% CI [57.67% – 59.17%] (Table 1).
IPTp-SP coverage varied by religious affiliation. It was 66.9%, 95% CI [65.91%–67.02%] among Christian women and 48.0%, 95% CI [47.38%–49.17%] among Muslim women (Table 1). Considering the parity, the ITP-SP was completed in 56.5%, 95% CI [53.2% – 59.8%] primiparous, 49.7%, 95% CI [48.1%-51.3%] in grand multiparous, 48.3%, 95% CI [46.9% – 49.7%] in multiparous. Among women who consulted a qualified health personnel during antenatal care visits, those who completed the ITP-SP were 55.5%, 95% CI [52.4%-58.6%] while they were 16.9%, 95% CI [15.3%-18.5%] among those who consulted a non-qualified health personnel. It is important to note that, in the field of maternal health, qualified health personnel refer to trained and authorized health professionals who provide obstetric and antenatal care in accordance with national standards and the recommendations of the World Health Organization (WHO). This category includes physicians, midwives, nurses or obstetric nurses, as well as qualified medical assistants or health technicians. Within women who performed at least four ANC visits, completed ITP-SP treatment was 63.4%,95% CI [61.3%-65.3%] whereas among those who performed not more than three and no ANC visit, it was 35.08%, 95% CI [33.38%-36.78%] and 5.53%, 95% CI [3.73%-7.33%] respectively (Table 2).
Factors associated with non-completion of intermittent preventive treatment for malaria in pregnancy (IPTp-SP) bivariate analysis
The bivariate analysis highlights significant disparities in the coverage of intermittent preventive treatment with sulfadoxine–pyrimethamine (IPTp-SP) among pregnant women in Guinea, according to regions and characteristics of antenatal care. The proportion of women who received at least three doses of IPTp-SP was highest in the N’Zérékoré region compared with other regions of the country. Compared with this reference region, significantly lower coverage was observed among women living in Kankan (PR = 2.59; 95% CI: 1.56–4.28), Boké (PR = 2.41; 95% CI: 1.38–4.22), Conakry (PR = 2.26; 95% CI: 1.21–4.21), as well as Labé (PR = 2.28; 95% CI: 1.20–4.36), and Mamou (PR = 2.26; 95% CI: 1.15–4.44). Place of residence was also an associated factor, with women living in rural areas having lower coverage than those in urban areas (PR = 1.58; 95% CI: 1.10–2.65). The strongest associations, however, were related to antenatal care (ANC). Women who did not attend any ANC had extremely low IPTp-SP coverage (16.9%) and a significantly higher risk of receiving fewer than three doses (PR = 3.12; 95% CI: 2.81–5.24). Similarly, receiving ANC from unqualified personnel (PR = 9.65; 95% CI: 5.16–18.02) and having an insufficient number of ANC visits were strongly associated with low coverage. In contrast, sociodemographic characteristics such as maternal age, education level, household wealth index, and parity were not significantly associated with IPTp-SP coverage in this bivariate analysis (Table 3).
Multivariate analysis
The multivariate analysis identified several factors significantly associated with not completing IPTp-SP treatment among pregnant women in Guinea (Table 4). Compared with N’Zérékoré, women living in Boké (aPR = 2.43; 95% CI: 1.456 – 4.061; p<0.001), Conakry (aPR = 2.29; 95% CI: 1.368 – 3.836; p=0.002), Kankan (aPR = 2.22; 95% CI: 1.392 – 3.548; p<0.001), Labé (aPR = 2.34; 95% CI: 1.270 – 4.308; p=0.006), Mamou (aPR = 2.38; 95% CI: 1.245 – 4.539; p=0.009), and Kindia (aPR = 1.75; 95% CI: 1.029 – 2.970; p=0.039) had significantly higher Prevalence Ratio of not completing IPTp-SP treatment. Women who did not consult any health worker for antenatal care missed IPTp-SP doses nearly ten times (aPR = 9.59; 95% CI: 5.20–17.70; p< 0.001) more than those followed up by qualified health personnel. Women who had less than four antenatal visits had 3.55 times higher odds of non-completion of IPTp-SP doses (95% CI: 2.77–4.54; p < 0.001) compared with those who had at least four antenatal visits. In addition, women who did not read newspapers had higher odds of not completing IPTp-SP treatment (aPR = 2.61; 95% CI: 1.07–6.35; p = 0.035) than those who read newspapers at least once per week (Table 4).
Discussion
This study assessed the coverage of IPTp-SP and the factors associated with its non-completion among pregnant women in Guinea, using data from the 2021 Guinea Malaria and Anemia Indicator Survey (GMAIS). Only slightly more than half of women received the recommended minimum of three doses of IPTp-SP, underscoring persistent gaps in malaria prevention relative to the national target of 90%[21]. Factors associated with non-completion of IPTp-SP treatment included regional disparities, healthcare access, and media exposure. The observed IPTp-SP coverage is higher than many past national and multi-country estimates but remains suboptimal. WHO recommends at least three-monthly doses starting in the second trimester, yet uptake across sub-Saharan Africa is variable [5]. Compared to countries like Cameroon, where coverage reaches 97.7% [25], Guinea’s coverage of IPTp-SP is low, indicating the need to adopt successful strategies from higher-performing settings. Multi-country analyses show substantial heterogeneity in IPTp-SP coverage and suggest that strengthening community-based delivery strategies can significantly improve uptake where facility-based delivery alone is insufficient. [10,26].
The partial coverage categories (two doses, one dose, and no dose) observed in this study, reflects a common pattern in Africa, where many women initiate but fail to complete the regimen. Consistent with previous studies [26,27], not completing IPTp-SP treatment, may be associated with late initiation of ANC and missed opportunities during ANC contacts; SP stock-outs were not directly assessed in this study.
Socio-demographic and health-system determinants such as education, wealth, and place of residence further influence adherence. High education and frequent media consultation have been associated with higher IPTp-SP uptake, while poverty and late ANC booking predict lower use [27,28]. Interventions targeting these determinants, such as promoting early ANC visit, enhancing media outreach, community-Based strategy (sensitization for pregnant women for early ANC visit, active research and distribution of SP to pregnant women with not completing IPTp-SP tretment and strengthening health systems, are critical to improving IPTp-SP coverage.
Recent evidence suggests several programmatic actions to improve coverage: strengthening ANC quality and ensuring consistent SP availability at the point of care; promoting early and regular ANC attendance; expanding community-based distribution (C-IPTp) to reach women in remote areas; and enhancing behaviour-change communication, particularly among younger and less-educated women. These interventions are aligned with WHO’s 2023 recommendations [5,20,26], and have shown success in multi-country evaluations [20,26].
The multivariate analysis of this study revealed significant regional disparities in IPTp-SP completion in Guinea. Regional variations were notable: women in Boké, Conakry, Kankan, Labé, Mamou, and Kindia had higher odds of not completing IPTp-SP treatment compared to those in N’Zérékoré. Differences in healthcare infrastructure, accessibility, and regional health policies are likely to contribute to this pattern . Addressing disparities requires region-specific strategies, including strengthening healthcare systems, improving service availability, and adapting health policies to local contexts to enhance IPTp-SP coverage nationwide.
Access to qualified ANC providers and visit frequency were the strongest determinants of IPTp-SP completion. Women with no ANC visits or fewer than four visits had 9.6- and 3.5-fold higher odds, respectively, of not completing IPTp-SP tretment. Similar findings have been reported in other studies conducted in sub-Saharan Africa[29,30]. However, ANC attendance alone is insufficient; the qualifications of healthcare staff and facility readiness are critical. Studies in Cameroon and Ghana showed that being attended by non-qualified providers or visiting under-resourced facilities markedly reduced IPTp uptake[30,31]. Strengthening provider training, supportive supervision, and uninterrupted drug supply are essential to addressing these system-level bottlenecks.
Health-system readiness assessments across multiple African countries also reveal missed opportunities, eligible women attending ANC but not receiving SP due to provider oversight or stock-outs[32]. Combining client-side demand creation with provider-side readiness is crucial for improving IPTp-SP uptake. Innovative strategies, such as mobile ANC units, community health worker outreach, and transport vouchers, can address geographic and financial barriers, especially in rural areas. Media exposure strongly affects IPTp-SP completion; women with limited access to malaria-related messages—via print, radio, or television—are less likely to complete three or more doses. Evidence from sub-Saharan Africa shows regular mass media exposure enhances awareness, adherence, and correct use. Policies should diversify and strengthen communication channels, using local languages and multiple platforms, to reach women with lower literacy or restricted access and promote full uptake of preventive treatment during pregnancy [27,33,34].
Strengths and limitations of the study
This study’s strength lies in using recent, nationally representative data with standardized measures, allowing generalizable findings and robust multivariable analysis of factors affecting IPTp-SP coverage. Stratified analyses highlighted geographic and sociodemographic disparities, while examining ANC attendance and media exposure provided insights into behavioral and health system influences. This study has some limitations. The cross-sectional design precludes establishing temporal or causal relationships. As a secondary data analysis, the study relied on pre-existing variables, limiting control variable definition. Self-reported measures may be subject to memory bias. Moreover, the study did not include qualitative interviews to explore reasons for non-coverage of intermittent preventive treatment for malaria. Due to the cross-sectional design of the study, the results were interpreted as associative factors rather than causal relationships. During primary data collection, recall bias was minimized through the use of standardized questionnaires, enhanced training of data collectors to reduce interviewer influence, and the preferential use of health service records to complete data collection forms. Despite this, findings are valuable for guiding Guinea’s National Malaria Control Program in improving ANC services, addressing regional gaps, and enhancing health education for pregnant women.
Conclusion
Despite national policy commitment and integration of IPTp-SP into Guinea’s malaria program, coverage among pregnant women remains low, with only half receiving the recommended three or more doses. Regional disparities and limited access to qualified ANC providers reveal ongoing inequities in service delivery. Strengthening ANC attendance and addressing sociocultural and informational barriers may help improve IPTp-SP uptake; community-based delivery approaches could be explored based on existing evidence. Achieving the 2023–2027 strategic plan targets requires coordinated efforts among national programs, local health systems, and communities. Future research on health system readiness, supply chain reliability, community interventions, and adherence will inform strategies to reduce maternal and neonatal malaria morbidity and mortality and reinforce Guinea’s malaria prevention program.
What is already known about the topic
- Malaria remains a major public health challenge, particularly affecting pregnant women and children
- Intermittent preventive treatment with sulfadoxine-pyrimethamine (at least three doses during pregnancy) is a key intervention to reduce malaria-related maternal morbidity and mortality
- However, coverage remains insufficient in many sub-Saharan African countries, ranging from 20% to 80%, considerably below the target of over 90%.This low uptake is influenced by socio-economic factors, such as education level, place of residence, and wealth, as well as health system determinants, including access to care, quality of services, and the availability of resources and medications
- In Guinea, despite gradual progress, the coverage of intermittent preventive treatment among pregnant women remains below the 90% target.
What this study adds
- Residing in a region other than Nzérékoré (an inland region of Guinea) is associated with a higher risk of non-completion of intermittent preventive treatment for malaria among pregnant women, which in turn indirectly increases the risk of severe malaria. These findings highlight the need to develop targeted urban strategies, particularly in large cities such as Conakry, where coverage of intermittent preventive treatment is expected to be higher due to better availability of health services
- This study highlights significant progress in Guinea’s coverage of intermittent preventive treatment for malaria among pregnant women. The proportion of women who received at least three doses of sulfadoxine–pyrimethamine increased steadily, rising from 3% in 2005 to 11% in 2012, then to 36% in 2018, and reaching 50.3% in 2021. Despite this encouraging trend, coverage remains below the established national target
- These findings provide key insights to guide future planning, strengthen interventions, and support decision-making in malaria control efforts in Guinea.
Acknowledgements
The authors express their deep gratitude to the National Malaria Control Program and the National Institute of Statistics of Guinea for their valuable support and for providing access to the primary data. They also warmly thank all the women who agreed to participate in the primary study.
| Variables | Frequency n (%) | Women who received at least three doses of IPTp-SP | p-value | |
|---|---|---|---|---|
| Yes n (%) | No n (%) | |||
| Total | 1,511 | 760 (50.30) | 751 (49.70) | |
| Region | <0.001 | |||
| Boké | 184 (12.2) | 76 (41.30) | 108 (58.70) | |
| Conakry | 192 (12.71) | 104 (54.17) | 88 (45.83) | |
| Faranah | 142 (9.42) | 85 (59.85) | 58 (40.85) | |
| Kankan | 297 (19.68) | 113 (38.05) | 185 (61.95) | |
| Kindia | 233 (15.44) | 125 (53.65) | 108 (46.35) | |
| Labé | 129 (8.55) | 60 (46.51) | 69 (53.49) | |
| Mamou | 91 (6.02) | 40 (43.95) | 50 (54.95) | |
| N’Zérékoré | 241 (15.98) | 157 (65.15) | 84 (34.85) | |
| Place of residence | 0.002 | |||
| Rural | 1,099 (72.78) | 526 (47.86) | 573 (52.14) | |
| Urban | 411 (27.22) | 234 (56.93) | 177 (43.07) | |
| Sex of the head of household | 0.165 | |||
| Male | 1,323 (87.64) | 657 (49.65) | 666 (50.35) | |
| Female | 187 (12.36) | 103 (55.08) | 84 (44.92) | |
| Age of the head of household (years) | 0.506 | |||
| <26 | 38 (2.50) | 19 (50.00) | 19 (50.00) | |
| 26-34 | 347 (22.99) | 182 (52.40) | 165 (47.60) | |
| 35-44 | 437 (28.95) | 205 (46.80) | 233 (53.20) | |
| 45-54 | 296 (19.58) | 151 (51.00) | 145 (49.00) | |
| 55+ | 392 (25.98) | 204 (52.00) | 188 (48.00) | |
| Household size | 0.173 | |||
| Medium | 626 (41.45) | 312 (49.84) | 314 (50.16) | |
| Large | 506 (33.53) | 270 (53.36) | 236 (46.64) | |
| Small | 378 (25.02) | 178 (47.10) | 200 (52.90) | |
| Household wealth index | 0.021 | |||
| Poor | 652 (43.16) | 319 (48.91) | 333 (51.07) | |
| Middle | 314 (20.76) | 143 (45.50) | 171 (54.50) | |
| Rich | 545 (36.08) | 298 (54.68) | 247 (45.32) | |
| Level of woman’s education | 0.013 | |||
| None | 960 (63.56) | 468 (48.75) | 492 (51.25) | |
| Primary | 305 (20.17) | 148 (48.52) | 157 (51.48) | |
| Secondary and above | 246 (16.27) | 145 (58.94) | 101 (41.06) | |
| Religion | <0.001 | |||
| Muslim | 1317 (87.22) | 632 (48.00) | 685 (52.00) | |
| Animist/No religion | 30 (2.01) | 19 (63.33) | 11 (36.70) | |
| Christian | 163 (10.77) | 109 (66.87) | 54 (33.13) | |
| Age of women (years) | 0.681 | |||
| Under 20 | 206 (13.61) | 108 (52.43) | 98 (47.57) | |
| 20–34 | 1,096 (72.61) | 544 (49.64) | 552 (50.36) | |
| 35–49 | 208 (13.78) | 107 (51.44) | 101 (48.56) | |
| Relationship status with head of household | 0.035 | |||
| Wife | 1,022 (67.68) | 491 (48.04) | 531 (51.96) | |
| Other relationship | 150 (9.94) | 81 (54.00) | 69 (46.00) | |
| Daughter | 338 (22.38) | 188 (55.62) | 150 (44.38) | |
| Frequency of reading newspaper | 0.047 | |||
| Not at all | 1,419 (93.94) | 706 (49.75) | 713 (50.25) | |
| Less than once a week | 47 (3.10) | 23 (48.89) | 24 (51.11) | |
| At least once a week | 45 (2.96) | 31 (68.89) | 14 (31.11) | |
| Frequency of listening to radio | 0.046 | |||
| Not at all | 784 (51.93) | 393 (50.13) | 391 (49.87) | |
| Less than once a week | 392 (25.96) | 181 (46.17) | 211 (53.83) | |
| At least once a week | 334 (22.11) | 185 (55.39) | 149 (44.61) | |
| Frequency of watching television | 0.208 | |||
| Not at all | 837 (55.42) | 421 (50.30) | 416 (49.70) | |
| Less than once a week | 281 (18.6) | 130 (46.26) | 151 (53.74) | |
| At least once a week | 392 (25.9) | 209 (53.31) | 183 (46.69) | |
| Variables | Frequency n (%) | Women who received at least three doses of IPTp-SP | p-value | |
|---|---|---|---|---|
| Yes n (%) | No n (%) | |||
| Total participants | 1,511 | 760 (50.30) | 751 (49.70) | – |
| Parity | 0.038 | |||
| Multiparous | 895 (59.23) | 432 (48.26) | 463 (51.74) | |
| Grand multiparous | 287 (18.99) | 142 (49.47) | 145 (50.53) | |
| Primiparous | 329 (21.78) | 186 (56.53) | 143 (43.46) | |
| Profile of the person who performed the antenatal care | <0.001 | |||
| Not a health personnel | 83 (5.50) | 14 (16.87) | 69 (83.13) | |
| Health personnel qualified for Antenatal Care | 1,009 (66.78) | 560 (55.50) | 449 (44.50) | |
| Health personnel not qualified for Antenatal Care | 419 (27.72) | 186 (44.39) | 233 (55.61) | |
| Status of antenatal care visit | <0.001 | |||
| No antenatal care | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| Antenatal care within the first trimester | 557 (36.86) | 330 (59.25) | 227 (40.75) | |
| Antenatal care beyond the first trimester | 870 (57.61) | 416 (47.82) | 454 (52.18) | |
| Number of ANC visits | <0.001 | |||
| No antenatal care visit | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| 1–3 antenatal care visit | 530 (35.08) | 177 (33.40) | 353 (66.60) | |
| At least 4 antenatal care visit | 897 (59.39) | 569 (63.43) | 328 (36.57) | |
| Place of ANC | <0.001 | |||
| No antenatal care visit | 83 (5.53) | 14 (16.87) | 69 (83.13) | |
| Unofficial health facilities | 12 (0.82) | 5 (41.67) | 7 (58.33) | |
| Official health facilities | 1,414 (93.65) | 741 (52.40) | 673 (47.60) | |
| Variables | Uptake of Sulfadoxine-Pyrimethamine during pregnancy | Crude Prevalence Ratio | 95% CI | p-value | ||
|---|---|---|---|---|---|---|
| < 3 doses n (%) | ≥ 3 doses n (%) | Lower | Upper | |||
| Region | ||||||
| Boké | 108 (58.70) | 76 (41.30) | 2.41 | 1.38 | 4.22 | 0.002 |
| Conakry | 88 (45.83) | 104 (54.17) | 2.26 | 1.21 | 4.21 | 0.011 |
| Faranah | 58 (40.85) | 85 (59.85) | 1.18 | 0.70 | 2.01 | 0.532 |
| Kankan | 185 (61.95) | 113 (38.05) | 2.59 | 1.56 | 4.28 | 0.001 |
| Kindia | 108 (46.35) | 125 (53.65) | 1.71 | 0.96 | 3.03 | 0.067 |
| Labé | 69 (53.49) | 60 (46.51) | 2.28 | 1.20 | 4.36 | 0.012 |
| Mamou | 50 (54.95) | 40 (43.95) | 2.26 | 1.15 | 4.44 | 0.018 |
| N’Zérékoré | 84 (34.85) | 157 (65.15) | 1.00 | – | – | – |
| Place of residence | ||||||
| Rural | 573 (52.14) | 526 (47.86) | 1.58 | 1.10 | 2.65 | 0.048 |
| Urban | 177 (43.07) | 234 (56.93) | 1.00 | – | – | – |
| Sex of the head of household | ||||||
| Male | 666 (50.35) | 657 (49.65) | 1.05 | 0.72 | 1.51 | 0.067 |
| Female | 84 (44.92) | 103 (55.08) | 1.00 | – | – | – |
| Age groups of head of household (years) | ||||||
| <26 | 19 (50.00) | 19 (50.00) | 1.14 | 0.52 | 2.49 | 0.743 |
| 35-44 | 233 (53.20) | 205 (46.80) | 1.32 | 0.95 | 1.83 | 0.102 |
| 45-54 | 145 (49.00) | 151 (51.00) | 1.34 | 0.91 | 1.96 | 0.140 |
| 55-95 | 188 (48.00) | 204 (52.00) | 1.40 | 0.92 | 2.12 | 0.115 |
| 26-34 | 165 (47.60) | 182 (52.40) | 1.00 | – | – | – |
| Parity | ||||||
| Multiparous | 463 (51.74) | 432 (48.26) | 1.35 | 0.97 | 1.88 | 0.080 |
| Grand multiparous | 145 (50.53) | 142 (49.47) | 1.33 | 0.83 | 2.13 | 0.233 |
| Primiparous | 143 (43.46) | 186 (56.53) | 1.00 | – | – | – |
| Profile of ANC provider | ||||||
| Not a health personnel | 69 (83.13) | 14 (16.87) | 9.65 | 5.16 | 18.02 | <0.001 |
| Not qualified | 233 (55.61) | 186 (44.39) | 1.22 | 0.93 | 1.61 | 0.147 |
| Qualified | 449 (44.50) | 560 (55.50) | 1.00 | – | – | – |
Table 4: Factors independently associated (multivariate analysis) with uptake of less than three doses of intermittent preventive treatment with Sulfadoxine-Pyrimethamine among women aged 15–49 whose last live birth occurred within the past two years before the study, Malaria and Anemia Indicators Survey, Guinea, 2021
| Variables | Parameter | Adjusted Prevalence Ratio | 95% CI | p-value | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| Socio-demographic characteristics | |||||
| Region | |||||
| Boké | 2.43 | 1.46 | 4.06 | <0.001 | |
| Conakry | 2.29 | 1.37 | 3.84 | 0.002 | |
| Faranah | 1.15 | 0.69 | 1.91 | 0.588 | |
| Kankan | 2.22 | 1.39 | 3.55 | <0.001 | |
| Kindia | 1.75 | 1.03 | 2.97 | 0.039 | |
| Labé | 2.34 | 1.27 | 4.31 | 0.006 | |
| Mamou | 2.38 | 1.25 | 4.54 | 0.009 | |
| N’Zérékoré (Ref) | 1.00 | – | – | – | |
| Frequency of reading newspaper | |||||
| Not at all | 1.69 | 0.87 | 3.28 | 0.120 | |
| Less than once a week | 2.61 | 1.07 | 6.35 | 0.035 | |
| At least once a week (Ref) | 1.00 | – | – | – | |
| Obstetric history | |||||
| Profile of the person who performed the antenatal | |||||
| Not a health personnel | 9.59 | 5.20 | 17.70 | <0.001 | |
| Health personnel not qualified for Antenatal Care | 1.28 | 0.99 | 1.66 | 0.062 | |
| Health personnel qualified for Antenatal Care (Ref) | 1.00 | – | – | – | |
| Number of antenatal care visits | |||||
| Less than 4 antenatal care visits | 3.55 | 2.77 | 4.54 | <0.001 | |
| At least 4 antenatal care visits (Ref) | 1.00 | – | – | – | |
References
- Simbeye AJ, Kumwenda S, Cohee LM, Omondi D, Masibo PK, Wao H, Awandu SS. Factors associated with malaria vaccine uptake in Nsanje district, Malawi. Malar J [Internet]. 2024 Apr 17 [cited 2026 Mar 23]; 23(1):105. doi:10.1186/s12936-024-04938-7 Available from: https://doi.org/10.1186/s12936-024-04938-7
- World Health Organization (WHO). World malaria report 2024. Addressing inequity in the global malaria response [Internet]. Geneva (Switzerland): World Health Organization; 2024 [cited 2026 Mar 23]; 293 p. Available from: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2024
- World Health Organization. Community health workers can help to fill the gap in malaria prevention for pregnant women [Internet]. Geneva (Switzerland): World Health Organization; 2023 Apr 14 [cited 2026 Mar 23]. [about 5 screens]. Available from: https://www.who.int/news-room/feature-stories/detail/community-health-workers-can-help-to-fill-the-gap-in-malaria-prevention-for-pregnant-women
- World Health Organization. Malaria [Internet]. Geneva (Switzerland): World Health Organization; 2025 Dec 4 [cited 2026 Mar 23]. [about 5 screens]. Available from: https://www.who.int/news-room/fact-sheets/detail/malaria
- World Health Organization. World malaria report 2022 [Internet]. Geneva (Switzerland): World Health Organization; 2022 Dec 8 [cited 2026 Mar 23]. 293 p. Available from: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2022
- World Health Organization. Intermittent preventative treatment to reduce the risk of malaria during pregnancy [Internet]. WHO (ELENA). 2023 [cited 2026 Mar 23]. Available from: https://www.who.int/tools/elena/interventions/iptp-pregnancy
- World Health Organization. Updated WHO recommendations for malaria chemoprevention among children and pregnant women [Internet]. World Health Organization. 2022 Aug 9 [cited 2026 Mar 23]. [about 1 screen]. Available from: https://www.who.int/news/item/03-06-2022-Updated-WHO-recommendations-for-malaria-chemoprevention-among-children-and-pregnant-women
- Olapeju B, Bride M, Gutman JR, Wolf K, Wabwire S, Atobrah D, Babanawo F, Akrofi OO, Atta-Obeng C, Soro BK, Touré F, Shekarau E, Hendrickson ZM. WHO antenatal care policy and prevention of malaria in pregnancy in sub-Saharan Africa. Malar J [Internet]. 2024 Jul 23 [cited 2026 Mar 23];23(1):218. doi:10.1186/s12936-024-05037-3 Available from: https://doi.org/10.1186/s12936-024-05037-3
- Thiam S, Kimotho V, Gatonga P. Why are IPTp coverage targets so elusive in sub-Saharan Africa? A systematic review of health system barriers. Malar J [Internet]. 2013 Oct 3 [cited 2026 Mar 23];12(1):353. doi:10.1186/1475-2875-12-353 Available from: https://doi.org/10.1186/1475-2875-12-353
- Kretchy IA, Atobrah D, Adumbire DA, Ankamah S, Adanu T, Badasu DM, Kwansa BK. Enhancing the uptake of intermittent preventive treatment for malaria in pregnancy: a scoping review of interventions and gender-informed approaches. Malar J [Internet]. 2025 Feb 18 [cited 2026 Mar 23];24(1):49. doi:10.1186/s12936-025-05275-z Available from: https://doi.org/10.1186/s12936-025-05275-z
- Pons-Duran C, Llach M, Sacoor C, Sanz S, Macete E, Arikpo I, Ramírez M, Meremikwu M, Mbombo Ndombe D, Méndez S, Manun’Ebo MF, Ramananjato R, Rabeza VR, Tholandi M, Roman E, Pagnoni F, González R, Menéndez C. Coverage of intermittent preventive treatment of malaria in pregnancy in four sub-Saharan countries: findings from household surveys. Int J Epidemiol [Internet]. 2021 May 17 [cited 2026 Mar 23];50(2):550–9. doi:10.1093/ije/dyaa233 Available from: https://doi.org/10.1093/ije/dyaa233
- Yaya S, Uthman O, Amouzou A, Bishwajit G. Use of Intermittent Preventive Treatment among Pregnant Women in Sub-Saharan Africa: Evidence from Malaria Indicator Surveys. TropicalMed [Internet]. 2018 Feb 11 [cited 2026 Mar 23];3(1):18. doi:10.3390/tropicalmed3010018 Available from: https://doi.org/10.3390/tropicalmed3010018
- Institut national de la Statistique (Guinée), ICF. Enquête sur les indicateurs du paludisme et de l’anémie en Guinée 2021 [Survey on malaria and anemia indicators in Guinea 2021] [Internet]. Rockville (Maryland): ICF; 2021 [cited 2026 Mar 23]. 150 p. Available from: https://www.stat-guinee.org/images/Documents/Publications/INS/rapports_enquetes/Rapport_final_EIPAG_2021.pdf
- Ministère de la Santé et de l’Hygiène Publique (Guinée). Annuaire statistique sanitaire 2022 [Health Statistics Yearbook 2022]. Conakry (Guinée); 2023 [cited 2026 Mar 23]. 140 p. Available from: https://www.stat-guinee.org/images/Documents/Publications/SSN/sante/Annuaire_statistique_sante_2022.pdf
- Severe Malaria Observatory. Guinea: Malaria Facts [Internet]. Geneva (Switzerland): Severe Malaria Observatory; 2024 [cited 2026 Mar 23]. [about 6 screens]. Available from: https://www.severemalaria.org/countries/guinea
- Unicef. Maternal mortality [Internet]. New York (NY): Unicef; 2025 Apr [cited 2026 Mar 23]. [about 3 screens]. Available from: https://data.unicef.org/topic/maternal-health/maternal-mortality/
- Barry I, Toure AA, Sangho O, Beavogui AH, Cisse D, Diallo A, Magassouba AS, Sylla Y, Doumbia L, Cherif MS, Camara AY, Diawara F, Tounkara M, Delamou A, Doumbia S. Variations in the use of malaria preventive measures among pregnant women in Guinea: a secondary analysis of the 2012 and 2018 demographic and health surveys. Malar J [Internet]. 2022 Nov 1 [cited 2026 Mar 23];21(1):309. doi:10.1186/s12936-022-04322-3 Available from: https://doi.org/10.1186/s12936-022-04322-3
- Diallo A, Touré AA, Doumbouya A, Magassouba AS, Traoré F, Cissé M, Barry I, Conté I, Cissé D, Cissé A, Camara G, Bérété AO, Camara AY, Conté NY, Beavogui AH. Factors Associated with Malaria Preventive Measures among Pregnant Women in Guinea. Infect Dis Obstet Gynecol [Internet]. 2021 Jul 1 [cited 2026 Mar 23];2021:1–9. doi:10.1155/2021/9914424 Available from: https://doi.org/10.1155/2021/9914424
- World Health Organization. Community deployment of intermittent preventive treatment of malaria in pregnancy with sulfadoxine-pyrimethamine: a field guide [Internet]. Geneva (Switzerland): World Health Organization; 2024 Jan 16 [cited 2026 Mar 23]; 57 p. Available from: https://www.who.int/publications/i/item/9789240086272
- Medicine for Malaria Venture. Malaria continues to strike pregnant women and children hardest and new tools are critical, according to WHO’s latest report [Internet]. Geneva (Switzerland): Medicine for Malaria Venture; 2019 Oct 5 [cited 2026 Mar 23]. [about 2 screens]. Available from: https://www.mmv.org/newsroom/news-resources-search/malaria-continues-strike-pregnant-women-and-children-hardest-and-new
- Koita K, Kayentao K, Worrall E, Van Eijk AM, Hill J. Community-based strategies to increase coverage of intermittent preventive treatment of malaria in pregnancy with sulfadoxine–pyrimethamine in sub-Saharan Africa: a systematic review, meta-analysis, meta-ethnography, and economic assessment. Lancet Glob Health [Internet]. 2024 Sep [cited 2026 Mar 23];12(9):e1456–69. doi:10.1016/S2214-109X(24)00228-6 Available from: https://doi.org/10.1016/S2214-109X(24)00228-6
- Programme National de Lutte Contre le Paludisme/Ministère de la Santé et de l’Hygiène Publique (Guinée). Plan Stratégique National de la Santé Communautaire 2023-2027 [National Strategic Plan for Community Health 2023-2027] [Internet]. Conakry (Guinée); 2023 Feb [cited 2026 Mar 23]. 101 p. Available from: https://p4h.world/app/uploads/2024/09/PSNSC-2023-2027_Guinee.x69485.pdf
- Institut National de la Statistique (Guinée), ICF. 2018. Enquête Démographique et de Santé en Guinée 2018 [2018 Demographic and Health Survey in Guinea]. Conakry, (Guinée): Institut National de la Statistique; 2019 Jul [cited 2026 Mar 23]. 609 p. Available from: https://dhsprogram.com/pubs/pdf/FR353/FR353.pdf
- Institut National de la Statistique (Guinée). Des statistiques fiables pour la prise de décision [Reliable statistics for decision-making] [Internet]. Conakry, (Guinée): Institut National de la Statistique; 2023 [cited 2026 Mar 23]. Available from: https://www.stat-guinee.org/
- Ministère du Plan et la Coopération Internationale (Guinée), Institut National de la Statistique (Guinée). Recensement Général de la Population et de l’habitat – 3 (RGPH-3), 2014 [General Population and Housing Census – 3 (RGPH-3), 2014] [Internet]. Conakry (Guinée): Institut National de la Statistique; 2027 Dec [cited 2026 Mar 23]. 192 p. Available from: https://dataspace.princeton.edu/handle/88435/dsp01pk02cd514
- Nishan MDNH, Akter K. Coverage and determinants of Intermittent Preventive Treatment in pregnancy (IPTp) in Cameroon, Guinea, Mali, and Nigeria. PLoS ONE [Internet]. 2024 Nov 1 [cited 2026 Mar 23];19(11):e0313087. doi:10.1371/journal.pone.0313087 Available from: https://doi.org/10.1371/journal.pone.0313087
- González R, Manun’Ebo MF, Meremikwu M, Rabeza VR, Sacoor C, Figueroa-Romero A, Arikpo I, Macete E, Mbombo Ndombe D, Ramananjato R, LIach M, Pons-Duran C, Sanz S, Ramírez M, Cirera L, Maly C, Roman E, Pagnoni F, Menéndez C. The impact of community delivery of intermittent preventive treatment of malaria in pregnancy on its coverage in four sub-Saharan African countries (Democratic Republic of the Congo, Madagascar, Mozambique, and Nigeria): a quasi-experimental multicentre evaluation. Lancet Glob Health [Internet]. 2023 Apr [cited 2026 Mar 23];11(4):e566–74. doi:10.1016/S2214-109X(23)00051-7 Available from: https://doi.org/10.1016/S2214-109X(23)00051-7
- Darteh EKM, Dickson KS, Ahinkorah BO, Owusu BA, Okyere J, Salihu T, Bio Bediako V, Budu E, Agbemavi W, Edjah JO, Seidu AA. Factors influencing the uptake of intermittent preventive treatment among pregnant women in sub-Saharan Africa: a multilevel analysis. Arch Public Health [Internet]. 2021 Oct 21 [cited 2026 Mar 23];79(1):182. doi:10.1186/s13690-021-00707-z Available from: https://doi.org/10.1186/s13690-021-00707-z
- Ameyaw EK. Uptake of intermittent preventive treatment of malaria in pregnancy using sulfadoxine-pyrimethamine (IPTp-SP) in Uganda: a national survey. Malar J [Internet]. 2022 Oct 7 [cited 2026 Mar 23];21(1):285. doi:10.1186/s12936-022-04299-z Available from: https://doi.org/10.1186/s12936-022-04299-z
- Apanga PA, Kumbeni MT, Chanase MAW. The Association Between Early Antenatal Care and Intermittent Preventive Treatment of Malaria in Pregnancy in Sub-Saharan Africa: Effect Modification by Planned Pregnancy Status. Ann Glob Health [Internet]. 2022 Jan 10 [cited 2026 Mar 23];88(1):4. doi:10.5334/aogh.3550 Available from: https://doi.org/10.5334/aogh.3550
- Asem L, Abdulia AG, Assuming PO, Abeka-Nkrumah G. The demand for intermittent preventive treatment of malaria in pregnancy using sulfadoxine-pyrimethamine in the Volta Region of Ghana. PLoS ONE [Internet]. 2024 Sep 5 [cited 2026 Mar 23];19(9):e0308321. doi:10.1371/journal.pone.0308321 Available from: https://doi.org/10.1371/journal.pone.0308321
- Guimsop DK, Talla AFK, Kodji H, Ateudjieu J. Factors associated with the uptake of intermittent preventive treatment for malaria during pregnancy in Cameroon: An analysis of data from the 2018 Cameroon Demographic and Health Survey. PLOS Glob Public Health [Internet]. 2024 Mar 27 [cited 2026 Mar 23];4(3):e0001245. doi:10.1371/journal.pgph.0001245 Available from: https://doi.org/10.1371/journal.pgph.0001245
- Xu X, Liang D, Zhao J, Mpembeni R, Olenja J, Yam EL, Huang J. The readiness of malaria services and uptake of intermittent preventive treatment in pregnancy in six sub-Saharan countries. J Glob Health [Internet]. 2024 Jun 28 [cited 2026 Mar 23];14:04112. doi:10.7189/jogh.14.04112 Available from: https://doi.org/10.7189/jogh.14.04112
- Yaya S, Uthman OA, Amouzou A, Bishwajit G. Mass media exposure and its impact on malaria prevention behaviour among adult women in sub-Saharan Africa: results from malaria indicator surveys. Glob Health Res Policy [Internet]. 2018 Jul 4 [cited 2026 Mar 23];3(1):20. doi:10.1186/s41256-018-0075-x Available from: https://doi.org/10.1186/s41256-018-0075-x
- De-Gaulle VF, Magnussen P, Kamgno J, Mbacham W, Orish VN, Tagbor H. Assessing health system factors affecting access and delivery of IPTp-SP and ITN to pregnant women attending ANC clinics in Ghana. BMC Health Serv Res [Internet]. 2021 Oct 5 [cited 2026 Mar 23];21(1):1056. doi:10.1186/s12913-021-07055-2 Available from: https://doi.org/10.1186/s12913-021-07055-2
