Outbreak Investigation | Open Access | Volume 9 (Suppl 13): Article 01 | Published: 23 Apr 2026
Yellow fever outbreak investigation potentially linked to zoonotic spill over in Eastern Uganda, April-July 2024
Menu, Tables and Figures
On Pubmed
Navigate this article
Tables
| Variable | Frequency (N=5) | Percentage |
|---|---|---|
| Sex | ||
| Female | 4 | 80.0 |
| Male | 1 | 20.0 |
| Age | ||
| 0-12 years | 2 | 40.0 |
| 13-17 years | 0 | 0.0 |
| 18-34 years | 1 | 20.0 |
| 35+ years | 2 | 40.0 |
| Subcounty of residence | ||
| Kapir | 1 | 20.0 |
| Ngora Town Council | 4 | 80.0 |
| Note: N = total number of cases (5). Percentages may not sum to 100 due to rounding. | ||
Table 1: Distribution of the cases by sex, age and area of residence
| Variable | Population at risk | Number of cases | Attack rate/100,000 |
|---|---|---|---|
| Sex | |||
| Female | 97,038 | 4 | 4.1 |
| Male | 116,739 | 1 | 0.9 |
| Subcounty of residence | |||
| Kapir | 29,658 | 1 | 3.4 |
| Ngora Town Council | 21,488 | 4 | 18.6 |
| Note: Attack rates calculated per 100,000 population. Population at risk based on subcounty estimates. | |||
Table 2: Yellow fever attack rates, stratified by sex and subcounty of residence
| Sub county | Target population | Number vaccinated | Coverage (%) |
|---|---|---|---|
| Ngora SC | 21,027 | 11,200 | 53.3 |
| Ngora Town council | 21,488 | 24,078 | 112.1 |
| Kapir | 29,658 | 17,121 | 57.7 |
| Kobuin | 20,775 | 14,159 | 68.2 |
| Mukura | 24,477 | 17,383 | 71.0 |
| Note: Coverage exceeding 100% may indicate inclusion of individuals from outside the target population or population movement. | |||
Table 3: Proportion of the target population vaccinated against yellow fever, by subcounty
| Variable | Frequency (N=4) | Percentage |
|---|---|---|
| Attended funeral before becoming ill | ||
| Yes | 2 | 50.0 |
| No | 2 | 50.0 |
| Travelled outside home village before becoming ill | ||
| Yes | 2 | 50.0 |
| No | 2 | 50.0 |
| Patient hospitalized before illness | ||
| Yes | 1 | 25.0 |
| No | 3 | 75.0 |
| Direct contact with animals | ||
| Yes | 3 | 75.0 |
| No | 1 | 25.0 |
| Note: N = total number of cases (4). Percentages may not sum to 100 due to rounding. | ||
Table 4: Proportion of cases reporting possible exposures to yellow fever
Figures
Keywords
- Yellow fever
- Uganda
- Outbreak
- Zoonosis
- Vaccination
Alex Mulyowa1,&, Cathelyne Kobumanzi1, Robert Mwesigwa1, Precious Ninsiima1, Irene Nakaziba1, Mary Ajumo1, Connie Nait1, Wilson Tusiime1, Bernard Lubwama2, Herbert Kazoora3, Suzanne Kiwanuka1
1Department of Health Policy, Planning and Management, School of Public Health, Makerere University, Kampala, Uganda, 2Division of Integrated Epidemiology and Surveillance, Ministry of Health, Kampala, Uganda, 3African Field Epidemiology Network, Kampala, Uganda
&Corresponding author: Alex Mulyowa, Department of Health Policy, Planning and Management, School of Public Health, Makerere University, Kampala, Uganda, Email: alexandermulyowa@gmail.com ORCID: https://orcid.org/0000-0003-3627-7637
Received: 29 Sep 2025, Accepted: 15 Apr 2026, Published: 23 Apr 2026
Domain: Field Epidemiology, Outbreak Investigation
Keywords: Yellow fever, Uganda, outbreak, zoonosis, vaccination
©Alex Mulyowa et al. Journal of Interventional Epidemiology and Public Health (ISSN: 2664-2824). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Alex Mulyowa et al. Yellow fever outbreak investigation potentially linked to zoonotic spill over in Eastern Uganda, April-July 2024. Journal of Interventional Epidemiology and Public Health. 2026; 9(Suppl 13):01. https://doi.org/10.37432/jieph-d-25-00211
Abstract
Introduction: Uganda has reported numerous yellow fever outbreaks in the past decade. Despite the rollout of control interventions such as vaccination, recurrent outbreaks continue to occur. This paper describes the yellow fever outbreak investigation in Ngora district, Uganda.
Methods: This was a descriptive outbreak investigation. We defined a suspected case of yellow fever as a resident or visitor of Ngora district who reported onset of unexplained fever (negative for malaria, or non-responsive to malaria treatment, and not explained by any other known reasons) from April to July 2024, plus two or more symptoms such as unexplained jaundice, abdominal pain, or headache that could not be explained by other causes. A confirmed case was a suspected case with laboratory confirmation of yellow fever-specific neutralising antibodies using the plaque reduction neutralisation test. Confirmed cases were identified through a review of records of twenty suspected cases reported between April and July 2024 at the sentinel surveillance site. A standardized case investigation form was used to collect information on exposures. We computed proportions to describe characteristics of the cases, and reviewed records to determine vaccination coverage in the district. Data were analysed using STATA v16.
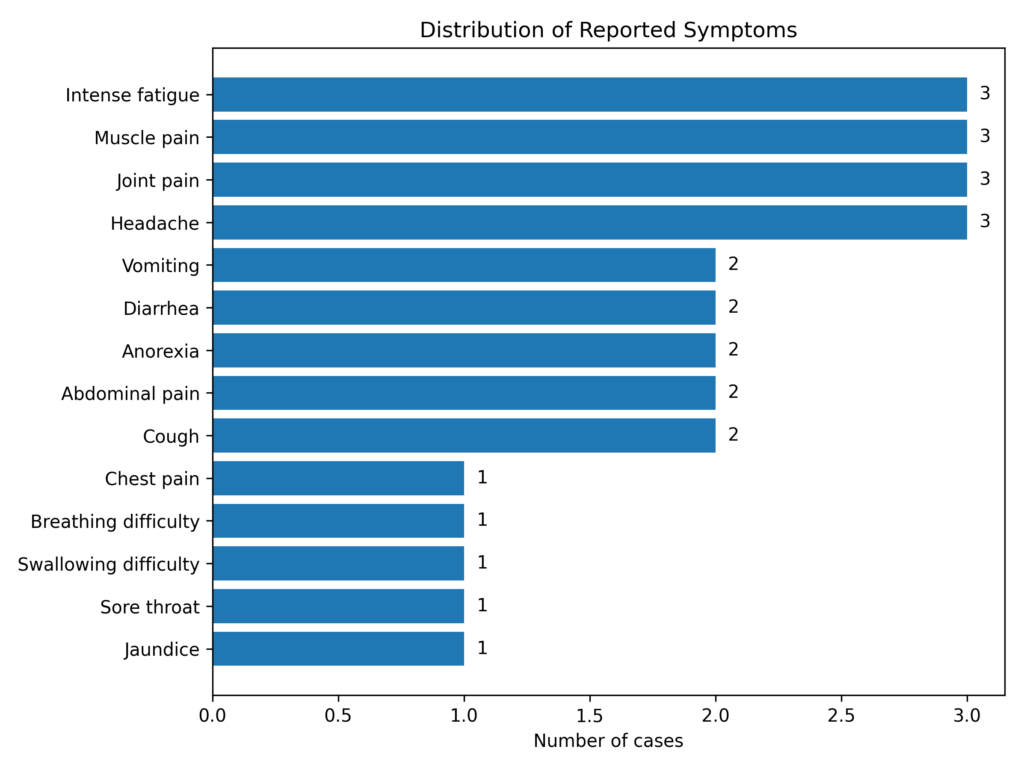
Results: Five confirmed cases of yellow fever were identified. Four were female, with an age range of 5-49 years. The fifth case resulted in a fatality; thus, no data was collected. One case had a previous history of vaccination. Fatigue (75%, n=3), muscle and joint pain (75%, n=3) and headache (75%, n=3) were the most commonly reported signs and symptoms. Three cases (75%) reported previous contact with animals, among which were monkeys.
Conclusion: This yellow fever outbreak was potentially linked to a zoonotic spillover. This study highlights the urgent need for targeted one health interventions, such as integrated vector control, aimed at controlling yellow fever and other zoonoses within the district.
Introduction
Yellow fever is a mosquito-borne arboviral illness, endemic within tropical regions of Africa and South America [1]. The illness has continuously re-emerged as a major global public health threat [2], transmitted through three cycles: sylvatic, urban, and intermediate [3]. It is reported largely among the unvaccinated population, with a total caseload of about 200,000 clinical cases and 30,000 deaths every year [4]. The risk of unvaccinated travellers contracting yellow fever during a two-week stay in a yellow fever endemic area is fifty cases per 100,000 population [5]. In Africa, yellow fever is the third most commonly reported type of disease outbreak, with an epidemic alert threshold of one case [6].
Uganda is a high-risk country in the African yellow fever belt [7]. The country has had at least one outbreak per year over the last four years [7]. The incidence rates have ranged from 3 to 13 cases per 100,000 population, with a 33% case fatality rate [8]. Previous studies have attributed the recent outbreaks to farming within or around forested areas [8, 9], climate change [10], and human interaction with monkeys [8]. Yet, even with the availability of safe and effective control interventions such as vaccination, yellow fever outbreaks continue to occur across the country [11]. Available evidence suggests that perhaps the sustained outbreaks could be because the control strategies are confined to only the affected districts and nearby areas, yet the disease is a nationwide burden [11]. Moreover, even with the mass national vaccination campaigns, it has been argued that this reactive approach initiated in response to outbreaks may not necessarily prevent future disease outbreaks [11]. This is plausible, given the numerous transmission dynamics such as mosquito population density, climate change, and human migration patterns, which may influence the risk of outbreaks, even in highly vaccinated areas [12]. Besides, there could also be a likelihood that herd immunity remains low, thus perpetuating a continuous cycle of outbreaks [13].
Ngora district is one of the 134 rural districts located in Eastern Uganda. The district population has more females than males (110,226 vs. 103,360), with an average household size of 7 persons, about 23% of whom are female-headed households [14]. Ngora is a district that has never reported any previous outbreak of yellow fever. However, the district remains at high risk of recrudescence, given its rocky terrain, erratic, unreliable rainfall patterns and partly forested vegetation cover – characteristics that allow for transmission of the yellow fever flavivirus among the local population. It is therefore not surprising that on 20th April 2024, two cases of yellow fever were detected in the district. On April 30th, 2024, one more case was detected, and by May 19th, 2024, an additional case had been detected. Samples from these cases were collected and taken to the arbovirology laboratory of the Uganda Virus Research Institute (UVRI). On June 17th, 2024, the Ministry of Health received a report from UVRI indicating that four samples had tested positive for yellow fever antibodies through the plaque reduction neutralisation test (PRNT). A yellow fever outbreak was declared in the district, and an outbreak investigation was instituted on 1st July, 2024. On July 5th, one yellow fever case fatality (detected on 18th June, 2024) was recorded. We conducted a case investigation to describe the characteristics of the yellow fever outbreak in Ngora district, Uganda.
Methods
Study setting
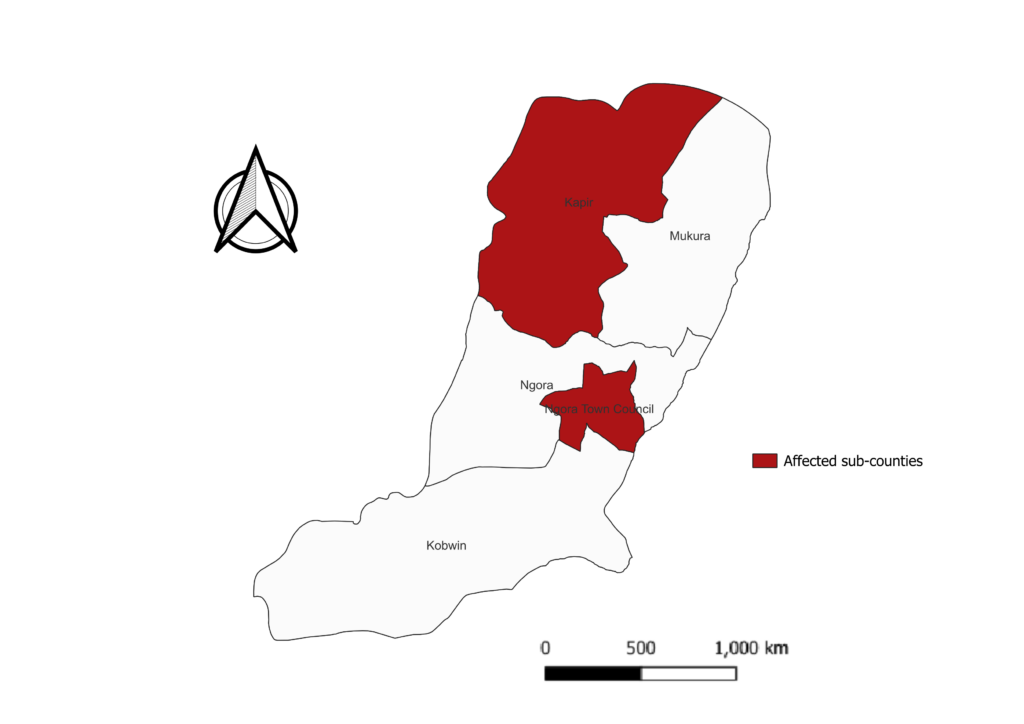
The study was conducted in Ngora district. Ngora District is located in the Teso sub-region, in the Eastern part of Uganda. The district has 5 sub-counties (Figure 1). The district was previously characterised by a modified equatorial type climate, and a bi-modal rainfall pattern with peaks in April-May and July-August, but more recently, rainfall patterns have become erratic and unpredictable. The district currently has low vegetation cover as a result of indiscriminate clearing of forested land, and the topography is generally flat and rocky. The interaction of the rainfall patterns and flat rocky topography that could create stagnant pools for mosquito breeding, as well as the low vegetation cover, which might increase human exposure to mosquitoes if they breed near human settlements, raises the likelihood of yellow fever transmission in Ngora. Ngora district has twelve health facilities: one hospital (private not-for-profit), one Health Center (HC) IV (public), six HC IIIs (public) and four HC IIs (3 public and 1 private not-for-profit). The average distance to the nearest health facility in the district is 5 kilometres, with 90% of the population residing within a 5km radius of a health unit [14].
Study population
This investigation was conducted among persons confirmed to have yellow fever, reported between April and July 2024. To identify the confirmed cases, we visited the UVRI sentinel surveillance site in Ngora district and reviewed records of all twenty (20) suspected cases reported within the same period. A suspected case of yellow fever was defined as a resident or visitor of Ngora district who reported onset of unexplained fever (negative for malaria, or non-responsive to malaria treatment, and not explained by any other known reasons) from April to July 2024, plus two or more symptoms such as unexplained jaundice, abdominal pain, or headache that could not be explained by other causes. We defined a confirmed case as a suspected case with laboratory-confirmed detection of yellow fever-specific neutralising antibodies using the plaque reduction neutralisation test. Since confirmed cases were defined based on PRNT, the impact of vaccination on case classification was considered negligible due to the high specificity of PRNT for distinguishing vaccine-induced from naturally acquired neutralising yellow fever antibodies.
Case finding
We reviewed the records corresponding to the samples confirmed for yellow fever at UVRI, to capture the contact and location details of these cases. Where the laboratory records did not provide sufficient information, we reviewed the outpatient records to gather additional details to allow us to locate the confirmed cases. Detailed interviews were then conducted, using standard case investigation forms. The cases were asked about the signs and symptoms they had experienced and their vaccination status. District-level vaccination records for the period March – April 2024 were also reviewed to assess the overall vaccination coverage.
Data analysis
We computed proportions to describe the socio-demographic characteristics of the cases and the signs and symptoms that they had experienced. Data are presented as frequencies and percentages. Statistical analysis was performed using STATA version 16.
Ethical consideration
Approval was granted by the Higher Degrees Research and Ethics Committee of Makerere University School of Public Health (REC No: SPH-2024-591). Verbal informed consent was obtained from all participants. No identifying information on the participants is presented. Consent was sought from a parent(s)/guardian(s) for participants below the age of 18 years. For children, interviews were conducted in the presence of the parent/ guardian. The investigation was carried out in compliance with the Helsinki Declaration.
Results
Socio-demographic characteristics of the cases
Most of the confirmed cases were female (n=4, 80%). Forty per cent (n=2) of the cases were aged 0-12 years, while 80% (n=4) resided within Ngora Town Council as detailed in Table 1. Females had the highest attack rate (4.1 per 100,000) for yellow fever in this outbreak. The highest attack rate was registered in Ngora Town Council, at 18.6 per 100,000 (Table 2).
Case fatality report
The case was a 91-year-old male from Amuria district, located in the Teso sub-region of Eastern Uganda, which shares borders with Ngora district. The case was a visitor in Ngora, where at the time of symptom onset, he was staying at a relative’s home in Komodo cell in Ngora Town Council (TC), Ngora district. Upon developing symptoms on 10th June, 2024, he sought medical care from the district’s sentinel surveillance site, where he was diagnosed with septicemia. This case had never been vaccinated against the yellow fever virus and had been deemed ineligible during the yellow fever mass vaccination campaign in April 2024. He returned for a review on 18th June, 2024, presenting with fever, abdominal pain, jaundice, and general body weakness.
He tested positive for the Hepatitis C virus but negative for Hepatitis B surface antigen (HBsAg). A sample for a yellow fever test was taken on the same day as the review, and he was discharged. Given that he was a visitor in Ngora, he returned to his home in Amuria district one week after the yellow fever sample was taken, but while there, his medical condition worsened. He was taken to a private clinic from where he was referred to Amuria hospital. He died two weeks later, on 5th July 2024. At the time of this outbreak investigation, we were not able to interview this case-patient because he was not available in Ngora district, thus, additional data pertaining to him aside his socio-demographics are not presented in the subsequent sections. The most commonly reported symptoms among the cases were fatigue (75%), muscle and joint pain (75%), headache (75%), vomiting (50%) and diarrhoea (50%) (Figure 2).
Description of the cases
The index case was a 49-year-old female from Agirigiroi village, Kapir sub-county. She was a farmer who spent most of her time around a partly forested and swampy area. She had been vaccinated against yellow fever on 5th April 2024, during the yellow fever mass vaccination campaign. She first experienced symptoms of fever, back pain, headache, fatigue, diarrhoea, and jaundice about a week after having been vaccinated. She sought treatment at a private clinic within Ngora TC. At the clinic, she could not be managed and was referred to the sentinel surveillance site, where she was diagnosed positive for malaria. Given the apparent yellowing of the eyes, a serum sample for a yellow fever test was taken from her and sent to UVRI. The sample returned positive on April 20th 2024.
At the time of this investigation, yellowing of the eyes was still evident. Although the temporal relationship between vaccination and symptom onset raised the possibility of an adverse event after vaccination, case classification was based on lab confirmation using the plaque reduction neutralisation test. Given the high specificity of PRNT for detecting yellow fever-specific neutralising antibodies, the likelihood of misclassification due to recent vaccination was considered minimal.
The second case was a 26-year-old female from Kobuku village, Ngora TC. At the time when she was traced, she was still reporting symptoms of joint pain and fatigue and was advised to go for a repeat yellow fever test. She had also never been vaccinated against yellow fever.
The third case, detected on April 30th, was a 9-year-old girl who reported a history of fever, general body weakness, headache, and at the time of sample collection (prior to the outbreak investigation), she presented with visible yellowing of the eyes, consistent with jaundice, which was still evident up until the time she was traced during the outbreak investigation. This case reported a history of travel to Amuria a few days prior to the onset of clinical signs and symptoms, and had also been previously clinically managed for malaria. However, there is no sufficient evidence to confirm that possible exposure could have come from Amuria district.
The 4th case, who was detected on May 19th 2024 was a 5-year-old girl residing in Komodo cell, Northern ward, Ngora TC. She reported a history of vomiting, abdominal pain, muscle pain, sore throat and jaundice, but no evident bleeding. She was initially taken to a nearby clinic to receive treatment, but she was then referred to the sentinel surveillance site for further management of signs and symptoms. Her laboratory results returned negative for malaria. At the time of this outbreak investigation, jaundice was still evident and therefore, this case was advised to return to the sentinel surveillance site for a repeat yellow fever test. Both the third and fourth cases similarly, had never been vaccinated against yellow fever.
Yellow fever vaccination coverage in Ngora district
Table 3 shows the vaccination coverage in Ngora, by subcounty. This vaccination coverage is based on the whole population of the district. About half of the population in Ngora subcounty (SC) had received the yellow fever vaccine, while in Ngora TC where most of the cases had been reported, vaccination coverage was above the target population, at 112% (24078/21488).
Summary of possible exposures to yellow fever
Table 4 shows that three-quarters (n=3) of the cases had previously been in direct contact with animals, while half (n=2) had travelled outside the home village before becoming ill. Furthermore, environmental observations also revealed that Ngora Town Council, where most of the cases resided, is characterised by a rocky terrain. Monkeys were observed in the trees around these rocks, and they were also seen around the homes of the two cases that were detected on April 30th and May 19th. In general, Ngora district has rocks scattered in most of the sub counties. These rocks are habitats for non-human primates which are the primary wild hosts of the yellow fever virus.
Discussion
This investigation found that most of the reported cases were from one subcounty and had never been vaccinated against yellow fever. Findings from our environmental observations and community interactions also revealed the presence of, and interaction with monkeys around residential areas, and breeding sites (stagnant pools of water in rocky terrain) for mosquitoes.
We found that most persons with a lab confirmation for yellow fever were female, and these had the highest attack rate. We could attribute the observed findings among females to cultural and socio-economic roles that influence risk levels. Females may have more frequent interactions with environments such as agricultural fields, or engage in activities that increase their exposure to mosquito bites [15]. Another plausible explanation could be health-seeking behaviour and access to health care among females. In many settings, women have better health-seeking behaviour and often access healthcare more frequently than men, which could increase the likelihood of case detection among them [16]. As such, it could be possible that while we did not find and/or interact with male cases to allow for meaningful comparisons, it can still reasonably be assumed that the transmission of yellow fever could occur among both genders, given similar exposures to agricultural fields and forested environments. Nonetheless, gendered considerations should be important when designing targeted prevention and control strategies against yellow fever, to address any unique exposure vulnerabilities of women in high-risk populations.
Our finding that apparent yellowing of eyes was still evident among three of the cases at the time of conducting the investigation is concerning. Although the cases may have received some prior treatment, there might have been missed opportunities for adequate supportive care and optimal case management [17, 18]. Timely outbreak response and access to supportive care for affected individuals should remain a priority consideration.
We found that three of the four cases reported a history of contact with animals, including monkeys. Qualitative insights also described ongoing human-monkey interactions within the area. This finding is similar to earlier studies investigating past yellow fever outbreaks in Uganda which found that the presence of sylvatic monkeys was a positive predictor for the risk of yellow fever transmission among the population [8, 19]. These findings closely align with known transmission dynamics for yellow fever, given that monkeys are natural hosts for flaviviruses, and thus, close contact with these animals can lead to human infection [20]. Our findings underscore the need for targeted behavior change strategies such as awareness campaigns about the risks of being in contact with wild monkeys.
Furthermore, our findings revealed that the vaccination coverage in the district was sub-optimal. Ngora TC had the highest vaccination coverage. It is however surprising that most of the cases originated from Ngora TC. This is possible, first, because these cases had never been vaccinated. Secondly, the particularly high attack rate in this subcounty could indicate a localised hotspot, suggesting some spatial clustering of risk factors related to environmental and ecological conditions that favour yellow fever transmission such as presence of monkeys, high mosquito density, frequent human-mosquito contact through agricultural activities, forest exposures, poor vector control within the area – all of which point towards possible intermediate transmission. Findings could therefore imply that even when there was a high vaccination coverage within Ngora TC, a small susceptible population that was missed during vaccination could still be affected, leading to a likely surge in cases. Collectively, this implies the need to implement strategies such as targeted vaccination in high-risk areas or supplementary immunization activities targeting unvaccinated individuals, vector control and monitoring, risk communication and community engagement or one health cross-sectoral coordination to guide preparedness and response.
Relatedly, a history of travel among half of the cases before the onset of clinical signs and symptoms of yellow fever was reported. Travel and/or frequent population movements are an important risk factor for onward amplification of the yellow fever virus, as it is introduced into crowded areas [21]. The impact is more pronounced in populations without herd immunity [21]. Although our findings do not necessarily point towards a cause-and-effect relationship, they underscore the need to strengthen surveillance and preparedness efforts in areas with frequent population movements, particularly those that border outbreak-prone districts or which have varying levels of vaccination coverage.
The observed finding that Ngora district is characterised by a rocky terrain indicates a likelihood that there could be stagnant pools of water that may be breeding sites for mosquitoes. Previous studies have indicated that stagnant pools of water play significant roles in the transmission of arboviral infections from vectors such as aedes mosquitoes [22, 23]. In our study, it is possible that the presence of stagnant pools contributed to increased mosquito breeding, resulting in higher mosquito density to facilitate transmission of yellow fever, especially among the unvaccinated population. Therefore, targeted integrated vector control strategies are urgently warranted to mitigate the risk for outbreak re-emergence.
This study had some strengths and limitations. First, our sample size was very small, and thus our study was not well-powered to detect any meaningful associations. It is for this reason that we were not able to perform a case control study which we believe could have established better conclusions around exposure. Secondly, at the time of conducting this investigation, we used only facility records to trace cases, but there could have been other suspected cases that we could have missed who were not captured at the facility. Our findings may therefore not necessarily be an accurate representation of the population characteristics, and thus ought to be interpreted with caution. Thirdly, given that we had a limited number of cases, we were not able to determine which type of outbreak this was. Additionally, we were not able to perform entomological assessments which could have revealed specific vector presence to confirm yellow fever transmission. These limitations notwithstanding, our study’s strengths are that the findings closely explain why the outbreak could have happened, even when there was high vaccination coverage in Ngora TC. Collectively, our findings underscore the need for targeted control interventions aimed at containing emergence and re-emergence of outbreaks of yellow fever across the country.
Conclusion
Our findings may potentially link this yellow fever outbreak to a zoonotic spillover, largely because three-quarters of the cases had previously been in contact with animals. However, this needs to be interpreted with caution since other factors such as presence of mosquito vectors in the community could also have contributed to this outbreak, particularly in the absence of entomological assessments in this study. Therefore, there is a need for targeted behavior change strategies such as risk communication and community engagement on yellow fever exposures among high-risk populations especially where vaccination coverage is low. Integrated vector control interventions aimed at preventing and controlling the re-emergence of future yellow fever outbreaks across the country are also necessary.
What is already known about the topic
- Yellow fever is endemic in Uganda, with recurrent outbreaks reported.
- It has an epidemic alert threshold of one case.
What this study adds
- This study shows that this yellow fever outbreak was likely due to a zoonotic spillover.
- The study underscores the need for targeted one health interventions such as integrated vector control, supplementary immunization activities, risk communication and behaviour change strategies
Acknowledgements
We acknowledge the support of the Africa Field Epidemiology Network (AFENET). We would like to thank the Ngora District Health Office, as well as the district sentinel surveillance site for their support in investigating the yellow fever outbreak. We also thank all the cases for their participation. We acknowledge Ms Angela Kisakye and Dr Chukwuma David Umeokonkwo for their review of the initial drafts.
Authors´ contributions
AM wrote the first draft of the manuscript, contributed to methodology design and performed formal analysis. CK, RM, PN, IN, MGA contributed to methodology design, editing the manuscript. CN, WHT, BL, HK and SNK supervised, reviewed and edited the manuscript for substantial intellectual content. All authors read and approved the final version prior to submission.
| Variable | Frequency (N=5) | Percentage |
|---|---|---|
| Sex | ||
| Female | 4 | 80.0 |
| Male | 1 | 20.0 |
| Age | ||
| 0-12 years | 2 | 40.0 |
| 13-17 years | 0 | 0.0 |
| 18-34 years | 1 | 20.0 |
| 35+ years | 2 | 40.0 |
| Subcounty of residence | ||
| Kapir | 1 | 20.0 |
| Ngora Town Council | 4 | 80.0 |
| Note: N = total number of cases (5). Percentages may not sum to 100 due to rounding. | ||
| Variable | Population at risk | Number of cases | *Attack rate/100,000 |
|---|---|---|---|
| Sex | |||
| Female | 97,038 | 4 | 4.1 |
| Male | 116,739 | 1 | 0.9 |
| Subcounty of residence | |||
| Kapir | 29,658 | 1 | 3.4 |
| Ngora Town Council | 21,488 | 4 | 18.6 |
| *Attack rates calculated per 100,000 population. Population at risk based on subcounty estimates. | |||
| Sub county | Target population | Number vaccinated | Coverage (%) |
|---|---|---|---|
| Ngora SC | 21,027 | 11,200 | 53.3 |
| Ngora Town council | 21,488 | 24,078 | 112.1* |
| Kapir | 29,658 | 17,121 | 57.7 |
| Kobuin | 20,775 | 14,159 | 68.2 |
| Mukura | 24,477 | 17,383 | 71.0 |
| *Coverage exceeding 100% may indicate inclusion of individuals from outside the target population or population movement. | |||
| Variable | Frequency (N=4) | Percentage |
|---|---|---|
| Attended funeral before becoming ill | ||
| Yes | 2 | 50.0 |
| No | 2 | 50.0 |
| Travelled outside home village before becoming ill | ||
| Yes | 2 | 50.0 |
| No | 2 | 50.0 |
| Patient hospitalized before illness | ||
| Yes | 1 | 25.0 |
| No | 3 | 75.0 |
| Direct contact with animals | ||
| Yes | 3 | 75.0 |
| No | 1 | 25.0 |
| Note: N = total number of cases (4). Percentages may not sum to 100 due to rounding. | ||


References
- WHO. Yellow fever [Internet]. Geneva (Switzerland): World Health Organization; 2025 Oct 20 [cited 2026 Apr 23]. Available from: https://www.who.int/news-room/fact-sheets/detail/yellow-fever
- Lindsey NP, Horton J, Barrett ADT, Demanou M, Monath TP, Tomori O, Van Herp M, Zeller H, Fall IS, Cibrelus L, Erin Staples J. Yellow fever resurgence: An avoidable crisis? npj Vaccines [Internet]. 2022 Nov 2 [cited 2026 Apr 23];7(1):137. doi:10.1038/s41541-022-00552-3 Available from: https://www.nature.com/articles/s41541-022-00552-3
- Gabiane G, Yen P, Failloux A. Aedes mosquitoes in the emerging threat of urban yellow fever transmission. Reviews in Medical Virology [Internet]. 2022 Feb 6 [cited 2026 Apr 23];32(4):e2333. doi:10.1002/rmv.2333 Available from: https://onlinelibrary.wiley.com/doi/10.1002/rmv.2333
- WHO. EYE strategy [Internet]. Geneva (Switzerland): World Health Organization; c2026 [cited 2026 Apr 23]. Available from: https://www.who.int/initiatives/eye-strategy
- Reno E, Quan NG, Franco-Paredes C, Chastain DB, Chauhan L, Rodriguez-Morales AJ, Henao-Martínez AF. Prevention of yellow fever in travellers: an update. The Lancet Infectious Diseases [Internet]. 2020 May 7 [cited 2026 Apr 23];20(6):e129–37. doi:10.1016/S1473-3099(20)30170-5 Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30170-5/abstract
- Mboussou F, Ndumbi P, Ngom R, Kassamali Z, Ogundiran O, Van Beek J, Williams G, Okot C, Hamblion EL, Impouma B. Infectious disease outbreaks in the African region: overview of events reported to the World Health Organization in 2018. Epidemiol Infect [Internet]. 2019 Nov 11 [cited 2026 Apr 23];147:e299. doi:10.1017/S0950268819001912 Available from: https://www.cambridge.org/core/journals/epidemiology-and-infection/article/infectious-disease-outbreaks-in-the-african-region-overview-of-events-reported-to-the-world-health-organization-in-2018/A504999B25CBA5E179B951FABB8CFE53
- WHO. Yellow fever – Uganda [Internet]. Geneva (Switzerland): World Health Organization; 2022 Apr 25 [cited 2026 Apr 23]. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON367
- Kwagonza L, Masiira B, Kyobe-Bosa H, Kadobera D, Atuheire EB, Lubwama B, Kagirita A, Katushabe E, Kayiwa JT, Lutwama JJ, Ojwang JC, Makumbi I, Ario AR, Borchert J, Zhu BP. Outbreak of yellow fever in central and southwestern Uganda, February–May 2016. BMC Infect Dis [Internet]. 2018 Nov 3 [cited 2026 Apr 23];18(1):548. doi:10.1186/s12879-018-3440-y Available from: https://link.springer.com/article/10.1186/s12879-018-3440-y
- Wamala JF, Malimbo M, Okot CL, Atai-Omoruto AD, Tenywa E, Miller JR, Balinandi S, Shoemaker T, Oyoo C, Omony EO, Kagirita A, Musenero MM, Makumbi I, Nanyunja M, Lutwama JJ, Downing R, Mbonye AK. Epidemiological and laboratory characterization of a yellow fever outbreak in northern Uganda, October 2010–January 2011. International Journal of Infectious Diseases [Internet]. 2012 May 10 [cited 2026 Apr 23];16(7):e536–42. doi:10.1016/j.ijid.2012.03.004 Available from: https://www.ijidonline.com/article/S1201-9712(12)00116-6/fulltext
- Baba MM, Ikusemoran M. Is the absence or intermittent YF vaccination the major contributor to its persistent outbreaks in eastern Africa? Biochemical and Biophysical Research Communications [Internet]. 2017 Jan 18 [cited 2026 Apr 23];492(4):548–57. doi:10.1016/j.bbrc.2017.01.079 Available from: https://www.sciencedirect.com/science/article/abs/pii/S0006291X17301298
- Mensah EA, Gyasi SO, Nsubuga F, Alali WQ. A proposed One Health approach to control yellow fever outbreaks in Uganda. One Health Outlook [Internet]. 2024 May 23 [cited 2026 Apr 23];6(1):9. doi:10.1186/s42522-024-00103-x Available from: https://link.springer.com/article/10.1186/s42522-024-00103-x
- Simon S, Amaku M, Massad E. Effects of migration rates and vaccination on the spread of yellow fever in Latin American communities. Revista Panamericana de Salud Pública [Internet]. 2023 Jun 1 [cited 2026 Apr 23];47:e86. doi:10.26633/RPSP.2023.86 Available from: https://iris.paho.org/items/259005c6-f2da-49b8-89e2-9ad586b4fe1b
- Ribeiro GS, Hamer GL, Diallo M, Kitron U, Ko AI, Weaver SC. Influence of herd immunity in the cyclical nature of arboviruses. Current Opinion in Virology [Internet]. 2020 Mar 17 [cited 2026 Apr 23];40:1–10. doi:10.1016/j.coviro.2020.02.004 Available from: https://www.sciencedirect.com/science/article/abs/pii/S1879625720300055
- UBOS. National Population and Housing Census 2024: Preliminary Results [Internet]. Kampala (Uganda): Uganda Bureau of Statistics; 2024 Jun [cited 2026 Apr 23]. 43 p. Available from: https://www.ubos.org/wp-content/uploads/publications/National-Population-and-Housing-Census-2024-Preliminary-Report.pdf
- Zahid MH, Van Wyk H, Morrison AC, Coloma J, Lee GO, Cevallos V, Ponce P, Eisenberg JNS. The biting rate of Aedes aegypti and its variability: A systematic review (1970–2022). Hol F, editor. PLoS Negl Trop Dis [Internet]. 2023 Aug 8 [cited 2026 Apr 23];17(8):e0010831. doi:10.1371/journal.pntd.0010831 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0010831
- Thompson AE, Anisimowicz Y, Miedema B, Hogg W, Wodchis WP, Aubrey-Bassler K. The influence of gender and other patient characteristics on health care-seeking behaviour: a QUALICOPC study. BMC Fam Pract [Internet]. 2016 Mar 31 [cited 2026 Apr 23];17(1):38. doi:10.1186/s12875-016-0440-0 Available from: https://link.springer.com/article/10.1186/s12875-016-0440-0
- Bosa HK, Majwala R, Nakiire L, Ario AR, Kiwanuka N, Kibuuka H, Downing RG, Lutwama JJ. Missed opportunities for Yellow Fever Surveillance in Uganda, July 2015 – May 2016. International Journal of Infectious Diseases [Internet]. 2016 Dec [cited 2026 Apr 23];53 Suppl:116–7. doi:10.1016/j.ijid.2016.11.291 Available from: https://www.ijidonline.com/article/S1201-9712(16)31509-0/fulltext
- Huebl L, Nnyombi A, Kihumuro A, Lukwago D, Walakira E, Kutalek R. Perceptions of yellow fever emergency mass vaccinations among vulnerable groups in Uganda: A qualitative study. Messer WB, editor. PLoS Negl Trop Dis [Internet]. 2024 May 13 [cited 2026 Apr 23];18(5):e0012173. doi:10.1371/journal.pntd.0012173 Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0012173
- Nabukenya I, Ssendagire S, Lubwama B, Aliddeki MD, Kyazze S, Makumbi I, Namusisi O. Investigation and response to Rift Valley Fever and Yellow Fever outbreaks in humans in Uganda, 2016. International Journal of Infectious Diseases [Internet]. 2016 Dec [cited 2026 Apr 23];53 Suppl:129. doi:10.1016/j.ijid.2016.11.319 Available from: https://www.ijidonline.com/article/S1201-9712(16)31537-5/fulltext
- Faria NR, Kraemer MUG, Hill SC, Goes De Jesus J, Aguiar RS, Iani FCM, Xavier J, Quick J, Du Plessis L, Dellicour S, Thézé J, Carvalho RDO, Baele G, Wu CH, Silveira PP, Arruda MB, Pereira MA, Pereira GC, Lourenço J, Obolski U, Abade L, Vasylyeva TI, Giovanetti M, Yi D, Weiss DJ, Wint GRW, Shearer FM, Funk S, Nikolay B, Fonseca V, Adelino TER, Oliveira MAA, Silva MVF, Sacchetto L, Figueiredo PO, Rezende IM, Mello EM, Said RFC, Santos DA, Ferraz ML, Brito MG, Santana LF, Menezes MT, Brindeiro RM, Tanuri A, Dos Santos FCP, Cunha MS, Nogueira JS, Rocco IM, Da Costa AC, Komninakis SCV, Azevedo V, Chieppe AO, Araujo ESM, Mendonça MCL, Dos Santos CC, Dos Santos CD, Mares-Guia AM, Nogueira RMR, Sequeira PC, Abreu RG, Garcia MHO, Abreu AL, Okumoto O, Kroon EG, De Albuquerque CFC, Lewandowski K, Pullan ST, Carroll M, De Oliveira T, Sabino EC, Souza RP, Suchard MA, Lemey P, Trindade GS, Drumond BP, Filippis AMB, Loman NJ, Cauchemez S, Alcantara LCJ, Pybus OG. Genomic and epidemiological monitoring of yellow fever virus transmission potential. Science [Internet]. 2018 Aug 23 [cited 2026 Apr 23];361(6405):894–9. doi:10.1126/science.aat7115 Available from: https://www.science.org/doi/10.1126/science.aat7115
- WHO. Disease Outbreak News: Yellow Fever – Uganda [Internet]. Geneva (Switzerland): World Health Organization; 2022 Apr 25 [cited 2026 Apr 23]. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON367
- Ferede G, Tiruneh M, Abate E, Kassa WJ, Wondimeneh Y, Damtie D, Tessema B. Distribution and larval breeding habitats of Aedes mosquito species in residential areas of northwest Ethiopia. Epidemiol Health [Internet]. 2018 Apr 23 [cited 2026 Apr 23];40:e2018015. doi:10.4178/epih.e2018015 Available from: https://www.e-epih.org/journal/view.php?doi=10.4178/epih.e2018015
- LaBeaud AD, Sutherland LJ, Muiruri S, Muchiri EM, Gray LR, Zimmerman PA, Hise AG, King CH. Arbovirus Prevalence in Mosquitoes, Kenya. Emerg Infect Dis [Internet]. 2011 Feb [cited 2026 Apr 23];17(2):233–41. doi:10.3201/eid1702.091666 Available from: https://wwwnc.cdc.gov/eid/article/17/2/09-1666_article
